- Safety & Recalls
- Regulatory Updates
- Drug Coverage
- COPD
- Cardiovascular
- Obstetrics-Gynecology & Women's Health
- Ophthalmology
- Clinical Pharmacology
- Pediatrics
- Urology
- Pharmacy
- Idiopathic Pulmonary Fibrosis
- Diabetes and Endocrinology
- Allergy, Immunology, and ENT
- Musculoskeletal/Rheumatology
- Respiratory
- Psychiatry and Behavioral Health
- Dermatology
- Oncology
A review of antithrombotic therapies in acute coronary syndrome: Should unfractionated heparin be replaced?
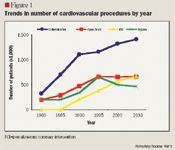
Cardiovascular disease afflicts more than 71.3 million people in the United States and accounts for more deaths annually than any other cause. The estimated direct and indirect costs associated with cardiovascular disease in 2006 ($403 billion) were more than double the costs associated with cancer ($190 billion), which is the second-leading cause of death in the United States.
Abstract
Acute coronary syndrome (ACS) is the cause of more than 13 million hospitalizations and emergency room visits annually in the United States. Treatment options currently include pharmacologic modalities with or without mechanical procedures to target new obstructions and to prevent existing thrombus growth and formation. Although unfractionated heparin (UFH) has historically been the agent of choice to prevent fibrin formation in ACS, complications in monitoring and increased bleeding risk have led to the search for alternative treatment options, including direct thrombin inhibitors (bivalirudin), pentasaccharide (fondaparinux), and low-molecular-weight heparins (enoxaparin). Emerging data suggest that these newer antithrombotic therapies may provide reasonable alternatives to UFH in certain patient populations. More data and clinical experience are needed to determine the optimal agent to replace UFH as antithrombotic therapy. (Formulary. 2007;42:150–164.)

ANTITHROMBOTIC AGENTS
Thrombosis occurs via the activation and interplay of clotting factors and tissue elements including platelets, which are major targets of antithrombotic drug therapies.4 Several classes of medications are available to prevent thrombus propagation and fibrin formation. These classes include unfractionated heparin (UFH), direct thrombin inhibitors (bivalirudin), pentasaccharide (fondaparinux), and low-molecular-weight heparins (LMWH) (enoxaparin).
Bivalirudin. Bivalirudin is a direct thrombin inhibitor that acts through inhibition of both free and clot-bound thrombin.9 Thrombin has a significant effect on tissue injury, coagulation, and platelet response, making it a logical target for selective therapy. Bivalirudin does not cause immunogenicity, unlike other hirudin analogs, because bivalirudin is a synthetic compound.9 Bivalirudin is currently indicated for use as an anticoagulant in patients with UA undergoing percutaneous transluminal coronary angioplasty, in patients undergoing PCI with provisional use of glycoprotein (GP) IIb/IIIa inhibitors, and in patients with or at risk for HIT or HIT thrombosis syndrome who are undergoing PCI.
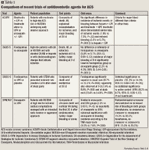
The efficacy of bivalirudin in ACS was studied in the Randomized Evaluation in PCI Linking Angiomax to Reduced Clinical Events (REPLACE-2) trial.10,11 The objective of this trial was to determine the efficacy of bivalirudin plus provisional GP IIb/IIIa inhibitor use during PCI versus UFH plus planned GP IIb/IIIa inhibitor use with regard to protection from periprocedural ischemic and hemorrhagic complications. A secondary end point was the incidence of death, MI, or urgent revascularization at 30 days. Investigators also performed a health economic analysis of treatment with bivalirudin plus provisional GP IIb/IIIa inhibitor use versus heparin plus planned GP IIb/IIIa inhibitor use.
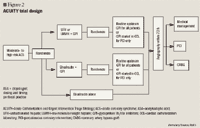
Two independent primary hypotheses were examined in the ACUITY trial: 1) bivalirudin plus routine GP IIb/IIIa inhibitor use would provide a noninferior or superior net clinical outcome (primary end point defined as the composite incidence of death, MI, unplanned revascularization for ischemia, and major bleeding) compared with heparin (UFH or enoxaparin) plus routine GP IIb/IIIa inhibitor use; and 2) bivalirudin alone (with only provisional use of GP IIb/IIIa inhibitors) would demonstrate a noninferior net clinical outcome and a superior reduction in major bleeding events versus heparin (UFH or enoxaparin) plus routine GP IIb/IIIa inhibitor use.
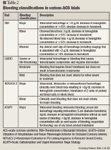
Major bleeding was defined in this study as non-CABG-related bleeding, intracranial bleeding, intraocular bleeding, retroperitoneal bleeding, access site bleed requiring intervention/surgery, hematoma ≥5 cm in diameter, hemoglobin decrease ≥3 g/dL with an overt source of bleeding or hemoglobin decrease ≥4 g/dL without an overt source of bleeding, blood product transfusion, or reoperation for bleeding5 (Table 2).
Patients with ACS undergoing an early invasive strategy who had experienced chest pain consistent with angina ≥10 minutes in duration within 24 hours of randomization and who had ≥1 of the following characteristics were included in the study: new or presumably new ST-segment depression or transient elevation in ≥2 contiguous leads; elevated troponin I, troponin T, or creatinine kinase MB within 24 hours of randomization; coronary artery disease documented by prior angioplasty; history of CABG; or history of MI. Other patients, if not meeting the previous criteria, were eligible if they were aged >65 years; were currently smokers; were treated with acetylsalicylic acid (ASA) within the previous 7 days; experienced ≥2 episodes of angina within the previous 24 hours; and had hypertension, hypercholesterolemia, diabetes, and a family history of heart disease.
Patients were excluded from the study if they had acute ST-segment elevation, cardiogenic shock, bleeding diathesis or a major bleed within the preceding 2 weeks, low platelet count (<100,000/mm3 ), history of HIT, creatinine clearance <30 mL/min, or any allergy to drugs or contrast media, or if they had been treated with abciximab or >2 prior LMWH doses. Patients were also excluded if researchers anticipated an inability to perform an angiography within 72 hours of randomization.
When UFH/enoxaparin plus GP IIb/IIIa inhibitors and bivalirudin plus GP IIb/IIIa inhibitors were compared, the regimen of bivalirudin plus GP IIb/IIIa inhibitors was demonstrated to be noninferior to UFH/enoxaparin plus GP IIb/IIIa inhibitors on the primary end point (11.8% vs 11.7%; 95% CI, 0.90–1.12; P<.001), the ischemic composite (7.7% vs 7.3%; 95% CI, 0.92–1.23; P=.015), and major bleeding (5.3% vs 5.7%; 95% CI, 0.78–1.10; P<.001), but the regimen did not demonstrate superiority to UFH/enoxaparin plus GP IIb/IIIa inhibitors (net clinical outcome, P=.93; ischemic composite, P=.39; major bleeding, P=.38); therefore, bivalirudin plus GP IIb/IIIa inhibitors was not demonstrated to be a superior replacement for heparin plus GP IIb/IIIa inhibitors in regard to ischemic events and incidence of bleeding.
Payers Recognize the Benefits, but Still See Weight Loss Drugs through a Cost Lens
April 12th 2024Jeffrey Casberg, M.S., R.Ph., a senior vice president of clinical pharmacy at IPD Analytics LLC, a drug intelligence firm that advises payers and pharmaceutical companies, talks about how payers are thinking about weight-loss drugs.
Drugs to Watch: Mental Health Conditions
April 11th 2024The FDA is reviewing two novel therapies: a psychedelic-assisted therapy for PTSD with a target action date of Aug. 11, 2024, and therapy for schizophrenia that does not directly block dopamine receptors with an action date of Sept. 26, 2024.
