- Safety & Recalls
- Regulatory Updates
- Drug Coverage
- COPD
- Cardiovascular
- Obstetrics-Gynecology & Women's Health
- Ophthalmology
- Clinical Pharmacology
- Pediatrics
- Urology
- Pharmacy
- Idiopathic Pulmonary Fibrosis
- Diabetes and Endocrinology
- Allergy, Immunology, and ENT
- Musculoskeletal/Rheumatology
- Respiratory
- Psychiatry and Behavioral Health
- Dermatology
- Oncology
Dronedarone for atrial fibrillation: An update of clinical evidence
Atrial fibrillation (AF) is the most common form of arrhythmia, affecting an estimated 2.2 million people in the United States. The goal of treatment is to reduce symptoms through rate or rhythm control and to prevent a cardioembolic event. Dronedarone (pronounced droe'' ne' da rone) is a noniodinated benzofuran derivative with characteristics of all 4 Vaughan-Williams antiarrhythmic classes. A search in clinicaltrials.gov for dronedarone phase 3 studies yielded 5 randomized controlled studies that investigated the efficacy and safety of the drug.

Key Points
Abstract
Atrial fibrillation (AF) is the most common form of arrhythmia, affecting an estimated 2.2 million people in the United States. The goal of treatment is to reduce symptoms through rate or rhythm control and to prevent a cardioembolic event. Dronedarone (pronounced droe'' ne' da rone) is a noniodinated benzofuran derivative with characteristics of all 4 Vaughan-Williams antiarrhythmic classes. A search in http://clinicaltrials.gov for dronedarone phase 3 studies yielded 5 randomized controlled studies that investigated the efficacy and safety of the drug. The ATHENA and EURIDIS/ADONIS clinical trials have shown dronedarone to reduce hospitalizations due to cardiovascular events and AF recurrence, respectively. The ANDROMEDA study was discontinued prematurely due to a significant increase in mortality with dronedarone when compared with placebo, mainly due to progressive heart failure. ERATO showed significant reduction in ventricular rate in patients with permanent AF. DIONYSOS compared dronedarone to amiodarone and demonstrated that amiodarone was more efficacious than dronedarone in preventing AF recurrence but with more premature drug discontinuation. There are 4 ongoing clinical trials, 3 of which have estimated completion dates in 2011. Another ongoing study (PALLAS) will evaluate the efficacy of dronedarone in preventing cardiovascular events, hospitalization, or death in permanent AF patients with additional cardiac risk factors; its estimated completion date is 2013. Although dronedarone has a favorable adverse effect profile when compared with amiodarone, it has a trade off of less efficacy. Its black box warning in heart failure patients is also a limitation. Dronedarone was approved for use by FDA in July 2009 and is considered a possible therapeutic option in AF management. Until long-term efficacy and safety studies are available, the key to dronedarone prescribing is appropriate patient selection to ensure the most effective therapeutic approach. (Formulary. 2010;45:275–283.)
Atrial fibrillation (AF) is the most common form of arrhythmia, accounting for approximately one-third of arrhythmias. An estimated 2.2 million people in the United States have paroxysmal or persistent AF. Prevalence of AF ranges from 0.4% to 1% of the population, increasing with age, such that the prevalence is estimated at 8% by the age of 80 years. The average annual cost to society for AF is estimated at $15.7 billion. The numbers of women and men with AF are about equal, but about 60% of people with AF over the age of 75 are women.1
TREATMENT OF AF
The goal of treatment is to reduce symptoms through ventricular rate control, conversion to sinus rhythm, maintenance of sinus rhythm, and prevention of cardioembolic stroke. Rate control is achieved by inhibiting atrioventricular (AV) nodal conduction. Drugs used in the management of rate control include beta-blockers, non-dihydropyridine calcium-channel blockers (eg, diltiazem, verapamil), digoxin, and amiodarone. Amiodarone is not a preferred first-line agent for rate control, but rather is used when other agents are ineffective or contraindicated. Restoration of sinus rhythm is mainly achieved through direct-current cardioversion or Vaughan-Williams class I and III pharmacologic agents. Maintenance of sinus rhythm may be achieved through pharmacologic agents such as amiodarone, dofetilide, flecainide, propafenone, or sotalol. Prevention of cardioembolic stroke is mainly accomplished with warfarin or aspirin.2
Nonpharmacologic treatment may be implemented when pharmacologic agents are ineffective or when intolerable adverse effects occur. Radiofrequency catheter ablation, such as AV nodal ablation with a permanent pacemaker or pulmonary vein isolation ablation, and surgical-based maze procedure are such interventions. Catheter ablations are minimally invasive, where catheters are threaded through blood vessels to reach the isolated atrial tissue and destroy the abnormal targeted area. AV nodal ablation with pacemaker is a rate-controlling method that disrupts the AV node by preventing electrical impulse transmission from the atria to the ventricles; a permanent pacemaker is implanted in patients to control the ventricular rate. Because AF may occur with this method, chronic anticoagulation medication is still required. Pulmonary vein isolation ablation is a rhythm-controlling method that involves delivery of radiofrequency energy to the targeted cardiac tissue to form scar tissue, thereby preventing abnormal electrical transmission to the rest of the atria.
Complications with catheter ablations have been reported in about 6% of patients, including thromboembolic stroke, pulmonary vein stenosis, and atrioesophageal fistula (rarely, but has high risk of mortality). Unlike catheter ablations, surgical maze procedure requires open heart surgery to create cardiac scar tissue to disrupt the abnormal electrical impulses in AF. Because it requires open heart surgery, this procedure may be considered when other options have failed or if other cardiac surgery is also planned, such as coronary artery bypass graft or mitral valve repair.3
RATE CONTROL VS RHYTHM CONTROL
The decision to use rate versus rhythm control often depends on the individual patient. Current guidelines recommend that rhythm control may be of benefit in young, symptomatic patients without underlying heart disease, whereas rate control may be indicated for old patients with minimal symptoms resulting from AF.1
Randomized controlled clinical trials have studied differences in outcomes based on rate versus rhythm control; the results have not shown statistically significant benefit with either approach.4-8 While no significant differences were found with the primary end points, significant differences were found in secondary end points. For example, patients assigned to rhythm control groups were more likely to be hospitalized and more likely to experience adverse effects associated with the treatment than the rate control groups.5
Limitations to antiarrhythmic agents are often related to their adverse effect profile. The Vaughan-Williams class I drugs (eg, procainamide [IA], flecainide [IC], and propafenone [IC]) are sodium-channel blockers that work by slowing conduction velocity and inhibiting ventricular automaticity. They are effective for conversion to and maintenance of sinus rhythm, but these drugs have been shown to increase mortality in patients with structural heart disease, most likely due to their proarrhythmic effects.9 Vaughan-Williams class III drugs (eg, amiodarone, dofetilide, and ibutilide) work by altering potassium flux, thereby delaying ventricular repolarization and prolonging the refractory period. With the exception of amiodarone, the class III agents are also associated with high risk of proarrhythmia. In addition to risk of proarrhythmia, some of the antiarrhythmic agents have complicated dosage titration schedules, dosage adjustment for renal insufficiency, or the need for in-hospital initiation.1
Amiodarone is considered a broad-spectrum antiarrhythmic with complex electrophysiologic effects. Although its exact mechanism of action is not known, it is classified as a Vaughan-Williams class III agent. Amiodarone also shows weak sodium channel blockade, depresses nodal automaticity and calcium channel blockade.10
Evidence from controlled clinical trials suggests that amiodarone may be more effective than other antiarrhythmic agents in AF management.11,12 Clinical evidence has demonstrated amiodarone to be more effective than sotalol or propafenone in preventing AF recurrence. During the course of the follow-up of 403 patients, 35% in the amiodarone group had AF recurrences, compared with 63% of patients in the sotalol or propafenone group (P<.001).11 Also, patients who received amiodarone experienced longer time to AF recurrence than patients who received sotalol or placebo (487 days, 74 days, and 6 days, respectively; P<.001).12
Major limitations with amiodarone are its adverse effects. While it has minimal proarrhythmic effects, long-term use of amiodarone is associated with serious end-organ toxicities. These include pulmonary fibrosis, thyroid abnormalities, hepatic toxicity, neuropathy, optic neuritis, corneal deposits, and photosensitivity.10
Dronedarone was approved by FDA in 2009 and is indicated to reduce the risk of cardiovascular hospitalizations in patients with paroxysmal or persistent AF or AFL, with a recent episode of AF/AFL and associated cardiovascular risk factors, who are in sinus rhythm or who will be cardioverted.13
CLINICAL PHARMACOLOGY
Dronedarone is a noniodinated benzofuran derivative with characteristics of all 4 Vaughan-Williams antiarrhythmic classes, similar to that of amiodarone. Dronedarone exhibits prolonged PR and QT intervals with dose-related increases. At the recommended dosage, dronedarone does not lower heart rate.13 Specific molecular structural modifications (elimination of the iodine substituents and addition of methane-sulfonamyl group) to dronedarone were established to minimize the non-cardiovascular adverse effects that are often associated with amiodarone, such as thyroid disorders, pulmonary toxicity, and hepatoxicity.14
PHARMACOKINETICS
Dronedarone has low systemic oral bioavailability, which is increased when it is administered with fatty meals. Upon repeated dronedarone administration, steady state concentration is reached within 4 to 8 days. Dronedarone and its active metabolite (N-debutyl metabolite) are highly protein bound (mainly to albumin), with estimated steady state volume of distribution of 1400 L. Dronedarone is highly metabolized by cytochrome P-450 (CYP) 3A isoenzyme. Most of the metabolites (84%) are excreted in the feces and about 6% in the urine. After intravenous administration, plasma clearance of dronedarone ranges from 130 to 150 L/hour. The addition of a methane-sulfonamyl group to dronedarone may contribute to its shorter half-life and lower lipophilicity when compared to amiodarone, leading to less tissue accumulation. The elimination half-life of dronedarone ranges from 13 to 19 hours.13,14
CLINICAL TRIALS
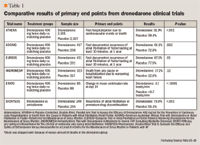
ATHENA. The Effect of Dronedarone on Cardiovascular Events in Atrial Fibrillation trial (ATHENA) was a randomized, placebo-controlled, double-blind, parallel arms study designed to assess whether dronedarone 400-mg twice-daily would reduce the rate of the composite outcome of hospitalizations due to cardiovascular events or death in patients with AF.15
The study enrolled patients with paroxysmal or persistent AF or atrial flutter (AFL) with at least 1 of the following inclusion criteria: age at least 70 years; arterial hypertension; diabetes; previous stroke, transient ischemic attack, or systemic embolism; left atrial diameter greater than or equal to 50 mm on echocardiography; and an ejection fraction <40%. The primary end point was first hospitalization due to cardiovascular events or death from any cause. Secondary outcomes were death from any cause, death from cardiovascular causes, and first hospitalization due to cardiovascular events.
The results showed that 31.9% and 39.4% of patients had a primary outcome event in the dronedarone and placebo groups, respectively (P<.001). This result was mainly attributed to the reduction in first hospitalization due to cardiovascular events (29.3% dronedarone and 36.9% placebo, P<.001). Additionally, deaths from cardiovascular cause were 2.7% in the dronedarone group compared with 3.9% in the placebo group (P=.03).
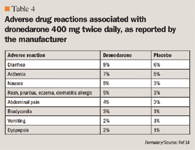
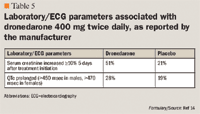
Adverse effects were experienced by 72.0% of patients in the dronedarone group and 69.3% in the placebo group (P=.048). Bradycardia, QT-interval prolongation, diarrhea, nausea, rash, and an increase in serum creatinine level were significantly more common in the dronedarone arm than placebo. Among the 696 patients who discontinued dronedarone prematurely, 12.7% were due to adverse events, compared with 8.1% of the 716 patients who dropped out of the placebo group (P<.001).
EURIDIS/ADONIS. The European Trial in Atrial Fibrillation or Flutter Patients Receiving Dronedarone for the Maintenance of Sinus Rhythm (EURIDIS) and the American–Australian–African Trial with Dronedarone in Atrial Fibrillation or Flutter Patients for the Maintenance of Sinus Rhythm (ADONIS) were both multicenter, randomized, double-blind, placebo-controlled trials. This complementary set of studies hypothesized that dronedarone is superior to placebo in maintaining sinus rhythm after electrical, pharmacologic, or spontaneous conversion from AF or AFL. Adult patients who qualified for inclusion had at least 1 episode of AF as seen on electrocardiography (ECG) in the previous 3 months.16
The primary end point was first documented recurrence of AF lasting at least 10 minutes and confirmed by 2 consecutive ECGs taken 10 minutes apart in 1 year. In ADONIS, the median time to recurrence was 158 days in the dronedarone group and 59 days in the placebo group. One-year recurrence occurred in 61.1% and 72.8% of patients in the dronedarone and placebo arms, respectively (P=.002). In EURIDIS, the median time to recurrence of AF in the dronedarone and placebo groups was 96 days and 41 days, respectively (P=.01). Recurrence at 12 months was 67.1% in the dronedarone group and 77.5% in the placebo group (P=.01). A post hoc analysis from the EURIDIS study showed that 21.2% and 32.0% of patients were hospitalized or died within 12 months in the dronedarone and placebo arms, respectively (P=.02), but no statistical difference was demonstrated from the ADONIS study (24.5% vs 29.8%, respectively; P=.22).16
Adverse effects were similar between both study arms. Elevation of serum creatinine levels was statistically significant between the dronedarone and placebo groups (2.4% and 0.2%, respectively; P=.004). A combined total from both trials of 148 patients in the dronedarone groups and 61 patients in the placebo groups discontinued treatment prematurely, but the reasons for the attrition were not described in the study.
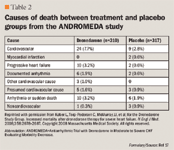
ANDROMEDA. The Antiarrhythmic Trial with Dronedarone in Moderate to Severe CHF Evaluating Morbidity Decrease (ANDROMEDA) was a randomized, double-blind, placebo-controlled trial, which hypothesized that dronedarone would reduce the rate of hospitalization due to HF and possibly also reduce mortality by reducing the incidence of death due to arrhythmia. Adult patients who were hospitalized with new or worsening HF and who had NYHA class III or IV HF or paroxysmal nocturnal dyspnea within a month before admission were included in the study.17
The primary end point was death from any cause or hospitalization for worsening HF. Secondary end points were death from all causes, hospitalization for cardiovascular causes, hospitalization for worsening HF, occurrence of AF or AFL, death from arrhythmia, or sudden death.
ERATO. The Efficacy and Safety of Dronedarone for the Control of Ventricular Rate During Atrial Fibrillation (ERATO) study was a randomized, double-blind, placebo-controlled trial. The objective of ERATO was to assess the efficacy of dronedarone in managing mean 24-hour ventricular rates in patients with permanent AF. Adult patients with documented, symptomatic permanent AF of greater than 6 months' duration were included in this trial.18
The primary end point was change in the mean ventricular rate measured by a 24-hour recording on day 14 compared to day 0. The mean reduction in 24-hour heart rate at day 14 was 11 beats/min in the dronedarone group compared with an increase of 0.7 beats/min in the placebo group (P<.0001). Further, the decrease in heart rate in the dronedarone group was sustained at 4 months when compared to placebo (–10.1 beats/min vs –1.3 beats/min, respectively; P<.001).
Dronedarone was well tolerated during the study. Overall treatment-related adverse effects occurred in 77% of patients in the dronedarone group and 60% of patients in the placebo group. Attrition rates due to adverse events were 15.3% and 10.1% of patients in the dronedarone and placebo groups, respectively.
DIONYSOS. The Efficacy and Safety of Dronedarone Versus Amiodarone for the Maintenance of Sinus Rhythm in Patients with Atrial Fibrillation (DIONYSOS) was a randomized, double-blind trial that compared the efficacy of amiodarone to dronedarone for maintaining sinus rhythm in patients with AF. Study patients were adults with AF lasting more than 72 hours for whom cardioversion and antiarrhythmic therapy were indicated.19
The primary end point was treatment failure, which was defined as recurrence of AF or study drug discontinuation for intolerance or lack of efficacy. Primary end point at 12 months occurred in 75.1% and 58.8% of patients in the dronedarone and amiodarone groups, respectively, (P<.0001). The primary end point result was driven mainly by AF recurrence, which occurred in 63.5% of dronedarone and 42.0% of amiodarone patients. Premature drug discontinuation, due to lack of efficacy or intolerance, occurred in 10.4% and 13.3% of patients in the dronedarone and amiodarone groups, respectively. Amiodarone was associated with greater thyroid abnormalities and neurologic events (sleep disorder and tremor) than dronedarone. Dronedarone was associated with more gastrointestinal adverse events than amiodarone (12.9% vs 5.1%).
ONGOING CLINICAL TRIALS
Four dronedarone clinical studies are currently recruiting patients.20-23 Dronedarone Pattern of Use in Patients Scheduled for Elective Cardioversion (ELECTRA) is a Canadian multi-center study that includes 500 patients with persistent AF who are undergoing elective cardioversion. The primary objective of the trial is to compare rate of AF recurrence within 6 months between treatment and control groups. The treatment group will receive dronedarone, 5 to 7 days prior to electrical cardioversion, and dronedarone continued for 6 months, while control group will receive placebo, 5 to 7 days prior to electrical cardioversion, and dronedarone given for 6 months. This study began November 2009 and is estimated to complete in June 2011.20
The Effect of Addition of Dronedarone to Standard Rate Control Therapy on Ventricular Rate During Persistent Atrial Fibrillation (AFRODITE) study compares the effect of dronedarone added to a standard rate-controlling agent (beta-blocker, calcium-channel blocker, or digoxin) to that of an existing conventional therapy dosage increase on ventricular heart rate after 1 week in patients with persistent AF. The trial has an approximate sample size of 596 patients, and it is estimated to complete in June 2011.21
The Effects of Dronedarone on Atrial Fibrillation Burden in Subjects With Permanent Pacemakers (HESTIA) study is a randomized, multicenter trial to evaluate the effects of dronedarone on AF burden, defined as percent of time in which a patient is in AF, based on electrogram observations in those with a permanent pacemaker. The study was started in July 2010 and is estimated to complete by August 2011.22
The Permanent Atrial fibriLLAtion Outcome Study Using Dronedarone on Top of Standard Therapy (PALLAS) study is a long-term trial with estimated enrollment of more than 10,000 patients who are aged >65 years with permanent AF and additional risk factors (coronary artery disease, prior stroke or TIA, symptomatic HF, left ventricular ejection fraction ≤40%, peripheral vascular occlusive disease, or aged >75 years with hypertension and diabetes mellitus).23 The objective of this study is to demonstrate the efficacy of dronedarone in preventing cardiovascular events (stroke, myocardial infarction, arterial embolism, or cardiovascular death) or cardiovascular-related hospitalization or death in permanent AF patients with additional risk factors. The study is recruiting patients and is estimated to complete August 2013.
ADVERSE EVENTS

DRUG INTERACTIONS
Dronedarone is highly metabolized by CYP3A4 isoenzyme. It is a moderate inhibitor of CYP3A4 and a weak inhibitor of CYP2D6. Concomitant administration of dronedarone and drugs that are strong CYP3A4 inhibitors (eg, ketoconazole, itraconazole, voriconazole, clarithromycin, and ritonavir) is contraindicated. Concurrent use of dronedarone and drugs that prolong the QTc interval (eg, class I and III antiarrhythmics, tricyclic antidepressants, phenothiazines) is also contraindicated due to potential risk of torsades de pointes. Coadministration of CYP3A inducers, such as rifampin, phenytoin, cabamazepine, phenobarbital, or St. John's wort, can decrease dronedarone exposure and should be avoided.13

DOSING AND ADMINISTRATION
Dosing range studies have shown dronedarone 400-mg twice-daily is effective in the prevention of AF relapse and in the long-term maintenance of sinus rhythm. It should be taken in the morning and evening with meals. No dosage adjustment is required for renal or moderate hepatic impairment.13
FORMULARY CONSIDERATIONS
Each of the published clinical studies evaluated patients who have different comorbidities and risks. Clinical evidence has demonstrated dronedarone to be efficacious in reducing AF recurrence and cardiac-related hospitalizations in AF patients.15,16 Some important issues should be considered when prescribing dronedarone.
One such factor is the balance of potential benefit in adverse effects provided by dronedarone with the efficacy in prevention of AF recurrence provided by amiodarone. A meta-analysis evaluating 9 trials, including DIONYSOS, found that amiodarone was more effective than dronedarone in prevention of AF recurrence, but it was associated with a greater rate of discontinuation due to adverse events than dronedarone (OR, 1.81; CI, 1.33–2.46; P<.001). Piccini et al estimated that for every 1,000 patients treated with dronedarone in place of amiodarone, there would be 228 more recurrent AF cases at 12 months in exchange for 9.6 fewer deaths and 62 fewer adverse events requiring drug discontinuation.24
A limitation to dronedarone is its black box warning in patients with severe, symptomatic HF. The ANDROMEDA study was discontinued early due to significant increase in mortality with dronedarone when compared to placebo, mostly due to deaths which resulted from progressive heart failure.17 Despite its problems in severe, symptomatic HF, dronedarone has demonstrated reduction in death or cardiac hospitalization in clinically stable patients with left ventricular dysfunction and low ejection fraction (<45%).15 The main factor that influenced the effect of dronedarone in the ANDROMEDA and ATHENA studies was the patient's clinical stability at the time of dronedarone initiation.15,17 Therefore, dronedarone should be avoided in patients with severe HF (NYHA class IV) or moderate HF (NYHA class II or III) with a history of decompensation within the last month. Dronedarone may be considered when the patient becomes clinically stable and demonstrates intolerance to other agents, such as amiodarone. Currently, amiodarone and dofetilide are the recommended agents for patients with HF.25,26 Clinical question continues for the role of dronedarone in clinically stable HF patients who later experience decompensation.
Monitoring of dronedarone use is another important consideration. Dronedarone significantly increases serum creatinine, but it appears to be reversible upon drug discontinuation. Tschuppert et al found that dronedarone significantly reduced renal creatinine clearance when compared to placebo in healthy male subjects (P=.04). While the increase in serum creatinine was related to reduction in renal creatinine clearance, it was not accompanied by decrease in glomerular filtration.27 Dronedarone dosage adjustment in renal impairment is not recommended; however, patients with renal insufficiency should be carefully monitored for worsening kidney function.
Many patients who are maintained in normal sinus rhythm may still require anticoagulation for stroke prevention. Drug interaction with warfarin is another clinical factor that should be taken into consideration. An analysis of the international normalized ratio (INR) from the DIONYSOS trial demonstrated greater proportion of patients with supratherapeutic INR levels (>4.5) in the amiodarone group compared to dronedarone throughout the study period, despite more frequent warfarin dose adjustments. Further, occurrence of bleeding events was significantly higher in the amiodarone group 11.4% (29/255) when compared to dronedarone, 5.6% (14/249), P=.03.19 While amiodarone can increase the anticoagulant effect of warfarin, which could result in major bleeding complications, dronedarone does not appear to show such significant interactions with warfarin.10,13
Based on currently available evidence, dronedarone appears to be an acceptable alternative to amiodarone for use in preventing AF recurrence in patients who cannot tolerate adverse effects associated with amiodarone. The clinician should take into consideration the patient's risk factors, concomitant medications, clinical status, and tolerability to other antiarrhythmic agents prior to initiating dronedarone. If dronedarone is started, the patient must be monitored for its efficacy and safety.
CONCLUSION
There are many treatment options for AF, ranging from pharmacologic agents to nonpharmacologic interventions. The selection of a specific agent often depends on the ultimate long-term goal of therapy and individualized patient preference. Dronedarone is considered a possible therapeutic option in AF management. Until long-term efficacy and safety studies are available, the key to dronedarone prescribing is appropriate patient selection to ensure the most effective therapeutic approach.
Dr Draskovich is a pharmacist for Walgreens, Stockton, Calif. At time of manuscript preparation, she was a student at Thomas J. Long School of Pharmacy and Health Sciences, University of the Pacific, Stockton, Calif.
Dr O'Dell is associate professor, pharmacy practice, Thomas J. Long School of Pharmacy and Health Sciences, University of the Pacific, Stockton, Calif.
Disclosure Information: The authors report no financial disclosures as related to products discussed in this article.
In each issue, the "Focus on" feature reviews a newly approved or investigational drug of interest to pharmacy and therapeutics committee members. The column is coordinated by Robert A. Quercia, MS, RPh, medical editor, University of Connecticut/Hartford Hospital, Evidence-Based Practice Center, Hartford, Conn., and adjunct associate professor, University of Connecticut School of Pharmacy, Storrs, Conn; and by Craig I. Coleman, PharmD, associate professor of pharmacy practice, University of Connecticut School of Pharmacy, and director, Pharmacoeconomics and Outcomes Studies Group, Hartford Hospital.
EDITORS' NOTE: The clinical information provided in "Focus on" articles is as current as possible. Due to regularly emerging data on developmental or newly approved drug therapies, articles include information published or presented and available to the author up until the time of the manuscript submission.
REFERENCES
1. Fuster V, Rydén LE, Cannom DS, et al; ACC/AHA Task Force Members and ESC Committee for Practice Guidelines. ACC/AHA/ESC 2006 Guidelines for the Management of Patients with Atrial Fibrillation: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and the European Society of Cardiology Committee for Practice Guidelines (Writing Committee to Revise the 2001 Guidelines for the Management of Patients with Atrial Fibrillation): developed in collaboration with the European Heart Rhythm Association and the Heart Rhythm Society. Circulation. 2006;114:e257-e354.
2. Tisdale JE. Arrhythmias. In: Chisholm-Burns MA, Wells BG, Schwinghammer TL, et al. Pharmacotherapy: principles and practice. 6th ed. New York, NY: McGraw-Hill. 2008.
3. Cheng JWM. New and emerging antriarrhythmic and anticoagulant agents for atrial fibrillation. Am J Health-Syst Pharm. 2010;67(suppl 5):S26–S34.
4. Hohnloser SH, Kuck KH, Lilienthal J, for the PIAF Investigators. Rhythm or rate control in atrial fibrillation–Pharmacological Intervention in Atrial Fibrillation (PIAF): a randomised trial. Lancet. 2000;356:1789–1794.
5. The Atrial Fibrillation Follow-up Investigation of Rhythm Management (AFFIRM) Investigators. A comparison of rate control and rhythm control in patients with atrial fibrillation. N Engl J Med. 2002;347:1825–1833.
6. Van Gelder IC, Hagens VE, Bosker HA, et al; for the Rate Control versus Electrical Cardioversion for Persistent Atrial Fibrillation Study Group. A comparison of rate control and rhythm control in patients with recurrent persistent atrial fibrillation. N Engl J Med. 2002;347:1834–1840.
7. Carlsson J, Miketic S, Windeler J, et al; for the STAF Investigators. Randomized trial of rate-control versus rhythm-control in persistent atrial fibrillation: the Strategies of Treatment of Atrial Fibrillation (STAF) study. J Am Coll Cardiol. 2003;41:1690–1696.
8. Opolski G, Torbicki A, Kosior DA, et al; for the Investigators of the Polish HOT CAFE Trial. Rate control vs rhythm control in patients with nonvalvular persistent atrial fibrillation: the results of the Polish How to Treat Chronic Atrial Fibrillation (HOT CAFE) Study. Chest. 2004;126:476–486.
9. The Cardiac Arrhythmia Suppression Trial (CAST) Investigators. Preliminary report: effect of encainide and flecainide on mortality in a randomized trial of arrhythmia suppression after myocardial infarction. N Engl J Med. 1989;321:406–412.
10. Cordarone (amiodarone) [package insert]. Philadelphia, PA: Wyeth Pharmaceuticals Inc.; August 2009.
11. Roy D, Talajic M, Dorian P, et al; for the Canadian Trial of Atrial Fibrillation Investigators. Amiodarone to prevent recurrence of atrial fibrillation. N Engl J Med. 2000;342:913–920.
12. Singh BN, Singh SN, Reda DJ, et al; for the Sotalol Amiodarone Atrial Fibrillation Efficacy Trial (SAFE-T) Investigators. Amiodarone versus sotalol for atrial fibrillation. N Engl J Med. 2005;352:1861–1872.
13. Multaq [package insert]. Bridgewater, NJ: Sanofi-Aventis U.S. LLC; 2009.
14. Sanofi-Aventis and FDA. Briefing Information for the March 18, 2009 Meeting of the Cardiovascular and Renal Drugs Advisory Committee. Available at: http://www.fda.gov/AdvisoryCommittees/CommitteesMeetingMaterials/Drugs/CardiovascularandRenalDrugsAdvisoryCommittee/ucm134972.htm. Accessed Sept. 3, 2010.
15. Hohnloser SH, Crijns HJ, van Eickels M, et al; for the ATHENA Investigators. Effect of dronedarone on cardiovascular events in atrial fibrillation. N Engl J Med. 2009;360:668–678.
16. Singh BN, Connolly SJ, Crijns HJ, et al; for the EURIDIS and ADONIS Investigators. Dronedarone for maintenance of sinus rhythm in atrial fibrillation or flutter. N Engl J Med. 2007;357:987–999.
17. Køber L, Torp-Pedersen C, McMurray JJ, et al; for the Dronedarone Study Group. Increased mortality after dronedarone therapy for severe heart failure. N Engl J Med. 2008;358:2678–2687.
18. Davy JM, Herold M, Hoglund C, et al; for the ERATO Study Investigators. Dronedarone for the control of ventricular rate in permanent atrial fibrillation: the efficacy and safety of dRonedArone for The cOntrol of ventricular rate during atrial fibrillation (ERATO) study. Am Heart J. 2008;156:527.e1–527.e9.
19. Le Heuzey JY, De Ferrari GM, Radzik D, Santini M, Zhu J, Davy JM. A short-term, randomized, double-blind, parallel-group study to evaluate the efficacy and safety of dronedarone versus amiodarone in patients with persistent atrial fibrillation: the DIONYSOS study. J Cardiovasc Electrophysiol. 2010;21:597–605.
20. Sanofi-Aventis. Dronedarone pattern of use in patients scheduled for elective cardioversion (ELECTRA). In: http://clinicaltrials.gov. Bethesda, MD: National Library of Medicine. 2009 [cited June 21, 2010]. Available at: http://clinicaltrials.gov/ct2/show/NCT01026090. NLM Identifier: NCT 01026090. Accessed Sept. 3, 2010.
21. Sanofi-Aventis. Effect of addition of dronedarone to standard rate control therapy on ventricular rate during persistent atrial fibrillation (AFRODITE). In: http://clinicaltrials.gov. Bethesda, MD: National Library of Medicine. 2009 [cited June 21, 2010]. Available at: http://clinicaltrials.gov/ct2/show/NCT01047566. NLM Identifier: NCT01047566. Accessed July 23, 2010.
22. Sanofi-Aventis. Effects of Dronedarone on Atrial Fibrillation Burden in Subjects With Permanent Pacemakers (HESTIA). In: http://clinicaltrials.gov. Bethesda, MD: National Library of Medicine. 2009 [cited July 21, 2010]. Available at: http://clinicaltrials.gov/ct2/show/NCT01135017. NLM Identifier: NCT01135017. Accessed Sept. 3, 2010.
23. Sanofi-Aventis. Permanent Atrial fibriLLAtion Outcome Study Using Dronedarone on Top of Standard Therapy (PALLAS). In: http://clinicaltrials.gov. Bethesda, MD: National Library of Medicine. 2009 [cited August 23, 2010]. Available at: http://clinicaltrials.gov/ct2/show/NCT01151137. NLM Identifier: NCT01151137. Accessed August 22, 2010.
24. Piccini JP, Hasselblad V, Peterson ED, Washam JB, Califf RM, Kong DF. Comparative efficacy of dronedarone and amiodarone for the maintenance of sinus rhythm in patients with atrial fibrillation. J Am Coll Cardiol. 2009;54:1089–1095.
25. Singh SN, Fletcher RD, Fisher SG, et al. Amiodarone in patients with congestive heart failure and asymptomatic ventricular arrhythmia. Survival Trial of Antiarrhythmic Therapy in Congestive Heart Failure. N Engl J Med. 1995;333:77–82.
26. Torp-Pedersen C, Møller M, Bloch-Thomsen PE, et al. Dofetilide in patients with congestive heart failure and left ventricular dysfunction. Danish Investigations of Arrhythmia and Mortality on Dofetilide Study Group. N Engl J Med. 1999;341:857–865.
27. Tschuppert Y, Buclin T, Rothuizen LE, et al. Effect of dronedarone on renal function in healthy subjects. Br J Clin Pharmacol. 2007;64:785–791.
Coalition promotes important acetaminophen dosing reminders
November 18th 2014It may come as a surprise that each year Americans catch approximately 1 billion colds, and the Centers for Disease Control and Prevention estimates that as many as 20% get the flu. This cold and flu season, 7 in 10 patients will reach for an over-the-counter (OTC) medicine to treat their coughs, stuffy noses, and sniffles. It’s an important time of the year to remind patients to double check their medicine labels so they don’t double up on medicines containing acetaminophen.
Support consumer access to specialty medications through value-based insurance design
June 30th 2014The driving force behind consumer cost-sharing provisions for specialty medications is the acquisition cost and not clinical value. This appears to be true for almost all public and private health plans, says a new report from researchers at the University of Michigan Center for Value-Based Insurance Design (V-BID Center) and the National Pharmaceutical Council (NPC).
Management of antipsychotic medication polypharmacy
June 13th 2013Within our healthcare-driven society, the increase in the identification and diagnosis of mental illnesses has led to a proportional increase in the prescribing of psychotropic medications. The prevalence of mental illnesses and subsequent treatment approaches may employ monotherapy as first-line treatment, but in many cases the use of combination of therapy can occur, leading to polypharmacy.1 Polypharmacy can be defined in several ways but it generally recognized as the use of multiple medications by one patient and the most common definition is the concurrent use of five more medications. The presence of polyharmacy has the potential to contribute to non-compliance, drug-drug interactions, medication errors, adverse events, or poor quality of life.
Medical innovation improves outcomes
June 12th 2013I have been diagnosed with stage 4 cancer of the pancreas, a disease that’s long been considered not just incurable, but almost impossible to treat-a recalcitrant disease that some practitioners feel has given oncology a bad name. I was told my life would be measured in weeks.
