- Safety & Recalls
- Regulatory Updates
- Drug Coverage
- COPD
- Cardiovascular
- Obstetrics-Gynecology & Women's Health
- Ophthalmology
- Clinical Pharmacology
- Pediatrics
- Urology
- Pharmacy
- Idiopathic Pulmonary Fibrosis
- Diabetes and Endocrinology
- Allergy, Immunology, and ENT
- Musculoskeletal/Rheumatology
- Respiratory
- Psychiatry and Behavioral Health
- Dermatology
- Oncology
ICER: Greater Transparency of Coverage Policies Needed
The Institute for Clinical and Economic Review’s fair access report found that for the drugs assessed, formularies provided fair access, but it was difficult to determine how well that translates into real-world access and affordability for patients.
Although payer coverage policies for the 18 drugs that the Institute for Clinical and Economic Review (ICER) assessed often met fair access criteria, there is still a need for transparency about coverage policy information and out-of-pocket costs for patients. These are the findings of ICER’s third Barriers to Fair Access assessment.
This year, ICER also did an exploratory analysis on how transparent plans were from a patient perspective, in other words, could patients and consumers find out information about what drugs were covered and what their costs would be.
Sarah K. Emond

“The numbers here show us that there is a lot more work PBMs and payers can do on making that information available,” Sarah K. Emond, president elect of ICER said in an interview. “Some of this was only behind a provider portal. We shouldn’t expect that every prospective plan member would know that it’s an okay place to go find this information.”
For this analysis, ICER assessed 18 drugs that the organization had reviewed in 2021 for cost-effectiveness against criteria it has determined are cornerstones of fair access. These drugs included:
- Benlysta (belimumab) and Lupkynis (voclosporin) for patients with lupus nephritis;
- Leqvio (inclisiran), Nexletol (bempedoic acid), and Nexlizet (bempedoic acid/ezetimibe) for patients with high cholesterol;
- Abecma (idecabtagene vicleucel) and Carvykti (ciltacabtagene autoleucel) for patients with multiple myeloma;
- Adbry (tralokinumab), Cibinqo (abrocitinib), Opzelura (ruxolitinib), and Rinvoq (upadacitinib) for patients with atopic dermatitis;
- Camzyos (mavacamten) for patients with hypertrophic cardiomyopathy;
- Cinryze (C1 esterase inhibitors), Haegarda (C1 esterase inhibitors), and Tahkzyro (lanadelumab) for patients with hereditary angioedema;
- Tezspire (tezepelumab) for patients with asthma;
- Soliris (eculizumab) and Vyvgart (efgartigimod) for patients with myasthenia gravis.
The criteria used to assess fair access include: cost-sharing, clinical eligibility, step therapy policies, and provider qualification restrictions.
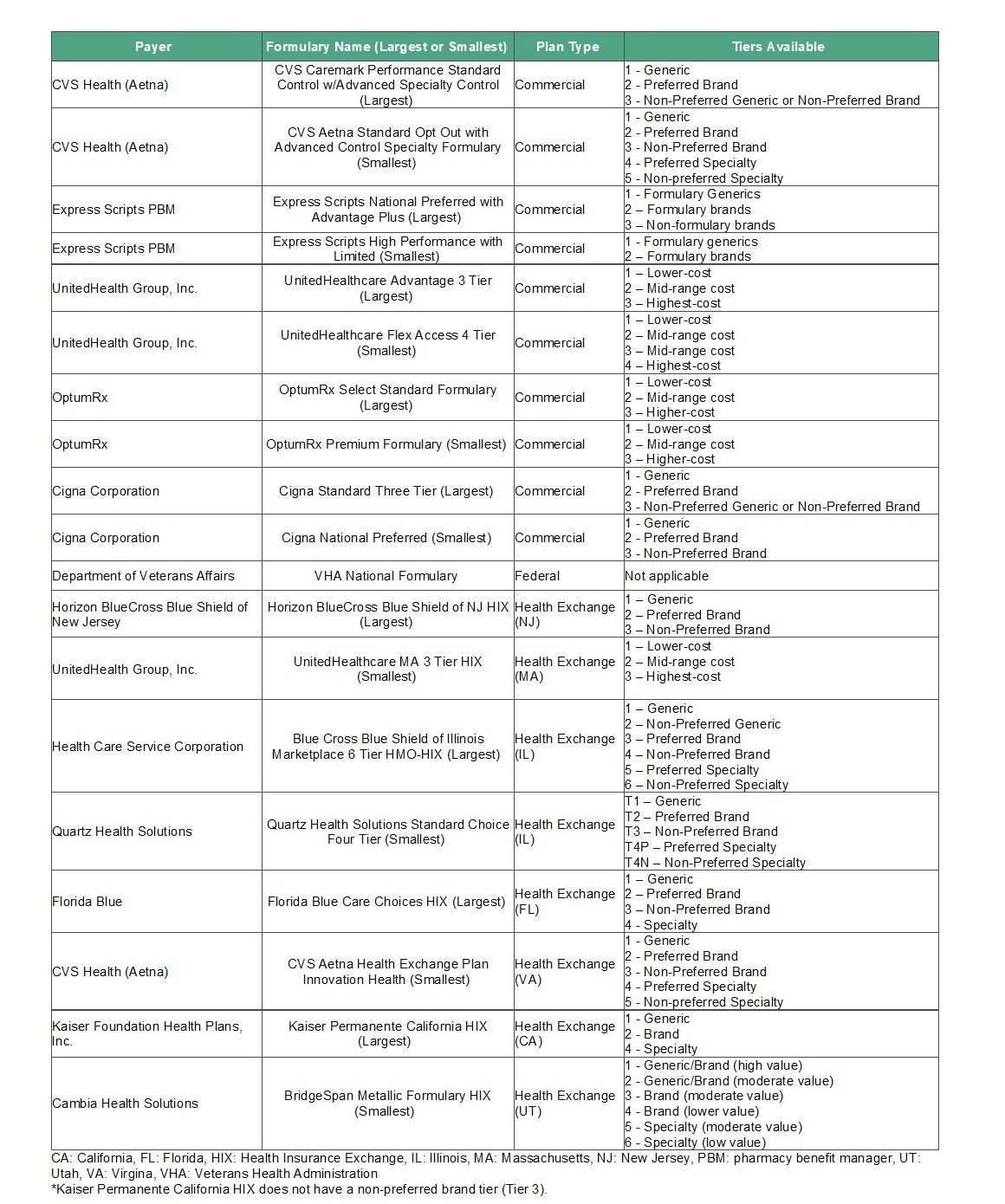
ICER looked at 19 formularies, including the largest and smallest formularies by number of covered lives offered by the five largest commercial payers in the United States, the Veterans Health Administration, and the largest and smallest state health exchange plan formularies. (See Table below.) These formularies represent about 42 million people.
ICER found that concordance with their criteria was 99% for clinical eligibility criteria, 99% for step therapy, and 100% for provider restrictions. Additionally, there were no differences by size of formulary, whether in the private market or in state health exchange plans.
Non-concordance with cost sharing, clinical eligibility, and step therapy criteria was clustered in a few drugs or condition areas, including treatments for lupus nephritis, atopic dermatitis, and myasthenia gravis. Drugs for atopic dermatitis and myasthenia gravis were also more likely to have coverage that did not meet fair access criteria. Although 81% of formularies had clinical criteria that aligned with fair access for Adbry, a drug for moderate-to-severe atopic dermatitis, those formularies lacking concordance often had narrow definitions of “moderate-to-severe” disease.
Alignment with ICER’s criteria for fair cost sharing was 62% across all formularies, but this analysis was limited because were on two drugs in this analysis — Benlysta and Nexlizet — whose net prices were found to meet cost-effectiveness standards. Nexlizet had a 78% concordance rate for cost-sharing.
“Most of the drugs in our 2021 cohort were priced out of line with value, so they’re not subject to this analysis on cost sharing,” Emond said. “Because it’s small, it’s hard to draw conclusions. But there was wide variation among the payers about what tier they were putting those drugs on, some were still putting it on a specialty tier, some were putting it on a non-preferred brand, and some were putting it on a preferred brand.”
Three formularies — Cambia BridgeSpan Metallic Formulary HIX, CVS Aetna Health Exchange Plan Innovation Health, and Express Scripts High Performance — listed the two drugs as nonformulary, and one plan, Horizon BlueCross BlueShield of NJ HIX, included Nexlizet on formulary, but not on the lowest tier.
Benlysta had a 47% concordance rate for cost sharing, with 10 formularies either listing the drug as non-formulary or placing it on a tier that did not represent the lowest relevant tier. Two health plans said that placing Benlysta on the specialty tier was appropriate because a clinical guideline recommends other, less expensive treatments as first-line options.
After ICER shared its analysis with payers, one revised its coverage policy for Abecma in a way that would bring it into alignment with ICER’s fair access criteria.
“My big take home is that we have two things we need to work on as a system,” Emond said. “One is more attention needs to be paid to the grand bargain of a fairly priced drug being on a on a low tier with low cost-sharing. We shouldn’t be punishing patients when the manufacturer has done the right thing with a fairly priced drug.
“The second is that tiering is an inadequate and unsatisfactory substitute for actual cost sharing,” she continued. “Because the plan sponsors — whether that’s the payer, the fully insured market, or a big employer in a self-insured market — are picking the cost sharing amount for that tier, we don’t have a lot of visibility into that.”
A product on a low tier, Emond said, may still have cost sharing that is prohibitive for patients. “We learned that the cost sharing for a preferred brand on tier two could have the exact same cost sharing as a tier-four specialty drug,” Emond said.
The ICER report notes that tiering level and actual out-of-pocket cost are not exact across formularies. Additionally, manufacturer coupons, patient assistance programs, and copay maximizer and accumulator programs complicate this analysis of cost sharing.
“Next year, we plan to do a dive deep into cost sharing because I think this is the crux of the access issue when it comes to payer policies,” Emond said.
Additionally, ICER assessed three drugs for transparency: Nexletol, Rinvoq, and Vyvgart. All plans made cost-sharing and tiering information available for these drugs, and between 70% and 93% of formularies provided clinical criteria. Only 57% of formularies disclosed whether they participate in a copay adjustment program, and just 36% to 64% of all formularies provided information about potential continuation of coverage.
Source: Institute for Clinical and Economic Review