- Safety & Recalls
- Regulatory Updates
- Drug Coverage
- COPD
- Cardiovascular
- Obstetrics-Gynecology & Women's Health
- Ophthalmology
- Clinical Pharmacology
- Pediatrics
- Urology
- Pharmacy
- Idiopathic Pulmonary Fibrosis
- Diabetes and Endocrinology
- Allergy, Immunology, and ENT
- Musculoskeletal/Rheumatology
- Respiratory
- Psychiatry and Behavioral Health
- Dermatology
- Oncology
Opioid abuse and dependence: Treatment review and future options
Pain is a significant medical problem and choosing the appropriate treatment may be complex. Opioids are considered a gold standard in the treatment of pain and as pain management has become a more prominent strategy, opioid dependence has become more frequent. The misuse and abuse of opioids have also increased. Successful management of opioid dependence requires utilization of both pharmacologic and nonpharmacologic treatment.

Key Points
Abstract
Pain is a significant medical problem and choosing the appropriate treatment may be complex. Opioids are considered a gold standard in the treatment of pain and as pain management has become a more prominent strategy, opioid dependence has become more frequent. The misuse and abuse of opioids have also increased. Successful management of opioid dependence requires utilization of both pharmacologic and nonpharmacologic treatment. There are many important factors to consider when treating opioid dependence pharmacologically including selecting the right form of treatment such as abstinence or maintenance with medications, and providing appropriate treatment of withdrawal symptoms during detoxification. Treatment options during withdrawal and maintenance therapy include inpatient and outpatient therapy with the use of drugs such as clonidine, naltrexone, methadone, buprenorphine, and naloxone. Newer abuse-deterrent formulations of opioids have recently become available and more are being developed, and these may help lessen misuse and abuse of medications commonly used in the management of chronic pain. (Formulary. 2010;45:284–291.)
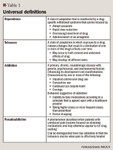
DIFFERENTIATING PHYSICAL DEPENDENCE, TOLERANCE, ADDICTION, AND PSEUDOADDICTION
MECHANISM OF ACTION OF OPIOIDS
Opioids provide analgesia by binding to specific receptors in the brain, spinal cord, and to a lesser degree, the peripheral nervous system. When the opioid binds to the appropriate receptor, transmission of pain impulses is inhibited.10 The 3 main classes of these opioid receptors that constitute the endogenous opioid system are referred to as mu, delta, and kappa.11 This system is involved in many functions including modulation of the response to painful stimuli and stressors; reward and reinforcement; and homeostatic adaptive functions such as regulation of food, water, and temperature.12 Mu receptors are responsible for supraspinal analgesia, respiratory depression, euphoria, sedation, decreased gastrointestinal (GI) motility, and physical dependence. Pure opioid agonists such as morphine, hydromorphone, oxymorphone, fentanyl, codeine, and methadone have a high affinity for mu receptors; a much lower affinity for delta and kappa receptors; and are the most potent analgesics. As the dose is increased, analgesia occurs in a log linear fashion and the degree of analgesia induced is only limited by intolerable dose-related adverse effects.11 In comparison, opioid agonists/antagonists and partial agonists including buprenorphine, nalbuphine, and pentazocine, exhibit a ceiling effect on the degree of analgesia they can produce as they simultaneously induce and block the receptors. The pure antagonists (naloxone and naltrexone) work on mu, kappa, and delta receptors to reverse opioids but the immediate-release action is short-lived.11 In addition, interactions between opioids and mu, kappa, and delta receptors increase dopamine release in the nucleus accumbens and initiate euphoria and other rewarding effects.13,14 While euphoria is not considered harmful, problems with addiction can become an issue if the focus of using the opioid is to achieve this side effect.9
OPIOID-RELATED ADVERSE EFFECTS
Opioids may cause potentially serious adverse effects. Constipation is quite frequent and tends to be persistent with chronic therapy, so patients should be counseled to consider a regimen of increasing fluids and fiber intake with the use of stool softeners and laxatives, if necessary.15,16 Nausea or vomiting is another common adverse effect that typically diminishes over the course of days to weeks of continuous opioid treatment.17 Sedation or inability to concentrate also tends to decrease over time; however, it can be more severe when combined with other drugs that are associated with sedation. Other common adverse effects include pruritis, myoclonus, and respiratory depression. Respiratory depression is of particular concern when initial opioid doses are too high, opioids are titrated too rapidly, or they are combined with other drugs that are associated with respiratory depression.17 With long-term opioid use, hyperalgesia, decreased libido, depression, decreased levels of testosterone, and other hormonal effects also are observed.18
SYMPTOMS OF OPIOID WITHDRAWAL
Withdrawal is a necessary first step for long-term treatment of opioid dependence.19 The most common signs and symptoms associated with withdrawal include irritability, anxiety, chills, nausea, diarrhea, sweating, sneezing, yawning, rhinorrhea, bone and muscle weakness, abdominal pains, and insomnia.19 The signs and symptoms of opioid withdrawal typically begin 2 to 3 half-lives after the last dose and reach peak intensity within 2 to 4 days. Signs and symptoms usually are not present until 36 to 48 hours after the last dose of methadone due to its longer half-life. Generally, most physical withdrawal signs are no longer obvious after 7 to 14 days.19 Symptoms of opioid withdrawal may be quite unpleasant, but are typically not life-threatening.15
CHARACTERISTICS OF OPIOID DEPENDENCE
A person who shows 3 or more of the following behaviors over a 12-month period is most likely opioid-dependent: tolerance to opioid therapy; has withdrawal symptoms when opioids are not used; takes other drugs to help relieve withdrawal symptoms; takes larger amounts of opioids than planned or prescribed and for longer periods of time; has a persistent desire to or unsuccessful attempts to quit; is spending a lot of time and effort to obtain, use, and recover from opioid use; gives up or reduces social or recreational activities; misses work; or continues to use opioids regardless of negative consequences.20
Opioid maintenance treatment and detoxification are the 2 general clinical treatment paths from which to choose.21 Most opioid-dependent individuals engage in both, in some cases multiple times. Success of the various treatment approaches and combinations of treatments is assessed in a number of different ways with the primary outcomes being retention in treatment and continued abstinence, and secondary outcomes including the reduction in harmful behaviors such as human immunodeficiency virus risk behaviors, illegal or criminal activity, and morbidity.21
TREATMENT SETTINGS
Inpatient hospital settings, outpatient treatment programs, and outpatient provider offices may all be used for managing opioid dependence. Treatment options involve acute detoxification and withdrawal followed by the maintenance phase. Opioid dependence treatment should include both pharmacologic and nonpharmacologic therapy. The facility of choice depends on the patient's perceived treatment needs and the clinical characteristics and preferences of the patient. The American Psychiatric Association (APA) guidelines identify the following treatment modalities as effective options for managing opioid dependence and withdrawal: Opioid substitution with methadone followed by a gradual taper; abrupt opioid discontinuation with the use of clonidine to suppress withdrawal symptoms; clonidine-naltrexone detoxification; buprenorphine substitution followed by abrupt or gradual discontinuation of buprenorphine; and use of other medications to treat the symptoms of opioid withdrawal.7
The inpatient hospital setting provides treatment for severe acute opioid overdoses that require emergency medical management to treat respiratory depression. Once the acute symptoms resolve, the patient can be treated for symptoms of opioid withdrawal. Inpatient use of clonidine and/or naltrexone detoxification is preferred, in order to closely monitor the patient.
The first treatment modality for opioid dependence was methadone replacement and it became widely available in the late 1960s.22 Currently, methadone for detoxification and outpatient maintenance of opioid dependence can only be provided by Opioid Treatment Programs that are registered with the Drug Enforcement Administration (DEA) and certified by the Federal Substance Abuse and Mental Health Services Administration.7 These are known as the federal and state licensed methadone clinics. As an exception, patients can continue to receive methadone maintenance therapy if admitted to the hospital for a condition other than opioid dependence if they require temporary methadone maintenance during the stay.7,23

Maintenance therapy with methadone or buprenorphine is appropriate for use in patients who have a history of dependence longer than 1 year.7 The availability of office-based treatment programs is a breakthrough for access to care for patients who cannot travel long distances to obtain treatment or who have difficulty attending daily outpatient programs.7
TREATMENT APPROACHES FOR OPIOID DEPENDENCE WITHDRAWAL
In the treatment of opioid dependence, clonidine and naltrexone are primarily used during acute opioid withdrawal therapy, but also have a role in maintenance therapy.
Alpha-2 agonist. Clonidine is generally considered to be a safe, non-narcotic medication used to help patients withdrawing from opioids. Typically used as an antihypertensive medication, it is a centrally acting alpha-2 agonist and works to minimize the noradrenergic hyperactivity seen with opioid withdrawal.7,26 Although not approved in the United States for opioid withdrawal, clonidine is administered for off-label use at various doses. Typical dosing is 0.1 mg to 0.3 mg up to every 6 hours orally, though patches have also been used.7,27 One advantage of clonidine is that it does not produce dependence or tolerance as do opioid medications and it can be given with naltrexone if needed.7 Contraindications to clonidine use include cardiac disorders, hypotension, and renal dysfunction. If clonidine-assisted detoxification is going to be provided in the outpatient setting, it should be under the guidance of experienced staff and the patient should not receive more than a 3-day supply of the medication because clonidine overdoses can be life-threatening.7
Opioid antagonist. In the United States, naltrexone is approved for the treatment of opioid dependence and is not a controlled substance. Naltrexone is considered a mu-receptor antagonist. It also acts as an antagonist at the kappa-receptor and weakly antagonizes the delta-receptor.11 The opioid antagonism is produced by naltrexone's active metabolite, 6-ß-naltrexo. Naltrexone is usually dosed between 50 mg and 100 mg daily or may be dosed 3 times per week because of its long duration of action (100 mg on Monday and Wednesday and 150 mg on Friday) for opioid withdrawal.7,27 This combination technique is useful in helping transition a patient to opioid antagonist treatment. The combination requires patients be monitored closely for the first 8 hours because of potential nal-trexone-induced withdrawal and for blood pressure monitoring.
MAINTENANCE PHASE TREATMENT OPTIONS
The maintenance phase is designed to keep patients opioid free. The maintenance phase also could include nonpharmacologic therapy to prevent relapse. Counseling services have an effect on outcomes in patients receiving pharmacologic treatment, with increases in the number and duration of services producing improved outcomes.27
Agonists. FDA approved methadone for the treatment of moderate to severe pain that is not responsive to non-narcotic pain medications, for the detoxification of opioid addiction, and for the maintenance treatment of opioid dependence in conjunction with medical and social services.23 Methadone is a Schedule II narcotic available in 5-mg and 10-mg tablets, a 40-mg dispersible wafer, and a 10-mg/ml liquid concentrate.28 It is an opioid receptor agonist at the mu receptor, but also an antagonist at the N-methyl-d-aspartate receptor.
Following oral administration, methadone has a bioavailability ranging from 36% to 100%, with peak plasma concentrations occurring between 1 and 7.5 hours.23 It is also 85% to 90% protein bound in plasma. Methadone is primarily metabolized by CYP3A4 and secondarily by CYP2D6. However, it is also metabolized by CYP2B6, CYP2C19, and CYP2C9. Macrolide antibiotics, fluvoxamine, clopidogrel, raloxifene, sertraline, and other inhibitors of those enzymes may increase methadone levels, enhancing its toxicities and effects. On the other hand, enzyme inducers such as phenobarbital, phenytoin, rifampin, and St. John's Wort may decrease levels of methadone, potentiating withdrawal symptoms.11,23 Providers and pharmacists should evaluate each patient's medication profile to identify potential drug interactions before adjusting methadone dose. A black box warning was added to methadone's prescribing information due to the risk of cardiac and respiratory-related deaths after the initiation of methadone treatment. Most of the cases of respiratory depression, QT prolongation, and cardiac arrhythmias (torsades de pointes) have occurred in patients on high doses of methadone, but have also been observed when it is used for maintenance of opioid dependence.23,29 Because of this warning, a baseline and follow-up electrocardiogram should be obtained.
When using methadone for opioid dependence and detoxification, the treatment period is usually at least 12 months and may continue for years. The length of treatment is patient dependent. The typical starting dose of 20 to 30 mg once daily is generally sufficient to suppress withdrawal symptoms. The typical maintenance dose range is between 80 and 120 mg per day, with adjustments being made based on withdrawal symptoms.23 Methadone is well absorbed orally, and with an average half-life of 15 to 25 hours, it allows for once-daily dosing. Common adverse effects associated with methadone use are sweating, constipation, decreased libido, weight gain, and irregular menstrual periods.28 Patients should be informed of the risk of addiction and abuse associated with methadone use, as well as the signs and symptoms of respiratory depression.
Partial agonists. Buprenorphine is a partial mu receptor agonist and acts as an antagonist at the kappa receptor. The receptor interactions are thought to account for buprenorphine's clinical implications in treating opioid dependence.7 The activity at the kappa receptor provides analgesic activity and provides benefits for use in opioid deterrence, maintenance, and detoxification.24,30 Because of the partial activation of the mu receptor, patients are less likely to abuse buprenorphine. The slow rate of dissociation along with the high rate of affinity for the mu receptor may block the effects of other opioids by displacing those other agents from the receptor.7,11,30
Treatment with buprenorphine is usually required for at least 6 months and as with methadone, may continue for years. Buprenorphine is available as 2-mg and 8-mg sublingual tablets as a single agent and is also available in combination with naloxone in 2-mg buprenorphine/0.5-mg naloxone or 8-mg buprenorphine/2-mg naloxone sublingual tablets.30 The sublingual forms of buprenorphine are the only approved dosage forms for office-based opioid treatment and are Schedule III controlled substances.24 The typical dose range for buprenorphine is 2 to 32 mg per day, with the average daily dose being about 16 mg. Buprenorphine has a long duration of action and may be dosed at longer intervals. When used in this manner, dose administration should be increased to compensate for the longer interval by doubling daily dose for a 48-hour interval or tripling the dose for a 72-hour interval.7 The most common side effects include nonspecific headache and constipation. However, there is some evidence that buprenorphine can produce mild elevations in liver function tests.7,30
Naloxone is a competitive antagonist at the mu and kappa receptors, with its primary action at the mu receptor. The bioavailability after oral or sublingual dosing is low, but with parenteral administration, it has a rapid onset of action that leads to rapid reversal of opioid effects.11,30 The buprenorphine-naloxone combination significantly reduces the risk of the medicine being diverted because naloxone will exert a potent opioid antagonist effect if the combination tablet is crushed and administered intravenously by an opioid-dependent patient.7,30
Due to high lipid solubility, this medication has excellent sublingual bioavailability with an onset of action within 30 to 60 minutes and a peak effect in approximately 90 minutes.24 The half-life of buprenorphine is approximately 37 hours and it is metabolized through the CYP3A4 isoenzyme. Patients concurrently taking CYP3A4 inhibitors or inducers should be closely monitored and dose adjustments may need to be made.30 Because of the extensive hepatic metabolism of both buprenorphine and naloxone, dosage adjustments may be considered in patients with decreased liver function along with monitoring for signs and symptoms of opioid withdrawal due to possibly elevated levels of naloxone.30
Regardless of the treatment strategy used, all medications appear to be efficacious for the treatment of opioid dependence. Treatment should be based on overall health status, patient preference, past response to treatment, and the probability of the patient achieving and maintaining abstinence.7
FUTURE TREATMENTS AND ABUSE-DETERRENT FORMULATIONS
Abuse is defined as the intentional self-administration of a medication for a nonmedical purpose.31 Although misuse and abuse frequently begin with overuse of oral opioids, nonmedical users often progress over time to manipulating the formulation or altering the route of delivery to extract more active ingredient and achieve more rapid euphoria, which may further enhance toxicity.32 The brain is particularly sensitive to toxicity because it is one of the most metabolically active tissues. Drugs of abuse may compromise important brain functions and should be taken into account when managing patients with chronic opioid therapy.33 Many clinical studies have demonstrated that chronic drug use changes the brain in fundamental ways and that the changes persist long after the individual has stopped taking the drug.34 Some research shows that opioid drug users display pronounced neuropsychological impairment in the domains of executive and memory function. Even after several years of drug abstinence, these impairments persist.35 These potentially life-long impairments necessitate the need for abuse-deterrent formulations.
Abusers will often crush tablets and smoke, snort, or inject them intravenously.32 Some extended-release products provide a theoretical lower potential for misuse and abuse due to the slower onset of action, however, longer acting products may be appealing to the nonmedical user because of the higher quantity of active opioid ingredient per dose.36 One approach in developing abuse-deterrent formulations includes incorporating a physical barrier that resists chewing, grinding, and mixing with alcohol or other solvents. Other mechanisms being developed in an attempt to reduce misuse and abuse include opioid agonist/antagonist combinations, and prodrugs, which require biotransformation in the GI tract in order for the opioid to become active.36
For example, a tamper-resistant form of oxycontin (OTR Oxycontin) received FDA approval with a required Risk Evaluation and Mitigation Strategy in April 2010. The new formulation prevents cutting, breaking, chewing, or crushing and when dissolved, forms a gel-like substance unsuitable for injection.37 Embeda, an extended-release opioid analgesic was approved by FDA in August 2009. This formulation consists of morphine sulfate pellets with an inner core of naltrexone. As a deterrent, if Embeda is crushed or chewed, naltrexone is released and absorbed with morphine, reversing morphine's effects.38
More recently in August 2010, FDA approved Suboxone (Reckitt Benckiser Pharmaceuticals) sublingual film. The proprietary PharmFilm technology offers a fast-dissolving sublingual film that cannot readily be crushed into a powder to snort.39 If this agent is extracted for injection, the naloxone component attenuates the effects of buprenorphine and precipitates acute withdrawal symptoms in opioid-dependent patients. It is also thought to decrease abuse potential by offering a dissolution time nearly twice that of Suboxone tablets, which may help patients be more compliant with treatment.39
Controlled-release high-viscosity liquid matrix oxycodone is one option under development. It is a long-acting formulation of oxycodone in an encapsulated liquid matrix that resists physical manipulations, including breaking, crushing, chewing, and extraction with common solvents such as alcohol or water.36 The viscosity of the formula makes it unsuitable for injection because it cannot be drawn into or expressed from a needle. FDA rejected a New Drug Application (NDA) for this oxycodone formulation in 2009 but a resubmission is expected by the end of 2010 after additional data on the stability of the formula is compiled. An oral sustained-release formula of oxycodone that resists injection and nasal inhalation, but is also designed to resist release of the entire dose when consumed with alcohol, is also under development.
Collegium Pharmaceuticals, Inc., is using a patented delivery system (DETERx) consisting of a multi-particulate matrix formulation enclosed in a capsule. The particles resist chewing, crushing, snorting, and extraction for use as an injection but can also be removed from the capsule and sprinkled on food for patients with difficulty swallowing. The delivery system is currently applied to an extended-release oxycodone (COL-003) and an extended release opioid formula (COL-172) that have both received FDA Fast Track Designation.36
Similarly, Egalet has developed an abuse-deterrent prolonged-release erosion matrix (ADPREM) tablet that is difficult to dissolve, crush or chew, is not easily extracted, will melt into a highly viscous gel, and is poorly combustible. An ADPREM extended-release morphine tablet is being evaluated in a phase 2 clinical trial and the technology is also being used to develop oxycodone, hydrocodone, and hydromorphone tablets.40 An agonist/antagonist combination of oxycodone and naltrexone (ELI-126) has completed phase 2 clinical trials and has an abuse-deterrent mechanism similar to the recently approved combination of extended-release morphine and naltrexone. When used as indicated, naltrexone remains latent; if crushed, the antagonist is released and reduces the effects of the agonist.36 More recently, the FDA has voted in support of using extended-release injectable naltrexone (Vivitrol) for the treatment of opioid dependence. While this product is not being marketed as a deterrent, it may offer more successful recovery for those opioid dependent patients seeking once-monthly medication therapy.41
Formulas of immediate-release oxycodone, immediate-release oxycodone and acetaminophen, and hydrocodone bitartrate and acetaminophen are being developed using technology that deters injecting and snorting by forming a viscous gel mixture when the tablet is dissolved.36,42,43 An NDA was filed for an immediate-release oxycodone formula using this technology in conjunction with niacin that was thought to incorporate the additional deterrent effects of flushing, itching, and sweating. However, in April 2010 the FDA's Anesthetic and Life Support Drugs and Drug Safety and Risk Management Advisory Committees voted 19 to 1 not to recommend approval primarily because the adverse effects of niacin could be easily avoided by taking the agent with food.42
Finally, a vaccine for the treatment of opioid dependence is in the early stages of development. It is being designed to prevent addictive substances from ever reaching the abuser's brain and prolongs the time between clinician visits. Vaccines for nicotine and cocaine addiction are further in development than for opioids, but perhaps this will be a treatment option in years to come.43
Opioids with a reduced abuse potential are one of the most important unmet needs in the management of chronic pain.44 Whether through physical barrier or pharmacodynamic means, abuse-deterrent and -resistant formulations may help meet this need. Successful products may encourage clinicians to prescribe these medications for more patients, which could enhance and increase treatment of pain, promote better patient compliance, and reduce adverse health outcomes.45 While these new agents could revolutionize the way prescribers manage chronic pain, cost and access to these medications still remain a concern.44
CONCLUSION
Opioid abuse and dependence are complex medical problems that healthcare professionals are faced with on a daily basis. Recent studies show that adverse drug events, emergency department visits, and even death are on the rise in some states as a result of increasing use and misuse of opioids.46 Providers should be aware that there are appropriate treatment options for these patients including opioid maintenance treatment and detoxification. It is important to adhere to currently accepted guidelines and recommendations for treating opioid-dependent patients, including the use of pharmacologic and nonpharmacologic therapies. Because all recommended treatment options are considered effective for the treatment of opioid dependence, treatment should be based on overall health status, patient preference, past response, and probability of success.7 Successful treatment is evaluated in different ways including retention in treatment, abstinence, and reduction in harmful lifestyle behaviors. Regulations and treatment modalities such as abuse-deterrent formulations likely are to continue to evolve in response to the growing incidence of dependence.
Dr Woods is clinical associate professor of pharmacy practice and Dr Hilaire is clinical associate professor of pharmacy practice, University of Wyoming School of Pharmacy, Laramie.
Disclosure Information: The authors report no financial disclosures as related to products discussed in this article.
REFERENCES
1. American Pain Society. Mission statement. Glenview, IL. Published 2001. Available at: http://www.ampainsoc.org/ce/cme_mission.htm. Accessed October 6, 2010.
2. Olsen Y, Daumit GL, Ford DE. Opioid prescriptions by U.S. primary care physicians from 1992 to 2001. J Pain. 2006;7(4):225–235.
3. Walwyn WM, Miotto KA, Evans CJ. Opioid pharmaceuticals and addiction: the issues, and research directions seeking solutions. Drug Alcohol Depend. 2010;108:156–165.
4. Caudill-Slosberg MA, Schwartz LM, Woloshin S. Office visits and analgesic prescriptions for musculoskeletal pain in US: 1980 vs. 2000. Pain. 2004;109:514–519.
5. Colliver JD, Kroutil LA, Dai L, Gfroerer JC. Misuse of prescription drugs: data from the 2002, 2003, and 2004 National Surveys on Drug Use and Health. DHHS Publication No. SMA 06-4192, Analytic Series A-28. 2006 Rockville, MD. Substance Abuse and Mental Health Services Administration, Office of Applied Studies. Available at: http://www.oas.samhsa.gov/prescription/Cover.htm. Accessed October 6, 2010.
6. National Center on Addiction and Substance Abuse (CASA) at Columbia University. You've Got Drugs! Prescription Drug Pushers on the Internet: 2006 Update. A CASA white paper. New York, NY. Published 2006. Available at: http://www.casacolumbia.org/templates/publications_reports.aspx?keywords=white+paper. Accessed October 6, 2010
7. American Psychiatric Association. Practice Guidelines for the treatment of patients with substance abuse disorders, 2nd ed. Published 2006. Available at: http://www.psychiatryonline.com/content.aspx?aID=142306. Accessed October 6, 2010.
8. American Pain Society. Definitions related to the use of opioids for the treatment of pain. Glenview, IL. Published 2006. Available at: http://www.ampainsoc.org/advocacy/opioids2.htm. Accessed October 6, 2010.
9. Savage SR, Kirsh KL, Passik SD. Challenges in using opioids to treat pain in persons with substance use disorders. Addict Sci Clin Pract. 2008;4(2):4–25.
10. Inturrisi CE. Clinical pharmacology of opioids for pain. Clin J Pain. 2002;18:S3–S13.
11. Trescot AM, Boswell MV, Atluri SL, et al. Opioid guidelines in the management of chronic non-cancer pain. Pain Physician. 2006;9:1–40.
12. Olson GA, Olson RD, Kastin AJ. Endogenous opiates: 1989. Peptides. 1990;11:1277–1304.
13. Koob GF. Drugs of abuse: anatomy, pharmacology, and function of reward pathways. Trends Pharmacol Sci. 1992;13(5):177–184.
14. Gianoulakis C. Endogenous opioids and addiction to alcohol and other drugs of abuse. Curr Top Med Chem. 2009;9(11):999–1015.
15. Chou R. 2009 Clinical guidelines from the American Pain Society and the American Academy of Pain Medicine on the use of chronic opioid therapy in chronic noncancer pain. Pol Arch Med Wewn. 2009;119(7-8):469–477.
16. Amabile DM, Bowman BJ. Overview of oral modified-release opioid products for the management of chronic pain. Ann Pharmacother. 2006;40:1327–1335.
17. Chou R, Fanciullo GJ, Fine PG, et al. Clinical guidelines for the use of chronic opioid therapy in chronic noncancer pain. J Pain. 2009;10:113–130.
18. Jan SA. Introduction: landscape of opioid dependence. JMCP. 2010;16(1b):S4–S8.
19. Gowing L, Farrell M, Ali R, White JM. Alpha2 adrenergic agonists for the management of opioid withdrawal. Cochrane Database of Systematic Reviews (online), 4, CD002024. Available at: http://mrw.interscience.wiley.com/cochrane/clsysrev/articles/CD002024/frame.html. Accessed October 6, 2010.
20. American Psychiatric Association. DSM-IV Criteria for substance dependence and abuse. In: Diagnostic and Statistical Manual of Mental Disorders: DSM-IV-TR. 4th ed. Test Revision. Washington, DC: APA, 2000.
21. Stotts AL, Dodrill CL, Kosten TR. Opioid dependence treatment: options in pharmacotherapy. Expert Opin Pharmacother. 2009;10(11):1727–1740.
22. Gouldin WM, Kennedy DT, Small RE. American Pain Society. Methadone: History and recommendations for use in analgesia. APS Bulletin. 2000:10(5). Available at: http://www.ampainsoc.org/pub/bulletin/sep00/upda1.htm. Accessed October 6, 2010.
23. Dolophine hydrochloride [package insert]. Columbus, OH: Roxane Laboratories, Inc; 2006.
24. Helm S, Trescot AM, Colson J, et al. Opioid antagonists, partial agonists, and agonists/antagonists: the role of office-based detoxification. Pain Physician. 2008;11(2)225–235.
25. Department of Health and Human Services. Substance Abuse and Mental Health Services Administration. Center for Substance Abuse Treatment. Drug Addiction Treatment Act of 2000 (DATA 2000). Title XXXV, Section 3502. Available at: http://buprenorphine.samhsa.gov/titlexxxv.html. Accessed October 6, 2010.
26. Amass L, Ling W, Freese TE, et al. Bringing buprenorphine-naloxone detoxification to community treatment providers: the NIDA clinical trials network field experience. Am J Addict. 2004(suppl 1):S42–S66.
27. Fiellin DA, O'Connor PG. Office based treatment of opioid-dependent patients. New Eng J Med. 2002.347(11):817–823.
28. Krantz MJ, Mehler PS. Treating opioid dependence: growing implications for primary care. Arch Intern Med. 2004;164(4):277–288.
29. Anchersen K, Causen T, Gossop M, et al. Prevalence and clinical relevance of corrected QT interval prolongation during methadone and buprenorphine treatment: a mortality assessment study. Addiction. 2009;104(6):993–999.
30. Suboxone [package insert]. Berkhire, UK; Reckitt Benckiser Pharmaceuticals, Inc: 2006.
31. Katz NP, Adams EH, Chilcoat H, et al. Challenges in the development of prescription opioid abuse-deterrent formulations. Clin J Pain. 2007;23:648–660.
32. Hays LR. A profile of oxycontin addiction. J Addict Dis. 2004;23(4):1–9.
33. Cunha-Oliveira T, Rego AC, Oliveira CR. Cellular and molecular mechanisms involved in the neurotoxicity of opioid and psychostimulant drugs. Brain Res Rev. 2008;58:192–208.
34. Leshner AI. Drug addiction research: moving toward the 21st century. Drug Alcohol Depend. 1998;51:5–7.
35. Ersche KD, Clark L, London M, Robbins TW, Sahakian BJ. Profile of executive and memory function associated with amphetamine and opioid dependence. Neuropsychopharmacology. 2006;31:1036–1047.
36. Webster L. Update on abuse-resistant and abuse-deterrent approaches to opioid formulations. Pain Med. 2009;10(S2):S124–S133.
37. FDA. 2010. Available at: http://www.fda.gov/NewsEvents/Newsroom/PressAnnouncements/ucm207480.htm. Accessed August 22, 2010.
38. Embeda [package insert]. Bristol, TN; King Pharmaceuticals, Inc: 2009.
39. Reckitt Benckiser Pharmaceuticals Inc. Advances in treatment. Available at: http://www.suboxone.com/hcp/about_suboxone/key_benefits.aspx. Accessed October 6, 2010.
40. Egalet Inc. Egalet innovators in pain management. Corporate presentation. 2008. Available at: http://www.egalet.com/multimedia/Egalet_Corp_Presentation_200913.pdf. Accessed October 6, 2010.
41. Alkermes Inc. Alkermes announces FDA advisory committee recommends approval of VIVITROL for opioid dependence. Available at: http://investor.alkermes.com/phoenix.zhtml?c=92211&p=RssLanding&cat=news&id=1472331. Accessed October 6, 2010.
42. Acura Pharmaceuticals Inc. King Pharmaceuticals and Acura Pharmaceuticals announce Plans to Submit an NDA for Acurox® Tablets without Niacin. 2010. Available at: http://www.kingpharm.com/Investors/News_Details.cfm?news_item_id=554. Accessed October 6, 2010.
43. Orson FM. The future of vaccines in the management of addictive disorders. Curr Psychiatry Rep. 2007;9:381–387.
44. Glabman M. Multiple therapies help curb opioid dependence. Manag Care. 2009;1(suppl 1):14–20.
45. King Pharmaceuticals Inc. News room. Available at: http://www.kingpharm.com/Products/index.cfm. Accessed October 6, 2010.
46. Braden JB. Emergency department visits among recipients of chronic opioid therapy. Arch Intern Med.2010;170(16):1425–1432.
Coalition promotes important acetaminophen dosing reminders
November 18th 2014It may come as a surprise that each year Americans catch approximately 1 billion colds, and the Centers for Disease Control and Prevention estimates that as many as 20% get the flu. This cold and flu season, 7 in 10 patients will reach for an over-the-counter (OTC) medicine to treat their coughs, stuffy noses, and sniffles. It’s an important time of the year to remind patients to double check their medicine labels so they don’t double up on medicines containing acetaminophen.
Support consumer access to specialty medications through value-based insurance design
June 30th 2014The driving force behind consumer cost-sharing provisions for specialty medications is the acquisition cost and not clinical value. This appears to be true for almost all public and private health plans, says a new report from researchers at the University of Michigan Center for Value-Based Insurance Design (V-BID Center) and the National Pharmaceutical Council (NPC).
Management of antipsychotic medication polypharmacy
June 13th 2013Within our healthcare-driven society, the increase in the identification and diagnosis of mental illnesses has led to a proportional increase in the prescribing of psychotropic medications. The prevalence of mental illnesses and subsequent treatment approaches may employ monotherapy as first-line treatment, but in many cases the use of combination of therapy can occur, leading to polypharmacy.1 Polypharmacy can be defined in several ways but it generally recognized as the use of multiple medications by one patient and the most common definition is the concurrent use of five more medications. The presence of polyharmacy has the potential to contribute to non-compliance, drug-drug interactions, medication errors, adverse events, or poor quality of life.
Medical innovation improves outcomes
June 12th 2013I have been diagnosed with stage 4 cancer of the pancreas, a disease that’s long been considered not just incurable, but almost impossible to treat-a recalcitrant disease that some practitioners feel has given oncology a bad name. I was told my life would be measured in weeks.
