- Safety & Recalls
- Regulatory Updates
- Drug Coverage
- COPD
- Cardiovascular
- Obstetrics-Gynecology & Women's Health
- Ophthalmology
- Clinical Pharmacology
- Pediatrics
- Urology
- Pharmacy
- Idiopathic Pulmonary Fibrosis
- Diabetes and Endocrinology
- Allergy, Immunology, and ENT
- Musculoskeletal/Rheumatology
- Respiratory
- Psychiatry and Behavioral Health
- Dermatology
- Oncology
Prenatal vitamins: A review of the literature on benefits and risks of various nutrient supplements
A look at the evidence in support of supplemental vitamin use during pregnancy.
Most pregnant women take a daily prenatal vitamin, but advising the patient can be difficult because so many different formulations are available.
Prenatal vitamins generally contain a variety of vitamins and minerals and may be similar to multivitamins used outside of pregnancy, with some notable differences. Some of these vitamins have been studied directly (eg, folic acid), and their recommended allowance comes from sound evidence. But for most vitamins, data are limited, and for multivitamins (also referred to as multimicronutrient supplements), study results can be biased by confounding variables. This article reviews the evidence in support of supplemental vitamin use during pregnancy. The discussion focuses primarily on data retrieved from the developed world, because the nutritional challenges of the developing world are distinct.
GENERAL NUTRITION REQUIREMENTS DURING PREGNANCY
Pregnancy represents a time of rapid change in maternal physiology and nutritional requirements. These changes allow the mother to meet the needs of the growing fetus and placenta and occur within weeks of the establishment of pregnancy. Dietary recommendations during pregnancy have focused on maintaining adequate caloric intake while avoiding substances that may harm the growing fetus. A particular focus on micronutrients during pregnancy has led to specific recommendations regarding nutrients such as calcium, folic acid, and iron.
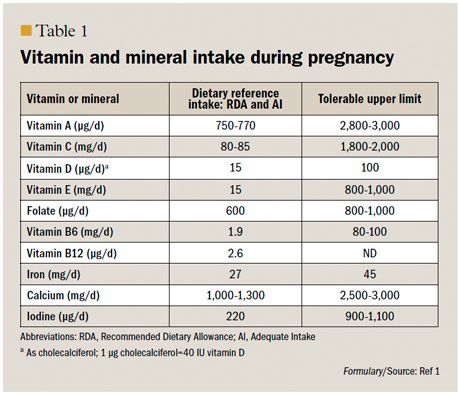
Since 1997, the Institute of Medicine’s (IOM) Food and Nutrition Board has been publishing dietary reference intakes (DRIs) for healthy people throughout the life stages, including pregnancy (Table 1, page 78).1 These tables include recommended daily allowances (RDAs) for which the committee believed there was enough information to make such a recommendation. In some circumstances, the Food and Nutrition Board did not have enough data to determine RDAs, and therefore, the available information combined with expert opinion, was used to develop adequate intake values. The American College of Obstetricians and Gynecologists (ACOG) has incorporated these tables into its recommendations for nutrition during the perinatal period.2
SPECIFIC VITAMIN REQUIREMENTS
Folic acid
The naturally occurring, water-soluble vitamin folate is integral to the synthesis and maintenance of DNA, metabolism of amino acids, production of red blood cells, and growth of the fetus and placenta.3 Folate is found in a number of foods and is most readily acquired from green leafy vegetables (ie, foliage), which gave it its name.3 Folic acid is a synthetic compound that is used as a folate supplement in various dosages.
Strong evidence links folic acid supplementation to a decreased incidence of neural tube defects, and all authorities recommend an increased allowance for women of childbearing age.
A Cochrane review summarized data from randomized and quasi-randomized trials of daily periconceptional folic acid supplementation at doses of 360 μg to 4000 μg (0.36 mg-4 mg).4 Supplementation was effective at reducing both primary (first occurrence) and recurrent neural tube lesions in this meta-analysis, which included 5 trials and 6,105 women. No negative effects were noted.
Folic acid supplementation may also be beneficial for preventing other congenital anomalies such as cleft lip/palate and heart defects; however, evidence from randomized trials is lacking.
Ideally, an adequate amount of folic acid is obtained from the diet. Fortification of grain products with folic acid began in 1998 in the United States, and the incidence of neural tube defects has decreased since then.5 Still, the majority of US women do not consume the recommended allowance of folic acid, so supplementation is recommended.6
Because the neural tube closes by 7 to 8 weeks’ gestation, before most women know they are pregnant, and given that approximately 50% of pregnancies are unplanned, it is prudent for all women of childbearing age to take supplemental folic acid. Unfortunately, a significant number of women do not take supplements, especially women with less education and of minority backgrounds. (This “folate gap” is discussed further in the December 2010 issue of Contemporary OB/GYN.7)
The RDA for folate during pregnancy is 600 μg/day.1 The US Preventive Services Task Force recommends that all women planning or capable of pregnancy take a daily supplement containing 400 µg to 800 µg (0.4 mg-0.8 mg) of folic acid.8 ACOG recommends 400 µg/day for low-risk women and 4000 µg (4 mg) daily for high-risk women, such as women with a history of an affected pregnancy and those taking antiepileptic medications.9
Omega-3 fatty acids
Docosahexaenoic acid (DHA) and eicosapentaenoic acid (EPA) are omega-3 (also known as n-3) long-chain polyunsaturated fatty acids (LCPUFAs), which are found in many prenatal vitamins and supplements and are promoted as beneficial for fetal neurodevelopment.3
Omega-3 fatty acids are associated with vasodilation, inhibition of thrombosis, and reduction in inflammation. In general, these nutrients are found in plants such as walnuts and in oils made from soy, canola, and flaxseed. Both DHA and EPA are found in fatty fish (salmon, tuna, trout, sardines, and mackerel) and in smaller amounts in lean fish and shrimp.
Also, DHA is the most abundant fatty acid in the brain, where it accumulates rapidly in the third trimester and neonatal period.10 Thus, maternal nutrition may affect the amount of DHA deposited in the growing brain.
Multiple observational studies in humans and animal studies have linked fish, fish oil, and/or LCPUFA supplementation with improved cognitive function in offspring.10 Unfortunately, observational studies are subject to various biases. For example, higher fish consumption in the United States is associated with higher socioeconomic status and other healthy habits.
The gold standard for the evaluation of causality is the randomized trial, and 4 major randomized trials have evaluated the use of fish oil supplements during pregnancy and lactation.11-14 These trials, none of which was performed in the United States, failed to show a consistent benefit of supplementation.
The largest study, dubbed the DHA to Optimize Mother Infant Outcome (DOMInO) trial, took place in Australia and recruited more than 2.000 women before 21 weeks’ gestation.12 The participants received fish oil capsules with 800 mg of DHA per day or vegetable oil capsules without DHA until birth. No differences were found in the infants’ cognitive or language development at 18 months or in visual acuity at 4 months.15 There were also no differences in maternal
postpartum depression.12
This study did find a lower rate of preterm birth (<34 weeks) and a smaller incidence of low birth weight (LBW; <2.500 g), although the numbers in these groups were small. An analysis of infants in this trial who were deemed to be at high hereditary risk of developing allergic disease (n=706) found no difference in immunoglobulin E-associated allergies (eczema or food allergy), but lower rates of atopic eczema and egg sensitization were noted.16
A similar trial was conducted in Norway comparing cod liver oil containing more than 1000 mg of DHA and 800 mg of EPA to corn oil that contained primarily linoleic and alpha-linoleic acid.11,17 At 9 months, there were no differences in cognitive development or growth.11
At 4 years, children in the cod liver oil group had higher scores on development and cognitive tests.17 However, at 7 years, there were no differences in IQ or body mass index.18 A major limitation of this study was the high dropout rate.
The 2 other smaller trials found modest benefits in eye-and-hand coordination,13 and although maternal DHA levels were positively associated with cognitive function, supplementation was not.14,19
The associations between omega-3 fatty acids and a number of other maternal, neonatal, and pediatric outcomes have been explored. A systematic review found no consistent effect of supplementation during pregnancy or lactation on infant and child body composition.20 A Cochrane review found no benefit in the prevention of preeclampsia or intrauterine growth restriction.21 However, this review, as in other studies,12,22 did find a significantly longer gestation in women receiving supplements, along with a lower risk of preterm delivery (<34 weeks), even after adjusting for possible confounding factors.
The association between omega-3 fatty acids and maternal depression is unclear, with observational studies suggesting a protective effect but with randomized trials and pooled observational studies showing no benefit.12,23,24
Similar to the lack of effect of DHA during pregnancy, fish oil and DHA supplementation during lactation also achieved no significant differences in primary neurodevelopmental outcomes, according to a Cochrane review of 6 randomized trials.25 In 2 trials, head circumference was larger among infants exposed to supplements. In some of these studies, supplementation was initiated during pregnancy, and a subgroup analysis is planned.
Because the benefits of DHA during pregnancy have not been proven, DRIs have not been established in the United States. However, many expert groups suggest 200 mg of DHA intake daily.26 This may be achieved in a number of ways but is ideally derived by eating 1 to 2 portions of fish per week. Approximately 1 g of EPA/DHA can be obtained from 100 g (3.5 oz) of oily fish. Because of the risk of mercury exposure, pregnant women should avoid shark, king mackerel, swordfish, and tilefish.27
In short, pregnant women should consume at least 2 meals of fish per week to achieve a DHA goal of 200 mg/day. Supplementation with DHA and EPA cannot be definitively recommended during pregnancy because of lack of data. However, evidence of risk is low.
Vitamin D
Vitamin D is an essential fat-soluble nutrient and has been the focus of considerable media attention over the past few years. The term vitamin D, or calciferol, generally refers to either vitamin D2 or vitamin D3.
Technically, vitamin D is not a vitamin at all, because it is synthesized in the skin primarily through ultraviolet irradiation of 7-dehydrocholesterol.28 In the Western world, where the amount of sun exposure is limited because of skin covering, sunscreen use, and prolonged time indoors, most vitamin D is obtained through ingestion of fortified milk and cheese products, and through vitamin supplementation.29 Vitamin D can be obtained from dietary sources such as fatty fish and egg yolks (Table 4). Vitamin D is measured in the serum as 25-hydroxyvitamin D (25OHD or calcidiol) in ng/mL or nmol/L.
Proposed perinatal effects of vitamin D deficiency include increased risk of preeclampsia, cesarean delivery (CD), intrahepatic cholestasis of pregnancy, preterm birth, small for gestational age (SGA), gestational diabetes, and others.30,31 The data regarding these associations are limited and conflicting, and the biologic mechanisms through which vitamin D deficiency mediates these effects are theorized. Vitamin D may mediate preeclampsia risk through its immunomodulatory effects at the time of placental implantation,32 through alteration of inflammatory cytokines in the placenta,33 or through effects on calcium homeostasis and overall cardiovascular health.34 Vitamin D may mediate CD risk through its effects on both smooth muscle and skeletal muscle.35
Trials of vitamin D supplementation in pregnancy have demonstrated a significantly increased vitamin D level in the mother; however, improved clinical outcomes have not been observed.36
There is not enough data to support the use of vitamin D supplements to decrease the risk of adverse perinatal outcomes.31 The current recommendations regarding vitamin D intake are geared toward the maintenance of bone health in the mother, fetus, and newborn. The normal range of vitamin D levels is debated. Currently, the IOM suggests that people with 25OHD levels less than 12 ng/mL (30 nmol/L) are at risk of deficiency, whereas those with levels greater than 20 ng/mL (50 nmol/L) are sufficient, although some experts suggest higher levels.29 The use of higher cutoffs has led to an overestimation of vitamin D insufficiency in the United States.
The DRI for vitamin D during pregnancy is 600 IU (15 μg) per day (Table 1, page 78). Exposure of arms and legs for 5 to 30 minutes (depending on time of day, season, latitude, and skin pigmentation) between the hours of 10 am and
3 pm twice a week is often adequate; however, care must be taken to balance the risks of cancer due to sun exposure.29 The upper limit is 4,000 IU (100 μg) per day; toxic levels do not occur from prolonged sun exposure. Routine assessment
of vitamin D levels (25OHD) in pregnancy is not recommended.
Iron
Adequate elemental iron intake is important during pregnancy to maintain the increase in red blood cell mass and meet the oxygen requirements of the uteroplacental circulation.37 Maternal anemia has been associated with LBW, preterm delivery, perinatal mortality, and maternal depression. The US Preventive Services Task Force38 and ACOG recommend that all pregnant women be routinely screened for anemia. Anemia during pregnancy is defined as hemoglobin and
hematocrit of less than 11 g/dL and 33%, respectively, in the first or third trimester, and as 10.5 g/dL and 32%, respectively, in the second trimester.
The DRI for iron is 27 mg/day and can be obtained from most prenatal vitamins (Table 1, page 78).1 This represents an increase of only 10 to 15 mg over the recommendation for nonpregnant adult women. Although the treatment of iron deficiency anemia is not debated, the benefits of iron supplementation in healthy women have been questioned, and some studies of iron supplementation have demonstrated harm.39
A Cochrane review of 49 trials involving more than 20,000 pregnant women showed that although prophylactic iron supplementation was associated with higher hemoglobin levels in the peripartum period, there were no differences in maternal or neonatal clinical outcomes.40 In addition, significant adverse effects were noted, especially hemoconcentration and constipation. This review was limited by significant heterogeneity in the study populations because the authors included trials from a variety of countries in different phases of economic development. A recent study found an association between iron supplementation and gestational diabetes.41
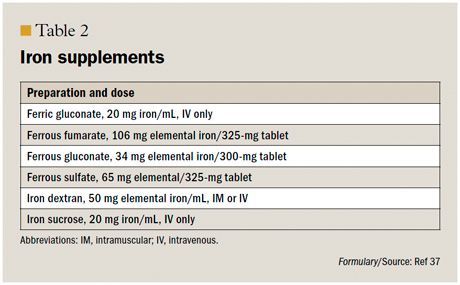
As mentioned earlier, the RDA for iron during pregnancy is 27 mg/day.1 In the setting of iron deficiency anemia, supplementation of 60 mg/day to 80 mg/day of elemental iron is suggested (Table 2).37 The lowest necessary dose should be used. Vitamin C increases the absorption of iron.
VITAMIN AND MINERAL EXCESS
Although the benefits of certain vitamins during pregnancy can be debated, some vitamins are associated with convincing evidence of harm when taken in excess doses.3 For example, more than 10,000 IU/day of vitamin A is associated with fetal malformations, yet adequate vitamin A is important to support healthy vision and immune function. Most US women do achieve adequate vitamin A intake from their diets, and prenatal vitamins contain 4000 to 5000 IU; therefore, additional supplementation is not advised.
Several trials have explored the efficacy of increased amounts of vitamin C (1,000 mg) and vitamin E (400 IU) for the prevention of preeclampsia.42 When the data were pooled, these studies demonstrated that not only were the vitamins not protective but also that women who received the supplements actually had increased risks of gestational hypertension and premature rupture of membranes.
Both an excess of iodine and a deficiency of iodine may lead to fetal goiter and developmental delay. Therefore, women are advised to stay within the RDA. Many prenatal vitamins do not contain iodine, and therefore dietary intake is important. Supplementation is frequently achieved through iodized salt; however, many gourmet and kosher salt preparations do not contain iodine. The RDA for pregnancy is 220 μg.
The tolerable upper limits of vitamin and mineral supplementation during pregnancy are listed in Table 1 (page 78).1
ROLE OF A HEALTHY DIET
Underweight has long been known as a risk factor for adverse pregnancy outcomes. Recently, overweight and obesity have become much more prevalent and are now recognized as significant risk factors as well. A healthy diet is necessary to modify these risks, and observational studies have linked specific dietary patterns, such as a Mediterranean-type diet, with improved pregnancy outcomes.43,44
Women whose diets are high in processed meats and high-fat dairy foods (compared with those who eat more fruits, vegetables, and fish) have a higher risk of having infants who are small for gestational age (SGA).45 However, given that observational studies are at risk for significant bias because of confounding factors, attempts have been made to perform randomized trials.
For example, the Cardiovascular Risk Reduction Diet in Pregnancy (CARRDIP) trial studied a low-cholesterol, low-saturated-fat, antiatherogenic diet compared with a usual diet among nonsmoking, healthy European women.46 This study found a reduction in preterm birth (<37 weeks) of 7.4% in women on the Mediterranean-type diet compared with 0.7% in the women on their usual diet and improved uteroplacental circulation as measured by Doppler velocimetry.47 Randomized trials of specific diets are exceedingly difficult to undertake, however, and so researchers have focused instead on the use of multivitamins and micronutrient supplements.
MULTIVITAMIN USE
In the developing world, where the risk of malnutrition is high, multiple trials have been performed to assess the effects of prenatal multimicronutrient supplementation. Two recent systematic reviews and meta-analyses have demonstrated a decreased risk of SGA48 and LBW49 infants in women who received multimicronutrient supplements versus those who received standard iron-folic acid supplementation. Reductions in miscarriage, preterm birth, and stillbirth have not been consistently noted, and vitamins have been associated with an increased incidence of multiple gestation.50
Most of the literature on multivitamin use in the developed world consists of observational studies, and many have found improved outcomes. Multivitamin use has been associated with a decreased risk of SGA infants,51 specifically in nonoverweight women,52 black women,53 and low-income urban women.54 The literature regarding preterm birth is conflicting, with some studies showing a decreased risk,52,54,55 some showing no effect,53 and 1 study showing an increased risk of preterm birth with multivitamin use.56 A decreased risk of placental abruption has also been noted,57 as well as a decreased risk of preeclampsia.58
These observational studies are inherently limited for a number of reasons. First, many were retrospective, relying on questionnaires administered after pregnancy, and thus were subject to recall bias. Second, in almost all studies, multivitamin use was significantly associated with a number of factors that are also known to improve pregnancy outcomes: higher socioeconomic status, not smoking, more education, married status, and nonminority race. Therefore, although these observational studies attempted to control for these factors, residual confounding is a major issue, and causality cannot be inferred. It just may be that healthier women take vitamins, and healthier women have better pregnancy outcomes.
SUMMARY
Vitamin and mineral supplements cannot replace a healthy diet, and there are not enough high-quality data to recommend multivitamin supplements for all American women. The differences between prenatal vitamins are slight, and no particular vitamin can be recommended over another. Pregnant women should stay below the upper limits of supplementation per IOM guidelines (Table 1, page 78).1 Folic acid is the 1 vitamin for which definitive evidence supports preventive supplementation in the periconceptional period.
Multivitamin supplements are recommended for pregnant women who cannot meet the RDAs through food intake; ideally, these women should be identified before conception and prescribed supplements at that time. At-risk populations include adolescents, women carrying multiple gestations, those with a substance abuse history (including tobacco), those with eating disorders, those taking certain medications that can alter absorption, and strict vegetarians or vegans.59
References
1. Food and Nutrition Board, Institute of Medicine, National Academy of Sciences. Recommended Dietary Allowance and Adequate Intake Values, Vitamins and Elements. Institute of Medicine Web site. www.iom.edu/Activities/Nutrition/SummaryDRIs/DRI-Tables.aspx. Updated September 12, 2011. Accessed July 26, 2012.
2. American College of Obstetricians and Gynecologists; American Academy of Pediatrics. Guidelines for Perinatal Care. 6th ed. Washington, DC: American College of Obstetricians and Gynecologists; 2007. Elk Grove Village, IL: American Academy of Pediatrics; 2007.
3. Insel P, Ross D, McMahon K, Bernstein M. Nutrition. 4th ed. Sudbury, MA: Jones and Bartlett; 2011.
4. De-Regil LM, Fernández-Gaxiola AC, Dowswell T, Peña-Rosas JP. Effects and safety of periconceptional folate supplementation for preventing birth defects. Cochrane Database Syst Rev. 2010;(10):CD007950.
5. Honein MA, Paulozzi LJ, Mathews TJ, Erickson JD, Wong LY. Impact of folic acid fortification of the US food supply on the occurrence of neural tube defects. JAMA. 2001;285(23):2981-2986.
6. Centers for Disease Control and Prevention (CDC). Use of dietary supplements containing folic acid among women of childbearing age-United States, 2005. MMWR Morb Mortal Wkly Rep. 2005;54(38):955-958.
7. Simpson JL, Shulman LP, Brown H, Holzgreve W. Closing the folate gap in reproductive-age women. Contemporary OB/GYN. 2010;55(12):34-40.
8. US Preventive Services Task Force. Folic acid for the prevention of neural tube defects: US Preventive Services Task Force recommendation statement. Ann Intern Med. 2009;150(9):626-631.
9. Cheschier N; ACOG Committee on Practice Bulletins-Obstetrics. ACOG practice bulletin. Neural tube defects. Number 44, July 2003. (Replaces committee opinion number 252, March 2001.) Int J Gynaecol Obstet. 2003;83(1):123-133. Reaffirmed: Obstet Gynecol. 2011;117(6):1471.
10. Carlson SE. Docosahexaenoic acid supplementation in pregnancy and lactation. Am J Clin Nutr. 2009;89(2):678S-684S.
11. Helland IB, Saugstad OD, Smith L, et al. Similar effects on infants of n-3 and n-6 fatty acids supplementation to pregnant and lactating women. Pediatrics. 2001;108(5):E82.
12. Makrides M, Gibson RA, McPhee AJ, Yelland L, Quinlivan J,Ryan P; DOMInO Investigative Team. Effect of DHA supplementation during pregnancy on maternal depression and neurodevelopment of young children: a randomized controlled trial. JAMA. 2010;304(15):1675-1683.
13. Dunstan JA, Simmer K, Dixon G, Prescott SL. Cognitive assessment of children at age 2(1/2) years after maternal fish oil supplementation in pregnancy: a randomised controlled trial. Arch Dis Child Fetal Neonatal Ed. 2008;93(1):F45-F50.
14. Escolano-Margarit MV, Ramos R, Beyer J, et al. Prenatal DHA status and neurological outcome in children at age 5.5 years are positively associated. J Nutr. 2011;141(6):1216-1223.
15. Smithers LG, Gibson RA, Makrides M. Maternal supplementation with docosahexaenoic acid during pregnancy does not affect early visual development in the infant: a randomized controlled trial. Am J Clin Nutr. 2011;93(6):1293-1299.
16. Palmer DJ, Sullivan T, Gold MS, et al. Effect of n-3 long chain polyunsaturated fatty acid supplementation in pregnancy on infants’ allergies in first year of life: randomised controlled trial. BMJ. 2012;344:e184.
17. Helland IB, Smith L, Saarem K, Saugstad OD, Drevon CA. Maternal supplementation with very-long-chain n-3 fatty acids during pregnancy and lactation augments children’s IQ at 4 years of age. Pediatrics. 2003;111(1):e39-e44.
18. Helland IB, Smith L, Blomén B, Saarem K, Saugstad OD, Drevon CA. Effect of supplementing pregnant and lactating mothers with n-3 very-long-chain fatty acids on children’s IQ and body mass index at 7 years of age. Pediatrics. 2008;122(2):e472-e479.
19. Campoy C, Escolano-Margarit MV, Ramos R, et al. Effects of prenatal fish-oil and 5-methyltetrahydrofolate supplementation on cognitive development of children at 6.5 y of age. Am J Clin Nutr. 2011;94(6 suppl):1880S-1888S.
20. Muhlhausler BS, Gibson RA, Makrides M. Effect of long-chain polyunsaturated fatty acid supplementation during pregnancy or lactation on infant and child body composition: a systematic review. Am J Clin Nutr. 2010;92(4):857-863.
21. Makrides M, Duley L, Olsen SF. Marine oil, and other prostaglandin precursor, supplementation for pregnancy uncomplicated by pre-eclampsia or intrauterine growth restriction. Cochrane Database Syst Rev. 2006;(3):CD003402.
22. Smuts CM, Huang M, Mundy D, Plasse T, Major S, Carlson SE. A randomized trial of docosahexaenoic acid supplementation during the third trimester of pregnancy. Obstet Gynecol. 2003;101(3):469-479.
23. Freeman MP, Davis M, Sinha P, Wisner KL, Hibbeln JR, Gelenberg AJ. Omega-3 fatty acids and supportive psychotherapy for perinatal depression: a randomized placebo-controlled study. J Affect Disord. 2008;110(1-2):142-148.
24. Ramakrishnan U. Fatty acid status and maternal mental health. Matern Child Nutr. 2011;7(suppl 2):99-111.
25. Delgado-Noguera MF, Calvache JA, Bonfill Cosp X. Supplementation with long chain polyunsaturated fatty acids (LCPUFA) to breastfeeding mothers for improving child growth and development. Cochrane Database Syst Rev. 2010;(12):CD007901.
26. Kris-Etherton PM, Grieger JA, Etherton TD. Dietary reference intakes for DHA and EPA. Prostaglandins Leukot Essent Fatty Acids. 2009;81(2-3):99-104.
27. US Food and Drug Administration (FDA), US Environmental Protection Agency (EPA). What you need to know about mercury in fish and shellfish. 2004 EPA and FDA advice for: women who might become pregnant, women who are pregnant, nursing mothers, young children. FDA Web site. www.fda.gov/Food/FoodSafety/Product-SpecificInformation/Seafood/FoodbornePathogensContaminants/Methylmercury/ucm115662.htm. Published March 2004. Updated November 21, 2011. Accessed July 31, 2012.
28. Dictionary of Food Science and Nutrition. London, England: A & C Black Publishers; 2006.
29. Holick MF. Vitamin D deficiency. N Engl J Med. 2007;(357):266-281.
30. Bodnar LM, Simhan HN. Vitamin D may be a link to black-white disparities in adverse birth outcomes. Obstet Gynecol Surv. 2010;(65):273-284.
31. Insitute of Medicine. Dietary Reference Intakes for Calcium and Vitamin D. Washington, DC: National Academies Press; 2011.
32. Hypponen E. Vitamin D for the prevention of preeclampsia? A hypothesis. Nutr Rev. 2005;(63):225-232.
33. Diaz L, Noyola-Martinez N, Barrera D, et al. Calcitriol inhibits TNF-alpha-induced inflammatory cytokines in human trophoblasts. J Reprod Immunol. 2009;(81):17-24.
34. Taylor SN, Wagner CL, Hollis BW. Vitamin D deficiency in pregnancy and lactation and health consequences. In: Holick MF, ed. Vitamin D Physiology, Molecular Biology, and Clinical Applications. 2nd ed. New York: Humana Press; 2010.
35. Lapillonne A. Vitamin D deficiency during pregnancy may impair maternal and fetal outcomes. Med Hypotheses. 2010;(74):71-75.
36. De-Regil LM, Palacios C, Ansary A, Kulier R, Pena-Rosas JP. Vitamin D supplementation for women during pregnancy. Cochrane Database Syst Rev 2012;2:CD008873.
37. American College of Obstetricians and Gynecologists. ACOG Practice Bulletin No. 95: anemia in pregnancy. Obstet Gynecol. 2008;112(1):201-207.
38. US Preventive Services Task Force (USPSTF). Screening for Iron Deficiency Anemia. www.uspreventiveservicestaskforce.org/uspstf/uspsiron.htm. Accessed July 31, 2012.
39. Berti C, Biesalski HK, Gärtner R, et al. Micronutrients in pregnancy: current knowledge and unresolved questions. Clin Nutr. 2011;30(6):689-701.
40. Peña-Rosas JP, Viteri FE. Effects and safety of preventive oral iron or iron+folic acid supplementation for women during pregnancy. Cochrane Database Syst Rev. 2009;(4):CD004736.
41. Bo S, Menato G, Villois P, et al. Iron supplementation and gestational diabetes in midpregnancy. Am J Obstet Gynecol. 2009;201(2):158.e1-158.e6.
42. Conde-Agudelo A, Romero R, Kusanovic JP, Hassan SS. Supplementation with vitamins C and E during pregnancy for the prevention of preeclampsia and other adverse maternal and perinatal outcomes: a systematic review and metaanalysis. Am J Obstet Gynecol. 2011;204(6):503.e1-503.e12.
43. Chatzi L, Mendez M, Garcia R, et al; INMA and RHEA study groups. Mediterranean diet adherence during pregnancy and fetal growth: INMA (Spain) and RHEA (Greece) mother-child cohort studies. Br J Nutr. 2012;107(1):135-145.
44. Mikkelsen TB, Osterdal ML, Knudsen VK, et al. Association between a Mediterranean-type diet and risk of preterm birth among Danish women: a prospective cohort study. Acta Obstet Gynecol Scand. 2008;87(3):325-330.
45. Knudsen VK, Orozova-Bekkevold IM, Mikkelsen TB, Wolff S, Olsen SF. Major dietary patterns in pregnancy and fetal growth. Eur J Clin Nutr. 2008;62(4):463-470.
46. Khoury J, Henriksen T, Christophersen B, Tonstad S. Effect of a cholesterol-lowering diet on maternal, cord, and neonatal lipids, and pregnancy outcome: a randomized clinical trial. Am J Obstet Gynecol. 2005;193(4):1292-1301.
47. Khoury J, Haugen G, Tonstad S, Frøslie KF, Henriksen T. Effect of a cholesterol-lowering diet during pregnancy on maternal and fetal Doppler velocimetry: the CARRDIP study. Am J Obstet Gynecol. 2007;196(6):549.e1-549.e7.
48. Haider BA, Yakoob MY, Bhutta ZA. Effect of multiple micronutrient supplementation during pregnancy on maternal and birth outcomes. BMC Public Health. 2011;11(suppl 3):S19.
49. Shah PS, Ohlsson A; Knowledge Synthesis Group on Determinants of Low Birth Weight and Preterm Births. Effects of prenatal multimicronutrient supplementation on pregnancy outcomes: a meta-analysis. CMAJ. 2009;180(12):
E99-E108.
50. Rumbold A, Middleton P, Pan N, Crowther CA. Vitamin supplementation for preventing miscarriage. Cochrane Database Syst Rev. 2011;(1):
CD004073.
51. Catov JM, Bodnar LM, Ness RB, Markovic N, Roberts JM. Association of periconceptional multivitamin use and risk of preterm or small-for-gestational-age births. Am J Epidemiol. 2007;166(3):296-303.
52. Catov JM, Bodnar LM, Olsen J, Olsen S, Nohr EA. Periconceptional multivitamin use and risk of preterm or small-for-gestational-age births in the Danish National Birth Cohort. Am J Clin Nutr. 2011;94(3):906-912.
53. Burris HH, Mitchell AA, Werler MM. Periconceptional multivitamin use and infant birth weight disparities. Ann Epidemiol. 2010;20(3):233-240.
54. Scholl TO, Hediger ML, Bendich A, Schall JI, Smith WK, Krueger PM. Use of multivitamin/mineral prenatal supplements: influence on the outcome of pregnancy. Am J Epidemiol. 1997;146(2):134-141.
55. Vahratian A, Siega-Riz AM, Savitz DA, Thorp JM Jr. Multivitamin use and the risk of preterm birth. Am J Epidemiol. 2004;160(9):886-892.
56. Alwan NA, Greenwood DC, Simpson NA, McArdle HJ, Cade JE. The relationship between dietary supplement use in late pregnancy and birth outcomes: a cohort study in British women. BJOG. 2010;117(7):821-829.
57. Nilsen RM, Vollset SE, Rasmussen SA, Ueland PM, Daltveit AK. Folic acid and multivitamin supplement use and risk of placental abruption: a population-based registry study. Am J Epidemiol. 2008;167(7):867-874.
58. Bodnar LM, Tang G, Ness RB, Harger G, Roberts JM. Periconceptional multivitamin use reduces the risk of preeclampsia. Am J Epidemiol. 2006;164(5):470-477.
59. Koebnick C, Hoffmann I, Dagnelie PC, et al. Long-term ovo-lacto vegetarian diet impairs vitamin B-12 status in pregnant women. J Nutr. 2004;134(12):3319-3326.
Coalition promotes important acetaminophen dosing reminders
November 18th 2014It may come as a surprise that each year Americans catch approximately 1 billion colds, and the Centers for Disease Control and Prevention estimates that as many as 20% get the flu. This cold and flu season, 7 in 10 patients will reach for an over-the-counter (OTC) medicine to treat their coughs, stuffy noses, and sniffles. It’s an important time of the year to remind patients to double check their medicine labels so they don’t double up on medicines containing acetaminophen.
Support consumer access to specialty medications through value-based insurance design
June 30th 2014The driving force behind consumer cost-sharing provisions for specialty medications is the acquisition cost and not clinical value. This appears to be true for almost all public and private health plans, says a new report from researchers at the University of Michigan Center for Value-Based Insurance Design (V-BID Center) and the National Pharmaceutical Council (NPC).
Management of antipsychotic medication polypharmacy
June 13th 2013Within our healthcare-driven society, the increase in the identification and diagnosis of mental illnesses has led to a proportional increase in the prescribing of psychotropic medications. The prevalence of mental illnesses and subsequent treatment approaches may employ monotherapy as first-line treatment, but in many cases the use of combination of therapy can occur, leading to polypharmacy.1 Polypharmacy can be defined in several ways but it generally recognized as the use of multiple medications by one patient and the most common definition is the concurrent use of five more medications. The presence of polyharmacy has the potential to contribute to non-compliance, drug-drug interactions, medication errors, adverse events, or poor quality of life.
Medical innovation improves outcomes
June 12th 2013I have been diagnosed with stage 4 cancer of the pancreas, a disease that’s long been considered not just incurable, but almost impossible to treat-a recalcitrant disease that some practitioners feel has given oncology a bad name. I was told my life would be measured in weeks.

