- Safety & Recalls
- Regulatory Updates
- Drug Coverage
- COPD
- Cardiovascular
- Obstetrics-Gynecology & Women's Health
- Ophthalmology
- Clinical Pharmacology
- Pediatrics
- Urology
- Pharmacy
- Idiopathic Pulmonary Fibrosis
- Diabetes and Endocrinology
- Allergy, Immunology, and ENT
- Musculoskeletal/Rheumatology
- Respiratory
- Psychiatry and Behavioral Health
- Dermatology
- Oncology
A drug use evaluation of amphotericin B lipid complex injection following institution of new treatment guidelines at a tertiary teaching medical center
A drug use evaluation at Wake Forest University Baptist Medical Center (WFUBMC) was initiated following a change in institution-approved guidelines for the administration of amphotericin B lipid complex injection (ABLC) (Abelcet, Enzon). This study was conducted to determine compliance with the new guidelines among prescribers and to characterize the population of patients receiving ABLC. A total of 153 patients received ABLC from April 2001 through December 2002. One hundred thirty-three patients (87%) met 1 or more of the institutional criteria for ABLC administration and 20 (13%) receiving ABLC did not meet the guidelines. The mean baseline serum creatinine (SCr) value among patients meeting institutional guidelines (n=133) was 2.0 mg/dL (range, 0.5–5.0 mg/dL). Among patients who did not meet the guidelines (n=20), the mean baseline SCr level was 1.7 mg/dL (range, 0.6–2.9 mg/dL). The use of new guidelines, which were less stringent than previous guidelines, resulted in earlier administration..
Abstract

Amphotericin B has the broadest spectrum of activity of any antifungal agent. For decades it has been considered the gold standard for the treatment of severe systemic fungal infections.1 However, the efficacy of the conventional formulation, amphotericin B deoxycholate, is outweighed by a significant toxicity profile, often leading to severe renal damage. Adverse infusion-related events are also of concern, sometimes resulting in discontinuation of therapy.
There are currently 3 lipid formulations approved by FDA for clinical use: amphotericin B lipid complex injection (ABLC) (Abelcet, Enzon), amphotericin B cholesteryl sulfate (ABCD) (Amphotec, InterMune), and liposomal amphotericin B (L-AmB) (AmBisome, Fujisawa). Each of these agents is indicated for the treatment of systemic mycoses in patients who are intolerant of or refractory to conventional amphotericin B. Specifically, amphotericin B lipid complex allows for more specific tissue penetration and organ targeting; the lipid complex is rapidly and extensively taken up by the reticuloendothelial system and achieves high tissue concentrations in the liver, spleen, and lungs with minimal renal uptake.2 Acute infusion-related reactions are more common with the lipid-based formulations of amphotericin B compared with amphotericin B deoxycholate. Similarly, liver toxicity has been reported with the lipid-based formulations but is generally not associated with the conventional formulation.3,4
Other agents available for the treatment of systemic fungal infections include the azole antifungals, which act by blocking the activity of 14-alpha-demethylase, leading to inhibition of ergosterol synthesis; and the recently introduced echinocandin antifungals, which inhibit synthesis of an essential component of the fungal cell wall––beta (1,3)-D-glucan.3 These agents are typically associated with fewer adverse events than amphotericin B, particularly the echinocandins since they act on a component that is absent in mammalian cells; however, their spectrum of activity is not as broad as that of the polyenes (ie, amphotericin B).3
Hospital pharmacy departments are under ever-increasing pressure to rein in drug expenditures and to ensure that treatments are administered in the most cost-effective manner possible. At the same time, physicians are demanding access to a growing number of therapeutic options for their patients. In order to balance the fiscal responsibilities of the pharmacy department with the therapeutic needs of patients, it is necessary to develop drug administration guidelines that help control costs while providing flexibility with regard to the treating physician's judgment.
Though justifiable in terms of patient response, reduced toxicity profile, and patient outcomes, lipid formulations of amphotericin B have much higher acquisition costs than the conventional formulation, which carries a cost of approximately $20 per day. The least expensive lipid preparation is ABCD, at a cost of approximately $448 per day for a 70-kg patient, but this agent is associated with a higher incidence of infusion-related adverse events than the other lipid formulations.5 ABLC treatment for a 70-kg patient costs approximately $800 per day. L-AmB ranges in cost depending on the required dosage; for a 70-kg patient, this cost can range from $790 to $2,000 per day.6

The efficacy and safety of ABLC as therapy for invasive fungal infections was established in an open-label emergency-use study.7 Of the 556 patients with invasive fungal infections, 291 had mycologically confirmed fungal infections and met the criteria for the evaluation of therapeutic response. The overall response rate, defined as complete or partial response, was 57%. Overall response rates by pathogen were 42% for aspergillosis (55/130), 71% for candidiasis (65/91), 71% for zygomycosis (17/24), 64% for Cryptococcus (7/11), and 82% for fusariosis (9/11). Of 556 cases evaluable for safety, 396 (66%) demonstrated either stable or improved serum creatinine (SCr) levels at the end of therapy. Trend analysis of the data also revealed that SCr values tended to decline and kidney function improved in patients receiving ABLC.
Comparative trials have been published that justify the selection of ABLC as the treatment of choice at WFUBMC. A randomized trial in leukemia patients compared ABLC with L-AmB, both dosed at 3 mg/kg/d for fever of unknown origin or 5 mg/kg/d for definite infection.8 The ABLC group had a significantly higher clinical response to therapy (27/43 [63%] vs 15/39 [39%], P=.03). Infusion-related toxicity was higher in patients receiving ABLC and rates of hepatic toxicity (liver enzyme elevation) were higher in patients receiving L-AmB. There was no statistical difference in nephrotoxicity. Another study in patients with refractory fungal infection or intolerance of prior antifungal therapy compared 46 ABLC-treated patients with 21 patients receiving L-AmB.9 Nephrotoxicity, defined as doubling of SCr values from baseline, was observed in 2 (4.4%) of the ABLC-treated patients and 4 (19%) of the L-AmB-treated patients.
In a randomized, double-blind, comparative trial, the safety of ABLC and L-AmB was evaluated in 244 patients in 18 centers.10 Nephrotoxicity, defined as doubling of SCr values from a mean baseline value of 0.7 mg/dL, was observed in 14.1% and 14.8% of L-AmB patients (3 and 5 mg/kg/d, respectively) and in 42.3% of ABLC recipients (5 mg/kg/d). However, the difference between changes in SCr values varied only modestly among the 3 treatment groups. The mean changes in SCr levels among L-AmB-treated patients were 0.5±0.8 mg/dL and 0.4±0.4 mg/dL (3 and 5 mg/kg/d, respectively). Patients receiving ABLC experienced a mean change in SCr level of 1.0±1.0 mg/dL. Consideration of these factors led the authors to question whether the differences between the agents were clinically meaningful.
GUIDELINES FOR ABLC ADMINISTRATION AT WFUBMC
In 1996, the WFUBMC Department of Pharmacy and Section on Infectious Diseases developed usage guidelines for ABLC that were approved by the pharmacy and therapeutics (P&T) committee. The guidelines were based on data available at the time, including guidelines for enrollment in compassionate-use studies conducted prior to ABLC approval (Table 1). A concurrent drug use evaluation (DUE) was conducted from April 1, 1997, through March 31, 1999.11 ABLC indication, dose, duration of therapy, and therapeutic outcome were assessed. One hundred fifty-six patients received ABLC for either documented (57%) or suspected (43%) fungal infection. Forty-six percent of patients met the guidelines. Fifty-four percent did not, but had worsening renal function or another indication for which ABLC use was deemed medically necessary by the treating physician. Though a majority of patients did not meet the guidelines, the Department of Pharmacy and the P&T committee chose to keep the guidelines in place. The goal of the guidelines was to ensure the most cost-effective use of ABLC. At that time there also was no approval process, making enforcement of the guidelines difficult.
Upon further review, it was determined that revision of the guidelines was warranted. In March 2001, the WFUBMC Center for Antimicrobial Utilization, Stewardship, and Epidemiology (CAUSE), a subcommittee of the P&T committee, drafted and implemented new guidelines (Table 1). Although these guidelines included a lower threshold of SCr for conversion from conventional amphotericin B to ABLC, data from Wingard et al demonstrated that these high-risk patients were at an increased risk for renal failure leading to dialysis and ultimately to increased risk of mortality.12 The revised guidelines were designed to be more inclusive and, in contrast to the old guidelines, did not require that patients meet multiple, concurrent criteria. In particular, the new guidelines recognize the importance of initiating ABLC earlier in the treatment continuum. As the data will demonstrate, this change resulted in the maintenance of more stable, lower-level SCr values (2.0 mg/dL) at WFUBMC than had been observed in the previous evaluation (2.6 mg/dL) under the old guidelines.
The Department of Pharmacy initiated another DUE at that time to assess compliance with the new guidelines and characterize the patients who were receiving ABLC. Though clinical response and long-term outcome data were not obtained, the impact of treatment on SCr values was assessed to determine the impact on renal function, as reduced or stable SCr values are considered predictors of a positive outcome.12
DATA COLLECTION

RESULTS



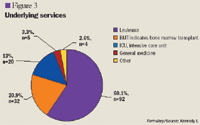
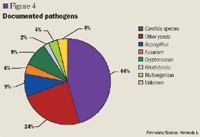

The mean baseline SCr value among patients meeting institutional guidelines (n=133) was 2.0 mg/dL (range, 0.5–5.0 mg/dL). Among patients who did not meet the guidelines (n=20), the mean SCr level was 1.7 mg/dL (range, 0.6–2.9 mg/dL). There was no significant difference in mortality rates among patients meeting the guidelines (27%, 36/133) and patients not meeting the guidelines (30%, 6/20) (Table 5).

The mean SCr value in the overall population did not change from baseline to the end of ABLC therapy, remaining at 2.0 mg/dL (Table 4). Among patients meeting the institutional guidelines, the mean SCr level exhibited an identical trend and remained at 2.0 mg/dL from baseline to end of therapy. Patients not meeting institutional guidelines experienced a 0.3-mg/dL increase in the mean SCr value from 1.7 to 2.0 mg/dL over the course of ABLC administration. An evaluation of other nephrotoxic medications (ie, aminoglycosides, cyclosporine, tacrolimus) was conducted and there was minimal use in this patient group.
Although 28% of patients (42/153) died during therapy (Table 5), the cause of death was not collected in this DUE. As these patients had a number of life-threatening conditions prior to ABLC treatment, it is difficult to attribute these deaths to any specific cause.
DISCUSSION
The new guidelines governing the administration of ABLC at WFUBMC are designed to improve the quality of patient care and better reflect the prescribing practices of physicians. By lowering the required SCr values for initiation of ABLC therapy, earlier therapeutic transition is possible from conventional amphotericin B to a less nephrotoxic alternative. As the data demonstrate, this resulted in the maintenance of stable SCr values at a lower level at WFUBMC (2.0 mg/dL) than had been seen in the previous evaluation under the old guidelines (2.6 mg/dL). The maintenance of SCr values at a mean of 2.0 mg/dL throughout ABLC therapy decreases the potential for renal toxicity. In addition, as a large number of patients requiring antifungal therapy are bone marrow transplant and leukemia patients, maintaining a lower SCr level may allow for the concomitant administration of potentially nephrotoxic drugs.
It should also be noted that strict adherence to the guidelines is not required for prescribing ABLC at WFUBMC. Rather, these guidelines serve to facilitate the cost-effective administration of ABLC. Although further study is warranted, the improved quality of care and earlier use of ABLC resulting from less stringent guidelines can diminish the overall institutional costs associated with antifungal treatment and long-term patient care. At press time, the guidelines reported were still in use at WFUBMC.
Dr Kennedy is pharmaceutical care coordinator, Hematology and Oncology, Wake Forest University Baptist Medical Center, Winston-Salem, NC. She can be reached at lakenned@wfubmc.edu
Disclosure Information: Enzon Pharmaceuticals provided an educational grant for the collection of data used in this evaluation. Dr Kennedy has served as a consultant and speaker for Enzon Pharmaceuticals.
REFERENCES
1. Ostrosky-Zeichner L, Marr KA, Rex JH, Cohen SH. Amphotericin B: time for a new gold standard. Clin Infect Dis. 2003;37:415–425.
2. Olsen SJ, Swerdel MR, Blue B, Clark JM, Bonner DP. Tissue distribution of amphotericin B lipid complex in laboratory animals. J Pharm Pharmacol. 1991;43:831–835.
3. Antifungal drugs. Treatment Guidelines from The Medical Letter. 2005;3:7–14.
4. Abelcet [package insert]. Bridgewater, NJ: Enzon Pharmaceuticals; 1999.
4. White TC, Marr KA, Bowden RA. Clinical, cellular and molecular factors that contribute to antifungal drug resistance. Clin Microbiol Rev. 1998;11:382–402.
5. Lewis RE. Antifungal therapy cost analysis. Available at: http:// http://www.doctorfungus.org/thedrugs/cost1.htm. Accessed August 16, 2006.
6. Walsh TJ, Hiemenz JW, Seibel NL, et al. Amphotericin B lipid complex for invasive fungal infections: analysis of safety and efficacy in 556 cases. Clin Infect Dis. 1998;26:1383–1396.
7. Fleming RV, Kantarjian HM, Husni R, et al. Comparison of amphotericin B lipid complex (ABLC) vs. ambisome in the treatment of suspected or documented fungal infections in patients with leukemia. Leuk Lymphoma. 2001;40:511–520.
8. Cannon JP, Garey KW, Danziger LH. A prospective and retrospective analysis of the nephrotoxicity and efficacy of lipid-based amphotericin B formulations. Pharmacotherapy. 2001;21: 1107–1114.
9. Wingard JR, White MH, Anaissie E, Raffalli J, Goodman J, Arrieta A; L Amph/ABLC Collaborative Study Group. A randomized, double-blind comparative trial evaluating the safety of liposomal amphotericin B versus amphotericin B lipid complex in the empirical treatment of febrile neutropenia. Clin Infect Dis. 2000;31:1155–1163.
10. Kennedy LD, Connelly JF, Kuzma KM. Two-year longitudinal evaluation of amphotericin B lipid complex injection in a tertiary care medical center. Hospital Pharm. 2001;36:176–181.
11. Bates DW, Su L, Yu DT, et al. Correlates of acute renal failure in patients receiving parenteral amphotericin B. Kidney Int. 2001;60:1452–1459.
Employers Face Barriers With Adopting Biosimilars
March 1st 2022Despite the promise of savings billions of dollars in the United States, adoption of biosimilars has been slow. A roundtable discussion among employers highlighted some of the barriers, including formulary design and drug pricing and rebates.
