- Safety & Recalls
- Regulatory Updates
- Drug Coverage
- COPD
- Cardiovascular
- Obstetrics-Gynecology & Women's Health
- Ophthalmology
- Clinical Pharmacology
- Pediatrics
- Urology
- Pharmacy
- Idiopathic Pulmonary Fibrosis
- Diabetes and Endocrinology
- Allergy, Immunology, and ENT
- Musculoskeletal/Rheumatology
- Respiratory
- Psychiatry and Behavioral Health
- Dermatology
- Oncology
Fampridine-SR: A potassium-channel blocker for the improvement of walking ability in patients with MS
Fampridine-SR is a sustained-release oral medication that is pending FDA approval for the symptomatic treatment of multiple sclerosis (MS).

Key Points
Abstract
Fampridine-SR is a sustained-release oral medication that is pending FDA approval for the symptomatic treatment of multiple sclerosis (MS). Fampridine (4-aminopyridine) is a potassium-channel blocker that has been demonstrated to improve impulse conduction in nerve fibers in which myelin has been damaged, a hallmark of MS. Although fampridine-SR is not a disease-modifying therapy, this agent does offer a novel approach to target MS symptoms, specifically walking ability and lower-extremity strength. A recent phase 3 trial in patients with various types of MS demonstrated that fampridine-SR is associated with clinically meaningful improvements in some patients regardless of concomitant therapy. If approved by FDA, fampridine-SR would be the first drug specifically indicated for the improvement of walking ability in patients with MS. (Formulary. 2009;44:293–299.)

CHEMISTRY AND PHARMACOLOGY
Myelin destruction, the primary effect of MS, exposes the potassium channels, creating impairments in conduction and generation of action potential through the axons. Fampridine-SR's mechanism of action targets the underlying axonal conduction block associated with this demyelination. Fampridine-SR is a monoamine and diamino derivative of pyridine (C5H6N2).4 Its primary mechanism of action is as a voltage-dependent, neuronal fast-potassium-channel blocker. At a cellular level, this increases neuronal excitability, potentiates synaptic and neuromuscular transmission, and relieves the conduction blockade in demyelinated axons. In myelinated axons, the blockade of fast potassium channels has little effect on the action potential. Fampridine-SR also more readily enters the open versus closed channels and remains trapped for an extended period of time in the closed channels, which accounts for the prolonged effects of the potassium-channel blockade.4,7
Potassium-channel blockade by fampridine-SR also results in greater than normal calcium influx at presynaptic terminals, which leads to enhanced neuroneuronal and neuromuscular transmission.4 Lastly, potassium-channel blockade has been demonstrated to have immunomodulatory properties. Experimental rat models of MS have demonstrated a delay in the hypersensitivity response to myelin protein and an improvement in the symptoms of the disease caused by blockade of potassium channels in T-lymphocytes.4
PHARMACOKINETICS
Blight and Henney8 conducted a pharmacokinetic study of a single dose of immediate-release fampridine in 4 healthy volunteers. After oral administration of fampridine 15 mg, peak plasma concentrations were reached in 1 hour after dosing. A median peak concentration of 72.9 ng/mL and a median half-life of 3.14 hours were observed with fampridine administration. Excretion was rapid and complete, occurring within 24 hours after dosing. Total cumulative recovery of the original dose was 96.36% with 95.85% excretion in the urine, suggesting that fampridine is unlikely to undergo substantial metabolic transformation.
Pharmacokinetic studies with fampridine-SR have been conducted mainly in patients with spinal cord injury.4 In patients taking fampridine doses of 10 to 25 mg administered once or twice daily for 1 week, mean plasma concentrations and area under the curve were linearly proportional to the dose administered, whereas other pharmacokinetic parameters were independent of dose. Peak plasma concentrations were reached at 3.2 to 3.7 hours after dosing, and an elimination half-life of 5.8 to 5.9 hours was observed. Steady-state concentrations were reached after 4 days of twice-daily dosing.4 The total clearance ranged from 34 to 34.8 L/h across all doses. In doses up to 60 mg twice daily, fampridine-SR demonstrated a mean time to maximum concentration of 2.2 to 3 hours and a mean half-life of 5.7 to 6.9 hours.9 Mean volume of distribution was 415.4 to 528 L, and apparent total clearance was 51.4 to 57.7 L/h; both these parameters were independent of dosage.
CLINICAL TRIALS
Fampridine-SR has been assessed primarily as a symptomatic treatment for MS. Although all deficits contribute to overall disability in MS, ambulatory impairment is often the key factor that determines functional status and is a common outcome measure in trials for MS. Therefore, the rating scales most commonly used in MS clinical trials depend largely on ambulatory deficits to characterize overall disease severity.10
One of the first trials to evaluate fampridine-SR was a randomized, double-blind, crossover study in 10 patients with MS and with stable motor deficits as defined by the Expanded Disability Status Scale (EDSS).11 The EDSS, a commonly used descriptor of MS severity, is an ordinal scale based entirely on ambulatory impairment in its middle range (EDSS 4.5–7.5). Scores are based on the maximal distance a patient can walk and whether the patient is using an ambulatory aid such as a cane. At the upper and lower ranges of the EDSS, patients have either no ambulation or relatively intact ambulation.10
Patients were randomized to receive either fampridine-SR 17.5 mg twice daily or placebo for 1 week with a 1-week washout period before crossing over to the other treatment group. Quantitative measures of motor function were evaluated, including the EDSS, time to walk 8 m (26.24 ft), time to climb 4 stairs, maximum voluntary isometric contraction measured quantitatively (MVICT), manual muscle testing (MMT), grip strength, and patient global impression. Results were mixed, with 9 of 10 fampridine-treated patients demonstrating an improvement in time to walk 8 m (fampridine-SR, 27.9±30 s; placebo, 35±25.3 s; P=.02). No other measures reached statistical significance.11
More recently, Goodman et al12 conducted a multicenter, randomized, double-blind, placebo-controlled, dose-ranging study in 36 patients with clinically definite MS. After a 4-week baseline period, patients were randomly assigned to receive either fampridine-SR (n=25) or placebo (n=11). The dose of fampridine-SR was increased from 10 mg twice daily to 40 mg twice daily in weekly incremental steps of 5 mg twice daily. Downward titration occurred during the final week. The primary outcome was the safety of the escalating dosing schedule, and the secondary outcome was the efficacy of and dose response to fampridine-SR. Efficacy was determined by assessing the daily Brief Fatigue Inventory, weekly completion of the Modified Fatigue Impact Scale, a timed 25-foot walk, the Lower Extremity Manual Muscle Testing (LEMMT), and the subject global impression of change.
No efficacy end points reached statistical significance in the analysis of variance (ANOVA); however, a post-hoc analysis of walking speed did demonstrate a significant difference between fampridine-SR and placebo (P=.03), and improvement in lower-extremity muscle strength was observed with fampridine-SR compared with placebo in a prospective analysis (P=.01).
The safety analysis demonstrated that treatment-related adverse events occurred more frequently in the fampridine-SR group than in the placebo group (84% vs 27%). Reported adverse events included dizziness, paresthesia, asthenia, nausea, headache, and tremor. The majority of adverse events were considered mild to moderate and transient; however, 5 patients discontinued fampridine-SR treatment at doses >25 mg twice daily, including 2 patients who experienced seizures at fampridine-SR doses of 30 and 35 mg twice daily. No patients in the placebo group discontinued treatment because of adverse events. Based on the safety and efficacy results, the authors concluded that future studies should employ fampridine-SR doses of up to 20 mg twice daily, and walking speed and lower extremity strength should be used as outcome measures.
On the basis of these recommendations, Goodman et al13 conducted another dose-comparison trial of fampridine-SR with doses of up to 20 mg twice daily. This multicenter, randomized, double-blind, placebo-controlled, parallel-group study enrolled 206 patients with clinically definite MS. After a single-blind, 2-week placebo run-in period, patients were randomized to receive either fampridine-SR 10, 15, or 20 mg twice daily or placebo for 15 weeks. Dose escalation occurred the first 2 weeks, followed by 12 weeks of stable dosing and 1 week of dose reduction. The primary outcome of this study was the percent change in walking speed based on a timed 25-foot walk. A variety of secondary outcomes was also assessed.
No significant differences were observed among the groups in the percent change in walking speed, although all 3 fampridine-SR-treated groups did demonstrate a trend toward improvement in walking speed. An increase from baseline in the LEMMT during the stable dosing period was observed in patients treated with fampridine-SR 10 or 15 mg versus those treated with placebo (P=.018 and .003, respectively). No other significant changes in efficacy outcomes were observed in fampridine-SR-treated patients versus placebo-treated patients. Of note, a post-hoc analysis demonstrated a subset of patients in each group with walking speeds that were consistently faster during treatment than during the nontreatment period. Treatment responders were defined as patients whose walking speed for ≥3 visits during the double-blind treatment period was faster than the maximum speed measured in 5 nontreatment visits. Under this treatment responder model, 35.3% of patients treated with fampridine-SR 10 mg, 36% of patients treated with fampridine-SR 15 mg, and 38.6% of patients treated with fampridine-SR 20 mg demonstrated a response compared with 8.5% of placebo-treated patients.

In a phase 3, multicenter, double-blind, placebo-controlled trial, 301 patients with MS of varying types were randomized to receive either fampridine-SR 10 mg twice daily (n=229) or placebo (n=72) for 14 weeks.14 The study included patients with clinically defined MS who could complete 2 trials of a timed 25-foot walk in an average time of 8 to 45 seconds. The primary outcome was the proportion of timed-walk responders in each group, defined as patients who achieved faster walking speeds in ≥3 of 4 assessments during the treatment period than during the off-treatment period. Change in walking speed was measured by the timed 25-foot walk. Secondary outcome measures included the Ashworth score for spasticity and the LEMMT. A 12-item multiple sclerosis walking scale (MSWS-12) that measures patients' impressions of their ambulatory disability was used to validate the clinical significance of the timed walk response, along with a subject global impression and clinician global impression scale. After a 2-week placebo run-in period, patients began the 14-week treatment period followed by 4 weeks with no treatment. Patients were assessed every 4 weeks during the treatment period.
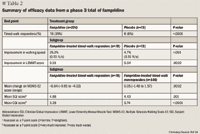
Eleven patients in the fampridine-SR group (5%) withdrew from the study because of adverse events (8 during the treatment phase). The incidence of insomnia, fatigue, back pain, and balance disorders was reported to be 50% higher in the fampridine-SR group than in the placebo group. A focal seizure that was deemed possibly related to fampridine-SR treatment also occurred in a patient who developed sepsis related to community-acquired pneumonia.
ADVERSE EVENTS
Based on recent clinical trials, adverse events are expected to occur in up to 80% to 87% of patients with MS taking fampridine-SR, with 15% to 20% of these events classified as severe. The most frequent adverse events observed in clinical trials with fampridine-SR 10 mg twice daily were fall (16%–19%), urinary tract infection (12%–14%), dizziness (4%–8%), insomnia (8%–10%), asthenia (6%–19%), headache (6%–12%), fatigue (6%–15%), and nausea (6%–10%).13,14
Seizures are also a concern with fampridine-SR because of its activity on potassium channels in the CNS.4,7 Unintentional overdose with compounded immediate release 4-AP has led to status epilepticus, and seizures have been noted in clinical trials with both immediate-release fampridine and fampridine-SR as described previously.15,16 A history of seizures was also an exclusion criterion in the largest phase 3 trial of fampridine-SR.14 A small trial of immediate-release fampridine demonstrated that the seizure risk may be associated with serum concentrations of the drug, but more information is needed to recommend therapeutic drug monitoring for fampridine-SR.7
Laboratory, electrocardiogram (ECG), and vital sign changes have been reported in fampridine-SR-treated patients in clinical trials, but specific changes were not described, although the authors did report that there were no clear trends or differences between the treatment groups.13,14 A study of long-term immediate-release fampridine treatment in patients with spinal cord injury and with impaired glucose tolerance actually demonstrated improvements in glucose levels, but more information from randomized clinical trials is needed to substantiate this observation.17 Fampridine-SR has not been associated with clinically significant changes in QT interval with doses ranging from 10 to 30 mg twice daily, according to a report released by Acorda.7
DRUG INTERACTIONS
To date, no formal drug interaction studies with fampridine-SR have been conducted. Because fampridine-SR may have the propensity to induce seizures, precautions should be taken in patients who are concomitantly taking medications that may lower the seizure threshold, such as tricyclic antidepressants, phenothiazines, and venlafaxine.7
DOSING AND ADMINISTRATION
Fampridine-SR is an enteric-coated tablet that is taken twice daily without regard to meals.7 Based on the recent large phase 3 clinical trial evaluating both efficacy and tolerability, fampridine-SR would likely be approved at a dose of 10 mg twice daily, although dose-ranging studies have been conducted with doses up to 50 mg twice daily.12–14 There is currently no information available on dosage adjustments in patients with hepatic or renal dysfunction.
Dr Feret is a clinical associate professor, University of Rhode Island College of Pharmacy, Kingston, Rhode Island.
Disclosure Information: The author reports no financial disclosures as related to products discussed in this article.
In each issue, the "Focus on" feature reviews a newly approved or investigational drug of interest to pharmacy and therapeutics committee members. The column is coordinated by Robert A. Quercia, MS, RPh, clinical manager and director of Drug Information, Department of Pharmacy Services, Hartford Hospital, Hartford, Conn, and adjunct associate professor, University of Connecticut School of Pharmacy, Storrs, Conn; and by Craig I. Coleman, PharmD, assistant professor of pharmacy practice, University of Connecticut School of Pharmacy, and director, Pharmacoeconomics and Outcomes Studies Group, Hartford Hospital.
EDITORS' NOTE: The clinical information provided in "Focus on" articles is as current as possible. Due to regularly emerging data on developmental or newly approved drug therapies, articles include information published or presented and available to the author up until the time of the manuscript submission.
REFERENCES
1. Who gets MS? National Multiple Sclerosis Society. http://www.nationalmssociety.org/about-multiple-sclerosis/who-gets-ms/index.aspx. Accessed September 8, 2009.
2. Calabresi PA. Diagnosis and management of multiple sclerosis. Am Fam Physician. 2004;70:1935–1994.
3. Goodin DS, Frohman EM, Garmany Jr, GP, et al; Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology and the MS Council for Clinical Practice Guidelines. Disease modifying therapies in multiple sclerosis: Report of the Therapeutic and Technology Assessment Subcommittee of the American Academy of Neurology and the MS Council for Clinical Practice Guidelines [erratum in Neurology. 2002;59:480]. Neurology. 2002;58:169–178.
4. Hayes KC. The use of 4-aminopyridine (fampridine) in demyelinating disorders. CNS Drug Rev. 2004;10:295–316.
5. October 14, 2009: Peripheral and Central Nervous System Drugs Advisory Committee meeting announcement. US Food and Drug Administration. http://www.fda.gov/AdvisoryCommittees/Calendar/ucm180089.htm. Accessed September 8, 2009.
6. Acorda Therapeutics announces FDA acceptance of Fampridine-SR New Drug Application for filing. Acorda Therapeutics. http://phoenix.corporate-ir.net/phoenix.zhtml?c=194451&p=irol-newsArticle&ID=1284888&highlight=. Accessed September 8, 2009.
7. Korenke AR, Rivey MP, Allington DR. Sustained-release fampridine for symptomatic treatment of multiple sclerosis. Ann Pharmacother. 2008;42:1458–1465.
8. Blight AR, Henney HR 3rd. Pharmacokinetics of 14C-radioactivity after oral intake of a single dose of 14C-labeled fampridine (4-aminopyridine) in healthy volunteers. Clin Ther. 2009;31:328–335.
9. Hayes KC, Potter PJ, Hsieh JT, Katz MA, Blight AR, Cohen R. Pharmacokinetics and safety of multiple doses of sustained-release 4-aminopyridine (Fampridine-SR) in subjects with chronic, incomplete spinal cord injury. Arch Phys Med Rehabil. 2004;85:29–34.
10. Schwid SR, Goodman AD, Mattson DH, et al. The measurement of ambulatory impairment in multiple sclerosis. Neurology.1997;49:1419–1424.
11. Schwid SR, Petrie MD, McDermott MP, Tierney DS, Mason DH, Goodman AD. Quantitative assessment of sustained-release 4-aminopyridine for symptomatic treatment of multiple sclerosis. Neurology. 1997;48:817–821.
12. Goodman AD, Cohen JA, Cross A , et al. Fampridine-SR in multiple sclerosis: A randomized, double-blind, placebo-controlled, dose-ranging study. Mult Scler. 2007; 13:357–368.
13. Goodman AD, Brown TR, Cohen JA, et al. Dose comparison trial of sustained-release fampridine in multiple sclerosis. Neurology. 2008;71: 1134–1141.
14. Goodman AD, Brown TR, Krupp LB, et al; Fampridine MsF203 Investigators. Sustained-release oral fampridine in multiple sclerosis: A randomised, double-blind, controlled trial. Lancet. 2009;373:732–738.
15. Burron JM, Bell CM, Walker SC, O'Connor PW. 4-aminopyridine toxicity with unintentional overdose in four patients with multiple sclerosis. Neurology. 2008; 71:1833–1834.
16. Schwam E. Severe accidental overdose of 4-aminopyridine due to a compounding pharmacy error. J Emerg Med. 2009 May 12 [Epub ahead of print].
17. Segal JL, Thompson JF, Tayek JA. Effects of long-term 4-aminopyridine therapy on glucose tolerance and glucokinetics in patients with spinal cord injury. Pharmacotherapy. 2007;27:789–792.
Employers Face Barriers With Adopting Biosimilars
March 1st 2022Despite the promise of savings billions of dollars in the United States, adoption of biosimilars has been slow. A roundtable discussion among employers highlighted some of the barriers, including formulary design and drug pricing and rebates.
