- Safety & Recalls
- Regulatory Updates
- Drug Coverage
- COPD
- Cardiovascular
- Obstetrics-Gynecology & Women's Health
- Ophthalmology
- Clinical Pharmacology
- Pediatrics
- Urology
- Pharmacy
- Idiopathic Pulmonary Fibrosis
- Diabetes and Endocrinology
- Allergy, Immunology, and ENT
- Musculoskeletal/Rheumatology
- Respiratory
- Psychiatry and Behavioral Health
- Dermatology
- Oncology
Aetna Updates Standard Formulary for the Fourth Quarter
Effective Oct. 1, 2021, Aetna has added several cancer drugs to its formulary and has updated exclusions and nonpreferred products.
Aetna has made several changes to its standard formulary effective Oct. 1, 2021.
Two cancer therapies have been added to the formulary and one cancer therapy has been moved from the nonpreferred tier to the preferred tier. Additions include two AstraZeneca products: Calquence (acalabrutinib) and Koselugo (selumetinib).
Calquence is approved to treat advanced mantle cell lymphoma and chronic lymphocytic leukemia. Koselugo was approved by the FDA in April 2020 to treat a rare disease that affects children. It treats patients 2 years of age and older with neurofibromatosis type 1 (NF1), a genetic disorder of the nervous system causing tumors to grow on nerves. Koselugo, a kinase inhibitor, is the first drug approved by the FDA to treat this progressive disease.
NF1 affects about 1 out of every 3,000 infants. Between 30% and 50% of patients born with NF1 develop one or more plexiform neurofibromas, which are tumors involving the nerve sheaths.
AstraZeneca’s Tagrisso (osimertinib) has moved from nonpreferred status to Aetna’s preferred tier. Tagrisso is a tyrosine kinase inhibitor that is approved as both adjuvant and first-line treatment metastatic nonsmall cell lung cancers whose tumors have EGFR exon 19 deletions or exon 21 L858R mutations. It was also approved in December 2020 for patients with early stage EGFR mutated long cancer.
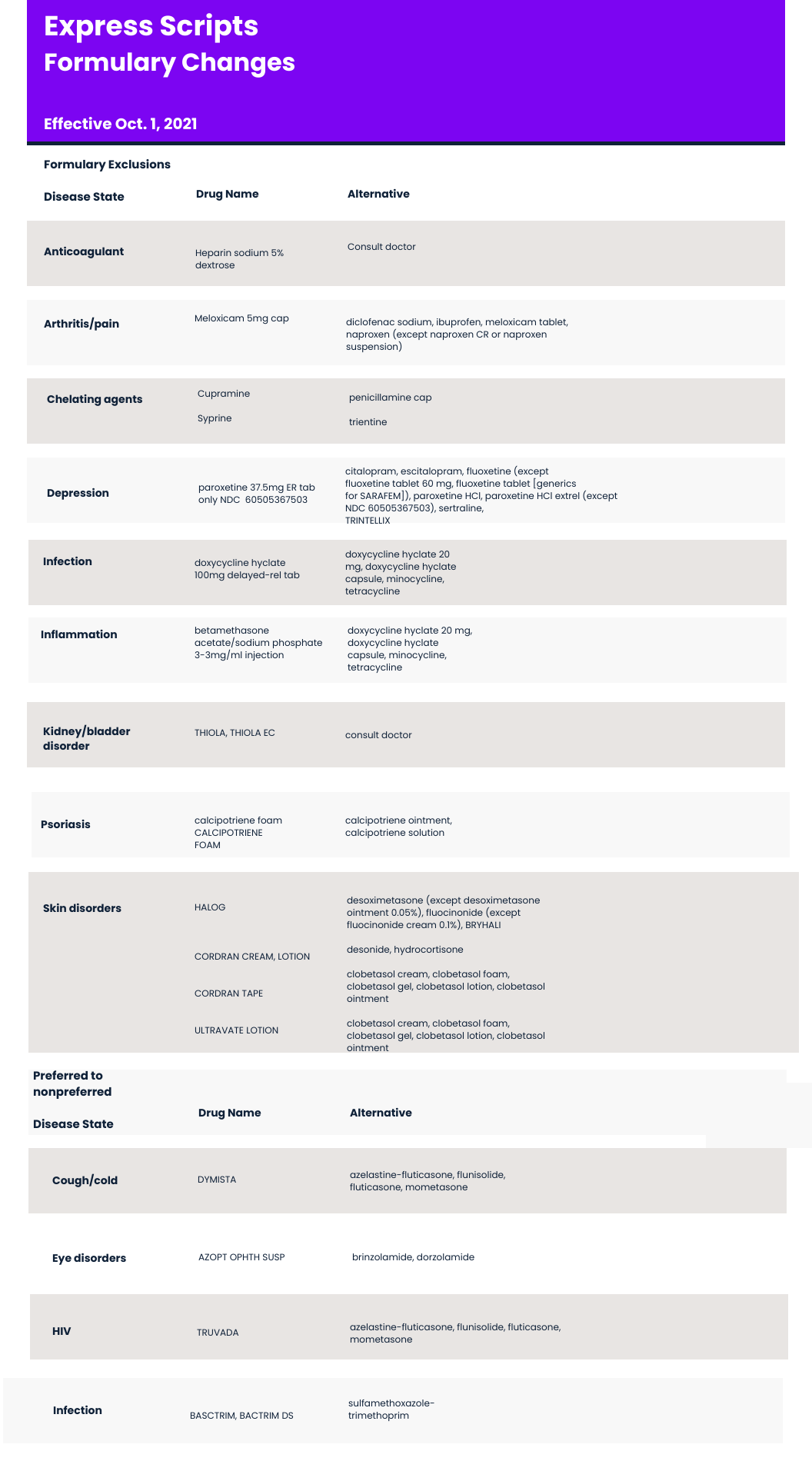
Formulary Exclusions and Nonpreferred
Several therapies have been excluded from Aetna’s standard formulary, including several therapies to treat psoriasis and skin disorders. Halog (halcinonide), Cordran (flurandrenolide), Ultravate (halobetasol) have been excluded. Aetna recommends desoximetasone, desonide, and clobetasol as alternatives.
Also excluded is Thiola and Thiola EC (tiopronin) from Travere Therapeutics (formerly Retrophin). Thiola is used to prevent cystine kidney stone formation in adults and children.
Aetna has also moved Truvuda (emtricitabine/tenofovir disoproxil fumarate) from preferred to nonpreferred status. Truvuda, manufactured by Gilead, is approved for pre-exposure prophylaxis (PrEP) for the prevention of HIV.
A complete list of changes to Aetna’s formulary exclusions and nonpreferred is below.
Payers Recognize the Benefits, but Still See Weight Loss Drugs through a Cost Lens
April 12th 2024Jeffrey Casberg, M.S., R.Ph., a senior vice president of clinical pharmacy at IPD Analytics LLC, a drug intelligence firm that advises payers and pharmaceutical companies, talks about how payers are thinking about weight-loss drugs.
Humira Biosimilars Have a Slow Uptake, Finds Samsung Bioepis Report
April 8th 2024Caps on Medicare Part D cost sharing as a result of the Inflation Reduction Act, could reduce members’ financial incentive for switching to a biosimilar, suggests the newest Samsung Bioepis Quarterly Biosimilar Market Report.
