- Safety & Recalls
- Regulatory Updates
- Drug Coverage
- COPD
- Cardiovascular
- Obstetrics-Gynecology & Women's Health
- Ophthalmology
- Clinical Pharmacology
- Pediatrics
- Urology
- Pharmacy
- Idiopathic Pulmonary Fibrosis
- Diabetes and Endocrinology
- Allergy, Immunology, and ENT
- Musculoskeletal/Rheumatology
- Respiratory
- Psychiatry and Behavioral Health
- Dermatology
- Oncology
Cost-effectiveness of 4 empiric antimicrobial regimens in patients with community-acquired pneumonia
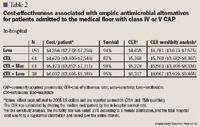
This study compares the cost-effectiveness of the 4 most common empiric antimicrobial regimens used for the treatment of adults with community-acquired pneumonia (CAP) at a community health system during a 6-month period. Associations between initial antimicrobials and total hospital costs were determined. Cost-effectiveness ratios were determined by dividing the total hospital costs by the percent survival. A total of 415 patients met criteria for the Pneumonia Severity Index (PSI) risk class IV or V. Costs (adjusted for inflation) were as follows (median, 25th and 75th percentile): total hospital costs ($5,078 [$3,218–$8,144]), pharmacy costs ($753 [$455–$1,357]), and antibiotic costs ($139 [$82–$229]). The most favorable cost-effectiveness ratio was observed for patients who received levofloxacin monotherapy ($4,635 per life saved), followed by ceftriaxone plus a macrolide ($5,278), ceftriaxone monotherapy ($5,368), and ceftriaxone plus levofloxacin ($6,317).
Abstract
This study compares the cost-effectiveness of the 4 most common empiric antimicrobial regimens used for the treatment of adults with community-acquired pneumonia (CAP) at a community health system during a 6-month period. Associations between initial antimicrobials and total hospital costs were determined. Cost-effectiveness ratios were determined by dividing the total hospital costs by the percent survival. A total of 415 patients met criteria for the Pneumonia Severity Index (PSI) risk class IV or V. Costs (adjusted for inflation) were as follows (median, 25th and 75th percentile): total hospital costs ($5,078 [$3,218–$8,144]), pharmacy costs ($753 [$455–$1,357]), and antibiotic costs ($139 [$82–$229]). The most favorable cost-effectiveness ratio was observed for patients who received levofloxacin monotherapy ($4,635 per life saved), followed by ceftriaxone plus a macrolide ($5,278), ceftriaxone monotherapy ($5,368), and ceftriaxone plus levofloxacin ($6,317). Among patients admitted to the medical floor for class IV or V CAP, empiric levofloxacin monotherapy was associated with greater cost-effectiveness than ceftriaxone monotherapy, ceftriaxone plus a macrolide, or ceftriaxone plus levofloxacin. (Formulary. 2005;40:298–303.)
Expenditures for community-acquired pneumonia (CAP) exceed $8 billion annually in the United States.1 Outpatient care is relatively inexpensive ($150–$350/patient), whereas costs increase greatly when patients are admitted to the hospital ($5,785/patient).1–3 Although less than one-quarter of CAP patients are hospitalized, these patients consume approximately 90% of all resources used for the treatment of CAP.1 Furthermore, the cost of care increases exponentially for patients admitted to the intensive care unit (ICU) ($21,144/patient).2 The fact that CAP imposes such a huge financial burden on society warrants careful attention to factors that impact cost of care.
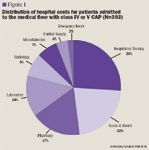
This study evaluated the cost of care for CAP patients admitted to the medical floor with PSI class IV or V disease.14 Risk classes IV and V were chosen because national recommendations state that these patients should be admitted to the hospital. After identifying patients who fell into these risk classes, the proportion of costs attributable to multiple aspects of patient care was determined. Then, a cost-effectiveness analysis was conducted to evaluate the cost-effectiveness of the 4 most common empiric antimicrobial regimens used within a private healthcare system (ie, levofloxacin monotherapy, ceftriaxone monotherapy, ceftriaxone plus a macrolide, and ceftriaxone plus levofloxacin).
METHODS
Prior to study initiation, approvals were obtained from the Institutional Review Board of the Baptist Health System and The University of Texas Health Science Center at San Antonio. All adult CAP patients admitted to Baptist Health System from November 1, 1999, to April 30, 2000, with a principal discharge diagnosis code (International Classification of Diseases, 9th Revision) of 481–484 and 486 were identified by the Department of Medical Records. Patients were excluded if they had a history of HIV or AIDS, were hospitalized within the previous 7 days, or were immunocompromised secondary to chemotherapy or solid organ transplant. Trained clinical pharmacists used a standardized data collection form to extract data from the medical record pertaining to patient demographics, past medical history, physical exam findings, laboratory values, antimicrobial start and stop dates, admission and discharge dates, discharge disposition, and in-hospital mortality. The clinical information was used to calculate a score for severity of illness according to the PSI.14 The PSI score was used to assign each patient to 1 of 5 PSI risk classes. Only patients managed on the medical floor who met criteria for risk class IV or V were evaluated in this analysis. Patients were stratified into 4 groups (ie, levofloxacin monotherapy, ceftriaxone monotherapy, ceftriaxone plus a macrolide, and ceftriaxone plus levofloxacin) based upon the antimicrobials received in the first 24 hours of hospitalization. These 4 groups were chosen because they were the 4 most common empiric antibiotic regimens used at the institution. Collectively, these 4 regimens were administered to 75% of class IV and V CAP patients.
The perspective of the cost-effectiveness analysis was the institution. Information on hospital charges was obtained from the billing department. All charges mentioned in this article have been adjusted to represent 2005 US dollars by multiplying the charges by the Consumer Price Index (CPI) for medical care from the US Department of Labor, Bureau of Labor Statistics. The CPI for 2005 was based on the "seasonally adjusted annual rate" through June 2005. Total hospital charges reflected the sum of charges from several different areas (eg, room and board, respiratory therapy, pharmacy, laboratory, radiology, central supply, emergency room, and miscellaneous). Patients were charged per episode of treatment by the respiratory department and the charge included the cost of some respiratory medication (eg, albuterol) but not antibiotics. Total hospital costs and pharmacy costs were derived by multiplying the total charges by the hospital cost-to-charge ratio. For antimicrobial costs, the actual cost per dose was obtained from the Department of Pharmacy. The cost per dose was multiplied by the number of dosage forms received during hospitalization.
Statistical analysis. All statistical comparisons were performed with JMP 5.0.1. Significance was defined as an alpha level less than 0.05. Costs were transformed with the natural logarithm function and tested with the Shapiro-Wilk test to ensure normality for total hospital costs (P=.9897), pharmacy costs (P=.9923), and antibiotic costs (P=.9658). Due to the relatively brief period of care, discounting was not performed. Baseline characteristics were compared using Chi-square for discrete variables. Log-transformation of patient age did not result in a normal distribution (P<.0001); therefore, Kruskal Wallis one-way ANOVA was used for age comparisons. One-way ANOVA was used to evaluate associations between antimicrobials received (independent variable) and the natural logarithm of total hospital cost (dependent variable).

RESULTS
A total of 649 patients were managed on the medical floor. Overall, 415 of 649 (64%) were stratified to risk class IV (65%) or V (35%). Median (25th and 75th percentiles) patient age was 80 (73–87) years and 52% of patients were female. Most patients were Caucasian (56%), followed by Latin-American (36%), and African-American (6%). Twenty-four percent of patients were admitted from the nursing home and 21% received antimicrobial therapy prior to admission. Heart failure was the most common comorbid condition (52%), followed by chronic obstructive pulmonary disease (42%), diabetes mellitus (33%), cerebrovascular disease (21%), renal disease (20%), cancer (20%), and liver disease (6%). Information regarding time-to-first-dose of antimicrobials was available for 376 of 415 (92%) of patients. Median (25th and 75th percentiles) time-to-first-dose of antimicrobials was 2.8 (1.6–5.0) hours. Patients received intravenous antimicrobials for 4 (3–7) days and remained hospitalized for 5 (3–7) days. The overall in-hospital mortality rate was 7%. In-hospital mortality was significantly higher among patients in risk class V compared to those in risk class IV (13% vs 4%, P=.0006).
Variables associated with increased cost. African-Americans accrued higher median (25th and 75th percentile) total hospital costs (n=20, $7,337 [$4,462– $15,603]) than Latin-Americans (n=110, $5,149 [$3,062–$7,785], P=.002) or Caucasians (n=174, $4,380 [$2,730–$6,849], P=.001). The cost difference among the different ethnic groups was attributable to significant differences in length of stay rather than severity of illness. The PSI score (median, 25th and 75th percentile) was not statistically different between Caucasians (116 [104–142] points), Latin-Americans (116 [100–132] points), and African-Americans (110 [96–124] points). However, African-Americans remained hospitalized the longest (6 [4–12] d), followed by Latin-Americans (5 [3–8] d), and Caucasians (4 [2–7] d) (P=.0038). The differences in length of stay were not due to age, as Caucasian patients were older (80 y) than Latin-American (77 y) or African-American (75 y) patients. However, African-Americans and Latin-Americans had higher rates of several comorbidities. Respective rates for African-Americans, Latin-Americans, and Caucasians were as follows: diabetes mellitus (46%, 44%, and 25%; P=.0004), stroke (35%, 21%, and 18%; P=.2536), renal disease (38%, 27%, and 13%; P=.0005), and liver disease (0%, 11%, and 4%; P=.0136).

DISCUSSION
In this study, antimicrobial acquisition costs accounted for only 3% of the total hospital costs and 18% of the total pharmacy costs. Nevertheless, antimicrobial choice has been shown to impact total hospital cost for CAP patients in 2 prior studies.4,21 Paladino and colleagues determined that azithromycin mono-therapy was more cost-effective than cefuroxime with or without erythromycin ($5,265 vs $6,145).4 In addition, Dresser and colleagues found that gatifloxacin monotherapy was more cost-effective than ceftriaxone plus a macrolide ($5,236 vs $7,047).21 These studies differed from the present study in that patients were randomized to treatment and the authors determined the costs attributable solely to pneumonia.

While costs were not vastly different ($5,173 vs $4,670), ceftriaxone plus a macrolide was associated with lower mortality than ceftriaxone monotherapy (2% vs 13%). Notably, patients who received ceftriaxone monotherapy were older and were much more likely to be admitted from the nursing home. The mortality rate for patients who received ceftriaxone plus a macrolide was lower than a comparison study,14 but the present study excluded ICU patients, which should have resulted in a lower mortality rate. Furthermore, the lower than expected in-hospital mortality for patients who received ceftriaxone plus a macrolide is potentially attributable to differences in baseline characteristics (eg, fewer patients admitted from a nursing home and fewer patients stratified to risk class V). It is important to recognize that 2 of the regimens evaluated in this study are not endorsed by current guidelines for the empiric selection of antibiotics for CAP (ie, ceftriaxone monotherapy and ceftriaxone plus levofloxacin).13,15,22 These guideline-discordant regimens were associated with poorer cost-effectiveness ratios than guideline-concordant regimens. In addition, the current study provides further reason to avoid the combination of ceftriaxone plus levofloxacin, because this regimen was associated with a very large incremental cost compared with levofloxacin monotherapy.
While antimicrobial choice impacts hospital costs, it is important to realize that antimicrobials account for only a small proportion of the total cost of hospitalization. The major factors that impact cost are respiratory services and room and board charges; therefore, attempts to reduce health-system expenditures should focus on shortening the length of hospital stay. Fortunately, criteria have been developed to assist with intravenous to oral switch and hospital discharge decisions.23,24 Finally, clinicians must consider factors other than cost-effectiveness when choosing an antimicrobial regimen (eg, likely pathogens, prior drug exposure, comorbid diseases, and antimicrobial resistance).25–27
CONCLUSIONS
Among patients admitted to the medical floor for class IV or V CAP, empiric levofloxacin monotherapy was associated with greater cost-effectiveness than ceftriaxone monotherapy, ceftriaxone plus a macrolide, or ceftriaxone plus levofloxacin. Combination therapy with ceftriaxone plus levofloxacin was not associated with improved cost-effectiveness compared to guideline-endorsed regimens; therefore, this antimicrobial regimen should be avoided. Finally, the best way to achieve cost-containment is to reduce the duration of hospitalization.
REFERENCES
1. Niederman MS, McCombs JS, Unger AN, et al. The cost of treating community-acquired pneumonia. Clin Ther. 1998;20:820–837.
2. Angus DC, Marrie TJ, Obrosky DS, et al. Severe community-acquired pneumonia: use of intensive care services and evaluation of American and British Thoracic Society Diagnostic criteria. Am J Respir Crit Care Med. 2002;166:717–723.
3. Lave JR, Fine MJ, Sankey SS, et al. Hospitalized pneumonia. Outcomes, treatment patterns, and costs in urban and rural areas. J Gen Intern Med. 1996;11:415-421.
4. Paladino JA, Gudgel LD, Forrest A, Niederman MS. Cost-effectiveness of IV-to-oral switch therapy: azithromycin vs cefuroxime with or without erythromycin for the treatment of community-acquired pneumonia. Chest. 2002;122: 1271–1279.
5. Castro-Guardiola A, Viejo-Rodriguez AL, Soler-Simon S, et al. Efficacy and safety of oral and early-switch therapy for community-acquired pneumonia: a randomized controlled trial. Am J Med. 2001;111:367–374.
6. Fine MJ, Pratt HM, Obrosky DS, et al. Relation between length of hospital stay and costs of care for patients with community-acquired pneumonia. Am J Med. 2000;109:378–385.
7. Siegel RE, Alicea M, Lee A, Blaiklock R. Comparison of 7 versus 10 days of antibiotic therapy for hospitalized patients with uncomplicated community-acquired pneumonia: a prospective, randomized, double-blind study. Am J Ther. 1999;6:217–222.
8. Rhew DC, Hackner D, Henderson L, et al. The clinical benefit of in-hospital observation in 'low-risk' pneumonia patients after conversion from parenteral to oral antimicrobial therapy. Chest. 1998;113:142–146.
9. Omidvari K, de Boisblanc BP, Karam G, et al. Early transition to oral antibiotic therapy for community-acquired pneumonia: duration of therapy, clinical outcomes, and cost analysis. Respir Med. 1998;92:1032–1039.
10. Siegel RE, Halpern NA, Almenoff PL, et al. A prospective randomized study of inpatient i.v. antibiotics for community-acquired pneumonia. The optimal duration of therapy. Chest. 1996;110:965–971.
11. Ramirez JA. Switch therapy in community-acquired pneumonia. Diagn Microbiol Infect Dis. 1995;22:219–223.
12. Merchant S, Mullins CD, Shih Y-CT. Factors associated with hospitalization costs for patients with community acquired pneumonia. Clin Ther. 2003;25:593–610.
13. Niederman MS, Mandell LA, Anzueto A, et al. Guidelines for the management of adults with community-acquired pneumonia. Diagnosis, assessment of severity, antimicrobial therapy, and prevention. Am J Respir Crit Care Med. 2001;163:1730-1754.
14. Fine MJ, Auble TE, Yealy DM, et al. A prediction rule to identify low-risk patients with community-acquired pneumonia. N Engl J Med. 1997;336:243–250.
15. Bartlett JG, Dowell SF, Mandell LA, et al, for the Infectious Diseases Society of America. Practice guidelines for the management of community-acquired pneumonia in adults. Clin Infect Dis. 2000;31:347–382.
16. Brown RB, Iannini P, Gross P, Kunkel M. Impact of initial antibiotic choice on clinical outcomes in community-acquired pneumonia: analysis of a hospital claims-made database. Chest. 2003;123:1503–1511.
17. Houck PM, MacLehose RF, Niederman MS, Lowery JK. Empiric antibiotic therapy and mortality among medicare pneumonia inpatients in 10 western states: 1993, 1995, and 1997. Chest. 2001;119:1420–1426.
18. Burgess DS, Lewis JS II. Effect of macrolides as part of initial empiric therapy on medical outcomes for hospitalized patients with community-acquired pneumonia. Clin Ther. 2000;22: 872–878.
19. Dudas V, Hopefl A, Jacobs R, Guglielmo BJ. Antimicrobial selection for hospitalized patients with presumed community-acquired pneumonia: a survey of nonteaching US community hospitals. Ann Pharmacother. 2000;34:446–452.
20. Gleason PP, Meehan TP, Fine JM, et al. Associations between initial antimicrobial therapy and medical outcomes for hospitalized elderly patients with pneumonia. Arch Intern Med. 1999;159:2562–2572.
21. Dresser LD, Niederman MS, Paladino JA. Cost-effectiveness of gatifloxacin vs ceftriaxone with a macrolide for the treatment of community-acquired pneumonia. Chest. 2001;119:1439–1448.
22. Heffelfinger JD, Dowell SF, Jorgensen JH, et al. Management of community-acquired pneumonia in the era of pneumococcal resistance: a report from the Drug-Resistant Streptococcus pneumoniae Therapeutic Working Group. Arch Intern Med. 2000;160:1399–1408.
23. Ramirez JA, Srinath L, Ahkee S, et al. Early switch from intravenous to oral cephalosporins in the treatment of hospitalized patients with community-acquired pneumonia. Arch Intern Med. 1995;155:1273–1276.
24. Halm EA, Fine MJ, Marrie TJ, et al. Time to clinical stability in patients hospitalized with community-acquired pneumonia: implications for practice guidelines. JAMA. 1998;279:1452–1457.
25. Nicolau D. Clinical and economic implications of antimicrobial resistance for the management of community-acquired respiratory tract infections. J Antimicrob Chemother. 2002;50(Suppl 1):61–70.
26. McGowan JE Jr. Economic impact of antimicrobial resistance. Emerg Infect Dis. 2001;7:286–292.
27. Howard D, Cordell R, McGowan JE Jr, et al. Measuring the economic costs of antimicrobial resistance in hospital settings: summary of the Centers for Disease Control and Prevention Emory Workshop. Clin Infect Dis. 2001;33: 1573–1578.
Employers Face Barriers With Adopting Biosimilars
March 1st 2022Despite the promise of savings billions of dollars in the United States, adoption of biosimilars has been slow. A roundtable discussion among employers highlighted some of the barriers, including formulary design and drug pricing and rebates.
