- Safety & Recalls
- Regulatory Updates
- Drug Coverage
- COPD
- Cardiovascular
- Obstetrics-Gynecology & Women's Health
- Ophthalmology
- Clinical Pharmacology
- Pediatrics
- Urology
- Pharmacy
- Idiopathic Pulmonary Fibrosis
- Diabetes and Endocrinology
- Allergy, Immunology, and ENT
- Musculoskeletal/Rheumatology
- Respiratory
- Psychiatry and Behavioral Health
- Dermatology
- Oncology
CVS Caremark Updates 2022 Formulary
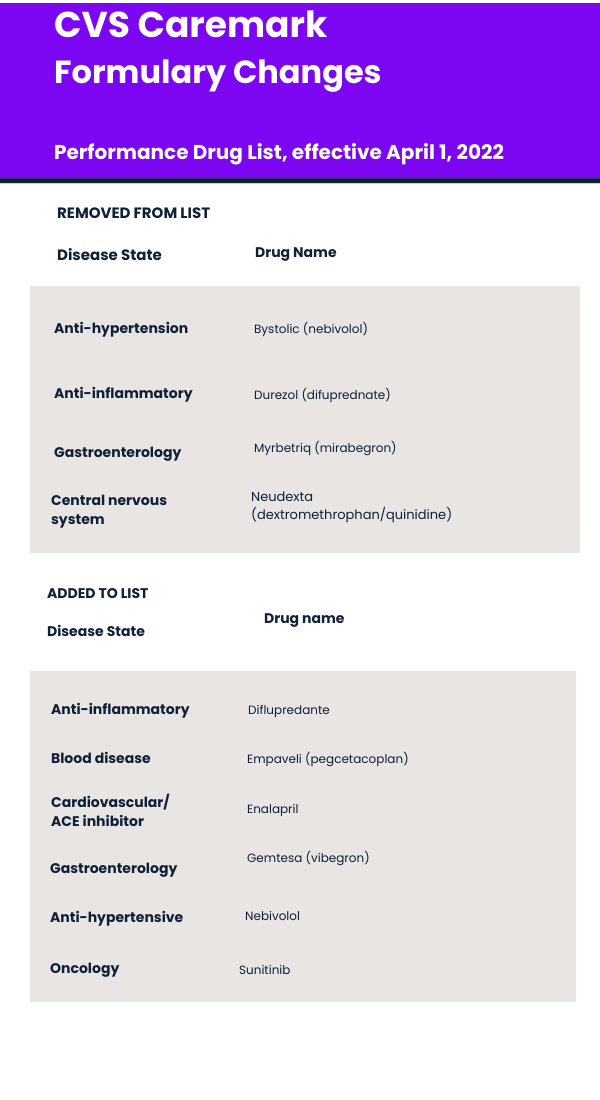
The PBM has removed 20 products and added a half dozen new therapies, including several generics. The changes are effective April 1, 2022.
CVS Caremark has released its Performance Drug List, Standard Control, for April 2022. Effective April 1, the company has removed 20 products from its Standard Control formulary when compared with the list effective from January 2021. CVS Caremark has also added several products, including some branded therapies. (See table below for a list of some of the products added and removed.)
“Our template formularies remain critical tools in helping clients save money as they recover from the economic impacts of COVID-19, while continuing to provide affordable prescription drug benefits to their members,” a CVS Caremark spokesperson said by email.
“It’s important to note that the vast majority — more than 99.6% — of our 100 million CVS Caremark members will be able to stay on their current therapy. We have and will continue to conduct robust, personalized outreach to the small number of members who will be impacted.”
Among those removed are Allergan’s Bystolic, Novartis’ Durezol, Astellas’ Mybretriq, and Avanir Pharmaceuticals’ Neudexta. For three of these therapeutics, generics are now available and are now CVS Caremark’s formulary. Bystolic (nebivolol) is a once-daily beta blocker for the treatment hypertension. Several generics launched in September 2021, including those from Camber/Hetero, Carlisle Medical, Torrent Pharmaceuticals, Ascend Laboratories, and ANI Pharmaceuticals.
Durezol (difluprednate) is a corticosteroid for the treatment of inflammation and pain associated with ocular surgery. The first generic, from Cipla, was approved in August 2021. Another generic from Carlisle Medical followed in September 2021 and a third Amneal followed in November 2021.
Nuedexta (dextromethorphan HBr and quinidine sulfate) was also removed. Nuedexta is a combination product used to treat pseudobulbar affect (PBA), which is characterized by episodes of sudden uncontrollable and inappropriate laughing or crying. It can occur in people who have had strokes, amyotrophic lateral sclerosis, multiple sclerosis, traumatic brain injury, Alzheimer’s and Parkinson’s disease. In one study, PBA symptoms were present in about 17.5% of nursing home residents.
A generic has been approved, but it is not yet available because of a patent dispute. A generic is likely to be available in July 2026. One source suggests possible alternatives include tricyclic antidepressants and selective serotonin reuptake inhibitors.
Also removed effective April 1 is Myrbetriq (mirabegron), which is used to treat overactive bladder. Developed by Astellas, Myrbetriq was also approved by the FDA in March 2021 to treat neurogenic detrusor overactivity (NDO) in children three years and older. It was approved in a new dosage form, an extended-release oral granule.
NDO is a type of bladder dysfunction caused by nerve damage and is marked by uncontrolled bladder contractions that can lead to symptoms of urinary frequency, urgency and incontinence. Eighty-five percent of children with NDO have spina bifida, a congenital spinal cord defect, according to Astellas.
Company officials said alternatives to Myrbetriq include darifenacin ext-rel, oxybutynin ext-rel, solifenacin, tolterodine, tolterodine ext-rel, trospium, trospium ext-rel, Gemtesa, and Toviaz.
The PBM has also added Urovant Science’s Gemtesa (vibegron), which was approved by the FDA in December 2020 for adults with overactive bladder. The therapy was launched in April 2021. Gemtesa is an oral, once-daily tablet containing 75 mg of vibegron, a small-molecule β3 agonist that helps relax the detrusor bladder muscle. It does not require dose titration.
Also added to CVS Caremark’s formulary is Empaveli (pegcetacoplan) injection, developed by Apellis Pharmaceuticals. The FDA approved the therapy in May 2021 for adults with paroxysmal nocturnal hemoglobinuria (PNH). It is approved for use in adults with PNH who are treatment-naïve as well as patients switching from the C5 inhibitors Soliris (eculizumab) and Ultomiris (ravulizumab). It was also approved in December in the European Union with the brand name of Aspaveli.
Payers Recognize the Benefits, but Still See Weight Loss Drugs through a Cost Lens
April 12th 2024Jeffrey Casberg, M.S., R.Ph., a senior vice president of clinical pharmacy at IPD Analytics LLC, a drug intelligence firm that advises payers and pharmaceutical companies, talks about how payers are thinking about weight-loss drugs.
Humira Biosimilars Have a Slow Uptake, Finds Samsung Bioepis Report
April 8th 2024Caps on Medicare Part D cost sharing as a result of the Inflation Reduction Act, could reduce members’ financial incentive for switching to a biosimilar, suggests the newest Samsung Bioepis Quarterly Biosimilar Market Report.
