- Safety & Recalls
- Regulatory Updates
- Drug Coverage
- COPD
- Cardiovascular
- Obstetrics-Gynecology & Women's Health
- Ophthalmology
- Clinical Pharmacology
- Pediatrics
- Urology
- Pharmacy
- Idiopathic Pulmonary Fibrosis
- Diabetes and Endocrinology
- Allergy, Immunology, and ENT
- Musculoskeletal/Rheumatology
- Respiratory
- Psychiatry and Behavioral Health
- Dermatology
- Oncology
Outpatient pharmacologic management of ACS using antiplatelet therapy: A review of recent guidelines and agents in the pipeline
Acute coronary syndrome (ACS) encompasses a series of heart conditions associated with myocardial ischemia.

Key Points
Abstract
Acute coronary syndrome (ACS) encompasses a series of heart conditions associated with myocardial ischemia. Three conditions are classified broadly as ACS: ST–segment elevation myocardial infarction (STEMI), non-ST-segment elevation MI (NSTEMI), and unstable angina (UA). According to American College of Cardiology (ACC)/American Heart Association (AHA) guidelines, aspirin is the cornerstone of long-term antiplatelet therapy in the management of ACS after hospital discharge; this agent should be continued indefinitely in the absence of any contraindications. The use of adjunctive clopidogrel after hospital discharge is also an established element of ACS management. Antiplatelet agents with novel mechanisms of action are currently being evaluated and compared with the current standard of care. The agents under investigation (prasugrel, ticagrelor, and SCH 530348) will need to demonstrate substantial improvements in their efficacy, safety, and tolerability profile compared with current treatments to gain a place in therapy. (Formulary. 2009;44:22–27.)
Acute coronary syndrome (ACS) encompasses a variety of heart conditions associated with myocardial ischemia. Cardiac ischemia occurs as a result of insufficient blood supply to the heart because of occlusion and often leads to myocardial infarction (MI). Three conditions are classified broadly as ACS: ST-segment elevation MI (STEMI), non-ST-segment elevation MI (NSTEMI), and unstable angina (UA). Specific criteria are measured for each condition when healthcare professionals make a differential diagnosis.
Per the American Heart Association (AHA) Statistics Committee, in conjunction with the Centers for Disease Control and Prevention (CDC), the National Institutes of Health (NIH), and other government agencies, an estimated 770,000 individuals will have had a new coronary attack in America in 2008.3 An estimated 430,000 Americans will have had a recurrent heart attack in the same year. In 2005, the National Center for Health Statistics along with the National Hospital Discharge Survey reported 1,413,000 hospitalizations for primary or secondary diagnosis of ACS, of which 838,000 were for MI alone and 558,000 were for UA alone (17,000 received both diagnoses).3 The estimated proportion of patients with MI classified as STEMI ranges from 29% to 47% depending on the registry and surveillance type (eg, National Registry of Myocardial Infarction 4 [NRMI-4], Global Registry of Acute Coronary Events [GRACE], Euro Heart Survey on ACS [EHS-ACS-II]). Given the high prevalence of ACS in the United States, the dire need for appropriate management and follow-up after hospital discharge is beyond question. There are numerous pharmacologic agents used in the outpatient management of ACS. Given the patient's individual clinical status, treatment options after discharge may include but are not limited to beta-adrenergic blockers, angiotensin-converting enzyme (ACE) inhibitors, lipid-lowering agents, anticoagulants if applicable, and antiplatelet drugs.
Atherosclerotic plaque rupture and aggregating platelets are primary factors in the pathophysiology of ACS. As such, it is necessary to understand the purpose and place of each of the various antiplatelet medications in the treatment of ACS by reviewing current guidelines as well as the emerging clinical studies evaluating the use of novel antiplatelet medications.
CURRENT ANTIPLATELET AGENTS FOR THE OUTPATIENT MANAGEMENT OF ACS
Aspirin, an integral component of antiplatelet therapy, is used in the management of ACS and prophylaxis of MI and exerts its activity through its inhibition of cyclooxygenase-dependent platelet activation and prostaglandin blockade. As a result, the formation of thromboxane A2 is prevented, and thus the otherwise enduring process of platelet stimulation is stopped.4 The recommended dosage of aspirin for MI prophylaxis is 75 to 325 mg/d; this dosage is typically continued indefinitely.
Clopidogrel is a widely accepted choice for adjunctive treatment of ACS. This agent has been demonstrated to reduce the rate of atherothrombotic events (eg, re-infarction) and death in patients with STEMI being treated medically.5 Clopidogrel is also used for the reduction in the rate of atherothrombotic events in patients with NSTEMI/UA who are being treated medically or by percutaneous coronary intervention (PCI). Clopidogrel is a thienopyridine that works through selective and irreversible inhibition of adenosine diphosphate (ADP) P2Y12 receptor binding, which thereby prevents the onset of the GPIIb/IIIa receptor complex and diminishes platelet aggregation.5,6 Outpatient dosing for clopidogrel is a single 75-mg tablet taken daily; the exact duration of therapy depends on presence and type of stenting.
Ticlopidine is usually reserved for the setting of stroke or is administered concomitantly with aspirin after stent placement to decrease the risk of subacute stent thrombosis.7 However, this agent is also a viable alternative for patients who have a contraindication to aspirin. Ticlopidine is associated with a risk of life-threatening hematologic disorders and should be administered with caution, with routine monitoring performed if ticlopidine use is a necessity. Also a thienopyridine, ticlopidine exerts its pharmacologic effects by irreversibly altering platelet membranes through the prevention of ADP stimulation and the prevention of platelet-fibrinogen binding.8 Unfortunately, ticlopidine also increases bleeding time. When indicated, ticlopidine is to be taken as a 250-mg dose twice daily with food.
Dipyridamole is an antiplatelet agent that is not approved by FDA for the management of ACS, nor has it shown a promising role in the treatment of ACS. Its main use is to decrease thrombosis after artificial heart valve placement.9 Dipyridamole blocks platelet aggregation and may also cause vasodilation by inhibiting adenosine deaminase and phosphodiesterase. There also exists a combination product on the market that combines immediate-release aspirin 25 mg with extended-release dipyridamole 200 mg. However, the overall use of this combination product is restricted to reduction in risk of stroke for patients who have had transient brain ischemia or completed ischemic stroke; this agent is not commonly used in the treatment of ACS.
RECOMMENDATIONS FOR TREATMENT
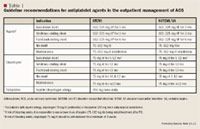
The 2007 updated guidelines also explicitly address the use of clopidogrel. Historically, guidelines have recommended treatment with clopidogrel for 1 to 6 months after stent placement (depending on the type of stent); however, this recommendation has been expanded. The guidelines now recommend that clopidogrel 75 mg should be administered for ≥1 month and ideally up to 12 months to those patients who receive a bare metal stent after PCI, with a minimum of 2 weeks of clopidogrel therapy if there is a concern about bleeding. For patients having a drug-eluting stent placed (ie, sirolimus or paclitaxel), clopidogrel 75 mg should be administered daily for 12 months if the risk of bleeding has been ruled out.
Newly added recommendations for clopidogrel also call for ≥14 days of clopidogrel treatment in patients who do not undergo stent placement after STEMI, regardless of whether reperfusion with fibrinolytics was done. Although long-term maintenance treatment with clopidogrel is recognized as reasonable in patients with STEMI who did not undergo stenting, this idea is not supported by data from randomized controlled trials beyond 30 days.
The 2004 ACC/AHA guidelines for STEMI recommended daily use of clopidogrel 75 mg as a preferred substitution for patients with aspirin resistance or allergy. Ticlopidine 250 mg administered twice daily is a second alternative. The 2004 recommendations for dipyridamole include use before discharge or early after discharge in patients with STEMI who are not undergoing cardiac catheterization to look for inducible ischemia in patients who are deemed unable to exercise. Patients with STEMI who have an ischemic stroke but are not treated with PCI and do not have a cardiac source of embolism or surgically important carotid stenosis may be treated with aspirin/extended-release dipyridamole 25/200 mg plus aspirin 81 mg/d. The use of dipyridamole does not appear to be suggested outside of these indications, and it is not addressed within the 2007 update.
Guideline recommendations for NSTEMI/UA. In the most recent 2007 ACC/AHA guidelines for the management of patients with NSTEMI/UA, aspirin 75 to 162 mg/d is still recommended indefinitely for any patient with UA or NSTEMI who has not undergone stent placement (Table 1).12 For patients who receive a bare metal stent, an initial aspirin dose of 162 to 325 mg/d is recommended for ≥1 month, with a subsequent dose of 75 to 162 mg/d to be continued indefinitely. In patients who undergo placement of a drug-eluting stent, aspirin 162 to 325 mg/d is advised for ≥3 months (sirolimus-eluting stent) or for 6 months (paclitaxel-eluting stent) as initial therapy. The dose may then be decreased and maintained at 75 to 162 mg/d thereafter for the life of the patient.
FDA Approves Combination Therapy for Pulmonary Arterial Hypertension
March 25th 2024J&J’s Opsynvi is single-tablet combination of macitentan, an endothelin receptor antagonist, and tadalafil, a PDE5 inhibitor. It will be priced on parity with Opsumit, which is also a J&J product to treat patients with PAH.
FDA Issues Complete Response Letter for Onpattro in Heart Failure Indication
October 9th 2023Alnylam Pharmaceuticals will no longer pursue this indication of Onpattro and will instead on focus on a label expansion for Amvuttra, which is in phase 3 development to treat patients with cardiomyopathy of ATTR amyloidosis.
