- Safety & Recalls
- Regulatory Updates
- Drug Coverage
- COPD
- Cardiovascular
- Obstetrics-Gynecology & Women's Health
- Ophthalmology
- Clinical Pharmacology
- Pediatrics
- Urology
- Pharmacy
- Idiopathic Pulmonary Fibrosis
- Diabetes and Endocrinology
- Allergy, Immunology, and ENT
- Musculoskeletal/Rheumatology
- Respiratory
- Psychiatry and Behavioral Health
- Dermatology
- Oncology
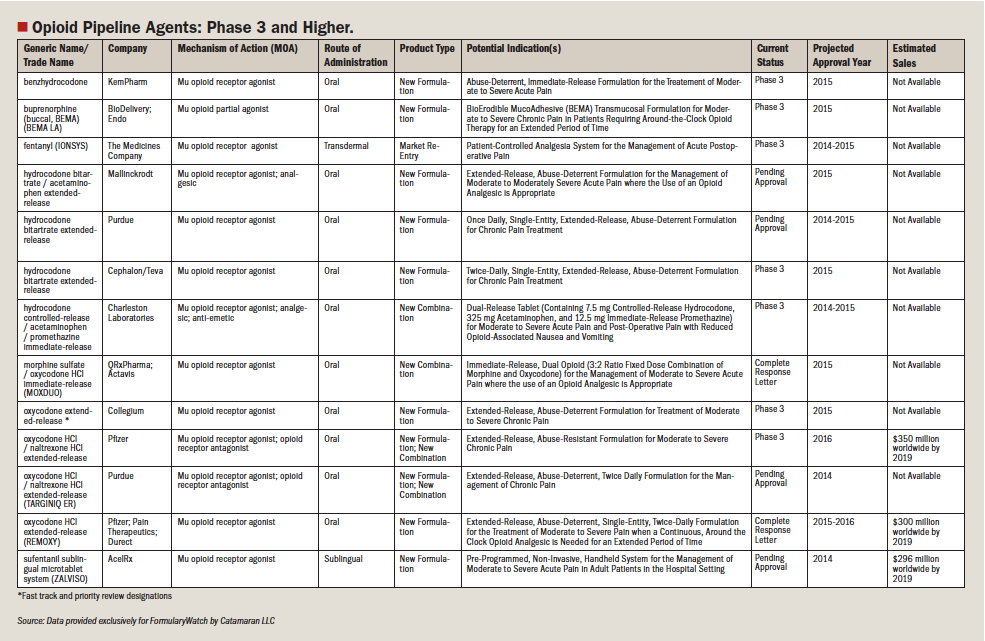
Drugs in Perspective: Zohydro ER (hydrocodone bitartrate extended release)
Pain management is gradually becoming a topic of conversation and gaining considerable attention as it relates to providing quality services for those with acute or chronic pain ailments. Pain can affect millions of Americans and it contributes significantly to national rates of morbidity, mortality, and disability.
Pain management is gradually becoming a topic of conversation and gaining considerable attention as it relates to providing quality services for those with acute or chronic pain ailments. Pain can affect millions of Americans and it contributes significantly to national rates of morbidity, mortality, and disability.1 As It currently stands, there is much disparity in the prevalence, degree of severity, and adequate level of treatment of pain management, which has produced a public health concern, particularly with chronic pain management.1 The experience of pain can include affective, behavioral, cognitive, sociocultural, or physiologic effects.2 There are a variety of interventions for chronic pain such as opioid medication or surgery with analgesic pharmacotherapy being considered the mainstay of pain management.2 For many individuals, continuous or long-term pain management can mean the difference between achieving an adequate quality of life.3
Zohydro ER (hydrocodone bitartrate) is an oral, extended-release capsule, analgesic opioid that was approved by FDA in October 2013 for the management of pain that is severe enough to require daily, around-the-clock, long-term treatment and with no other alternative treatments being effective.4 As an opioid analgesic, it binds to opioid receptors in the central nervous system to cause an inhibition of the ascending pain pathways, which ultimately changes an individual’s perception and response to pain.5 The drug is available as an extended-release oral capsule (12 hours) at 10 mg, 15 mg, 20 mg, 30 mg, 40 mg, and 50 mg.5,6 The suggested dosing regimen for the management of pain is 10 mg every 12 hours for those who are first-time users and higher doses can be given to those individuals who are opioid intolerant.5,6 There is a 40-mg single dose, 50-mg capsule, and total daily dose of 80 mg for those who are considered to be tolerant to the opioid.6 In individuals with renal impairment a lower initial dose is recommended, and for individuals with severe hepatic impairment an initial dose of 10 mg is recommended.
Relevance
Although there are many opioid analgesics on the market, this is the first oral, single entity without acetaminophen. This novel extended-release formulation of hydrocodone can be given every 12 hours, around the clock, for the management of moderate-to-severe chronic pain for long-term treatment.7 It is the first pure hydrocodone drug that has produced a mixture of both positive comments and concerns about its use and potential for abuse.
Studies
This hydrocodone bitartrate capsule was studied in over 1100 individuals who had chronic pain and participated in a phase 3 efficacy study or an open-label phase 3, long-term safety study. The phase 3 efficacy study with over 500 participants who had chronic low back pain reached the primary endpoint of significantly improved back pain with this drug compared to placebo.8 The secondary endpoint for the study, pain intensity, showed a 30% improvement from baseline to the end of study with the hydrocodone bitartrate capsule compared to placebo, at 67.5% and 31.1%, respectively.8
Potential advantages and disadvantages
This is the first hydrocodone product approved by FDA to offer the potential benefits of less-frequent dosing requirements with the ability to treat individuals with chronic pain without the risk of acetaminophen-related liver injury.9 Unfortunately, there is the concern that the pure hydrocodone is powerful enough that just one accidental overdose can kill an individual.10 Due the fact that this formulation of hydrocodone bitartrate has the ability to cause potentially life-threatening respiratory depression even with therapeutic uses, it includes an FDA-approved boxed warning related to the increased risk of this adverse outcome.11 The decision to approve this drug goes against the 2012 recommendation by the FDA advisory committee and came a day after it announced its intention to recommend to the DEA to move the combination hydrocodone product from schedule III to schedule II.12 FDA believed that the drug will only serve to worsen the current prescription drug addiction epidemic. The attorney general and critics argue that the number of overdose deaths related to prescription pain killers has increased over the last dozen years, so it is important to strive to reduce the potential damage that can be caused by this drug and other similar drugs.
Cost
This extended-release formulation of hydrocodone bitartrate became available on the market in March 2014.13 The price for a 100 count of the 10-mg strength is $702.00, 15 mg is $750.00, 20 mg is $774.00, 30 mg is $798.00, 40 mg is $822.00, and 50 mg is $858.00.4,13
Place in therapy
This drug will serve as a novel analgesic treatment option for individuals with chronic pain for long-term use when other treatment options are inadequate. As a schedule II medication it will be subjected to similar controls as other narcotics with the highest potential for abuse.14
Implications
FDA approval of this extended-release formulation of hydrocodone bitartrate has significant implications on chronic pain management, which is estimated to contribute to a national economic cost of $560 billion to $635 billion annually.15 Prior to the availability of this agent there have only been immediate-release drugs that contain combination analgesic ingredients, one of which was primarily acetaminophen. The overuse of acetaminophen can cause accidental overdose or liver damage, but this new drug does not contain acetaminophen and can be administered over an extended period of time to achieve pain management.
Dr Farinde serves on the faculty at Columbia Southern University, Orange Beach, Ala.
Disclosure information: The author reports no financial disclosures as related to products discussed in this article.
References
Gatchel RJ, McGeary DD, McGeary CA, Lippe B. Interdisciplinary chronic pain management: past, present, and future. Am Psychol. 2014;69(2):119–130.
Chiaretti A, Pierri F, Valentini P, et al. Current practice and recent advances in pediatric pain management. Eur Rev Med Pharmacol Sci. 2013;17 (Suppl 1):112–126.
O’Connor AB. Neuropathic pain: quality-of-life impact, costs and cost effectiveness of therapy. Pharmacoeconomics. 2009;27(2):95–112.
FDA. FDA approves extended-release, single-entity hydrocodone product.http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm372287.htm. October 25, 2013. Accessed May 27, 2014
Zohydro ER (Hydrocodone, extended release). Lexi-Comp Online, Lexicomp. Hudson, Ohio: Lexi-Comp, Inc.; March 3, 2014.
Zohydro ER [package insert]. San Diego, CA: Zogenix Inc; March 2014.
Zohydro ER. RxList. http://www.rxlist.com/zohydro-er-drug/indications-dosage.htm.
Accessed May 27, 2014.
Zogenix receives FDA approval for Zohydro (TM) ER (hydrocodone bitartrate) extended-release capsules. Wall Street Journal. October 25, 2013. http://online.wsj.com/article/PR-CO-20131025-911172.html. Accessed May 27, 2014.
Zogenix’s NDA for treatment of chronic pain receives FDA acceptance for review. PharmaWatch: CNS. 2012;11(9):16–17. http://pdc-connection.ebscohost.com/c/articles/79660203/zogenixs-nda-treatment-chronic-pain-receives-fda-acceptance-review. Accessed May 27, 2014.
FDA approves powerful new pain pill despite fears of abuse. (Louisville) Courier-Journal. February 28, 2014. http://www.courier-journal.com/article/20140228/NEWS01/302280046/1008/NEWS5001/. Accessed May 27, 2014.
Abele W. Herbal medication: potential for adverse interactions with analgesic drugs. J Clin Pharm Ther. 2002;27(6)391–401.
Young C. Controversy surrounds FDA approval of Zohydro. Pharmacy Today. 2013. http://www.pharmacist.com/controversy-surrounds-fda-approval-zohydro. Accessed May 27, 2014.
Zohydro ER. Zongenix, Inc. http://www.zohydroer.com/. Accessed March 3, 2014.
FDA oks new hydrocodone painkiller. HealthDay. October 25, 2013. http://consumer.healthday.com/bone-and-joint-information-4/pain-health-news-520/fda-oks-new-hydrocodone-painkiller-681512.html. Accessed May 27, 2014.
Zohydro ER. FDA accepts for review Zohydro ER NDA. Drugs.com. http://www.drugs.com/nda/zohydro_120716.html. Accessed May 27, 2014.
FDA Issues Complete Response Letter for Pz-Cel to Treat Epidermolysis Bullosa
April 22nd 2024Prademagene zamikeracel is a cell therapy designed to incorporate the functional collagen-producing COL7A1 gene into a patient’s own skin cells. The FDA is asking for additional information on manufacturing practices.
