- Safety & Recalls
- Regulatory Updates
- Drug Coverage
- COPD
- Cardiovascular
- Obstetrics-Gynecology & Women's Health
- Ophthalmology
- Clinical Pharmacology
- Pediatrics
- Urology
- Pharmacy
- Idiopathic Pulmonary Fibrosis
- Diabetes and Endocrinology
- Allergy, Immunology, and ENT
- Musculoskeletal/Rheumatology
- Respiratory
- Psychiatry and Behavioral Health
- Dermatology
- Oncology
A review of common sexually transmitted diseases
Although there have been significant advances in prevention, diagnosis, treatment, and education, the occurrence of sexually transmitted diseases (STDs) continues to increase. This article focuses on the diagnosis and treatment of the most common STDs.

Key Points
Abstract
In 2004, the Centers for Disease Control and Prevention (CDC) estimated that 19 million new sexually transmitted diseases (STDs) occur each year. Of the new cases, half occur in patients aged 15 to 24 years. Although there have been significant advances in prevention, diagnosis, treatment, and education, the occurrence of STDs continues to increase. The most common STDs include chlamydia, herpes simplex virus types 1 and 2, human papillomavirus (HPV), syphilis, gonorrhea, bacterial vaginosis, trichomoniasis, and pelvic inflammatory disease (PID). Diagnosis and treatment continue to be major public health challenges because of the asymptomatic nature of many of these infections. (Formulary. 2009;44:78–85.)

In 2004, the CDC reported that nearly half of the estimated new STD infections were in patients aged 15 to 24 years.1 Women aged 15 to 19 years have the highest reports of chlamydia and gonorrhea compared with women in other age groups, and women who contract human papillomavirus (HPV) infection typically do so in the early adolescent years.4 In most states, adolescents can consent to diagnosis and treatment of STDs without the knowledge or consent of their parents.4 Healthcare providers should be diligent when assessing for risk in this population to ensure that proper education and treatment are carried out.
MSM continue to be at high risk for acquiring STDs.4 The rate of HIV decreased during the 1980s, but the rates of chlamydia, syphilis, and gonorrhea have continued to increase in this population, likely because of unsafe sexual practices and multiple sex partners among MSM. Healthcare providers should assess for risk of STDs in men; this assessment should include a routine inquiry regarding the sex of the patient's partner. WSW have not demonstrated an increase in STD occurrences, but few data are available for this patient population, and risk can vary depending on sexual practices.
Historically, younger populations have been the target audience for sexual health programs aiming to decrease the risk of STDs; however, older populations are also at risk. A study conducted in the United Kingdom at a genitourinary medicine clinic demonstrated that STD incidence among older adults (patients aged ≥45 y) in 2003 was double the incidence reported in 1996.6 The populations most affected were men and patients aged 55 to 59 years. This study signifies the importance of continued STD surveillance later in life depending on the patient's lifestyle and risk factors.
COMMON STDS
Chlamydia. Chlamydia trachomatis infection is the most frequently reported sexually transmitted infectious disease in the United States and continues to be an important risk factor for pelvic inflammatory disease (PID), ectopic pregnancies, urethritis, cervicitis, chronic pelvic pain, and infertility.2,4 In 2007, there were 1,108,374 cases of chlamydia reported to the CDC. Black patients had an 8 times higher incidence than white patients. In women, the highest age-specific rates were among patients aged 15 to 19 years (3,004.7 cases per 100,000) and those aged 20 to 24 years (2,948.8 cases per 100,000).
Typically, patients are asymptomatic and are unaware of an infection with C trachomatis.7 If symptoms are present in women, they typically include an odorless, mucoid vaginal discharge, usually with no external pruritus. In men, symptoms can include a clear to white urethral discharge usually observed in the morning before the first void of the day. Because other infections can present with the same symptoms, healthcare providers cannot use symptoms alone to diagnose a chlamydial infection.
Screening is recommended in sexually active nonpregnant and pregnant women aged ≤24 years and in nonpregnant and pregnant women aged ≥25 years who are at increased risk for STDs.8 The benefits of screening men versus the potential harms have not been established.
Diagnosis can be performed via laboratory testing or point-of-care testing depending on the setting, sensitivity, specificity, ease of collection, and cost.9 Laboratory testing has a higher sensitivity but is not ideal for settings in which patient follow-up is not consistent; therefore, point-of-care testing is typically used in these cases.
Genital herpes. Herpes simplex virus type 1 (HSV-1) and type 2 (HSV-2) infections are both chronic, lifelong viral infections. Approximately 50 million people in the United States are infected with HSV.4 Historically, HSV-2 has been the causative agent in the majority of genital herpes cases, but HSV-1 has become more prevalent, causing approximately half of first episode outbreaks.
Symptoms of genital herpes do not differ between HSV-1 and HSV-2 infections. Patients will often experience prodromal symptoms of tingling, itching, paresthesias, and pain from an hour to a day before the outbreak.10 Antiviral treatment initiated during this time can decrease outbreak occurrences in some patients. Once lesions are present, antiviral therapy can be initiated for treatment. Without therapy, the lesions typically heal within 5 to 10 days. Shedding of HSV is highest in the 6 to 12 months after the first outbreak, signifying the importance of prompt diagnosis and initiation of therapy. In the first year after the initial outbreak, patients who do not receive antiviral therapy may experience approximately 4 to 10 outbreaks. Patients with HSV-1 infections typically have fewer outbreaks than patients with HSV-2 infections.
Screening for HSV is not routinely performed unless patients are at high risk, have a partner diagnosed with herpes, or have symptoms. Other infections presenting with genital ulcers include syphilis and chancroid.4
Diagnostic testing for HSV is readily available; these tests include viral culture, polymerase chain reaction (PCR), and serology testing.4,10,11 Each test has limitations and advantages. Viral cultures are most useful in symptomatic patients with active lesions but within 2 to 3 days after an outbreak, lose sensitivity and are associated with a high rate of false negatives. Serology testing is important in distinguishing HSV-1 from HSV-2. If the patient has been recently infected, there is a chance of a false negative, so these patients need to be retested at a later date.

Genital warts. Human papillomavirus (HPV), which is responsible for cervical cancer and genital warts, has received widespread attention since the introduction of the HPV quadrivalent vaccine Gardasil in 2006. The relationship between HPV and cervical cancer and genital warts has been recognized for many years.12 Although there are >100 genotypes of HPV, the 4 genotypes targeted by the vaccine cause the majority of cervical cancers and genital warts. HPV types 6 and 11 cause 90% of genital warts, and types 16 and 18 cause 70% of cervical cancers.4,12,13 The American Cancer Society (ACS) estimated that approximately 11,070 new cases of cervical cancer would be diagnosed in 2008.13
Screening for cervical cancer is routinely performed at a woman's annual gynecological examination. The first examination should be conducted within 3 years of the initiation of sexual intercourse or by the age of 21.12
The Advisory Committee on Immunization Practices (ACIP) currently recommends HPV vaccination for girls aged 11 to 12 years.14,15 Catch-up vaccination is also recommended in women aged ≤26 years. The age range approved by FDA is 9 to 26 years. The immunization schedule consists of a series of 3 intramuscular (IM) injections administered at 0, 2, and 6 months.16

Gonorrhea. Neisseria gonorrhoeae infection has been increasing over the past few years, causing a growing public health concern in the United States.4 Untreated gonorrhea can lead to cervicitis, urethritis, proctitis, PID, adverse outcomes in pregnancy, and increased susceptibility to and transmission of HIV.17 Gonorrhea rates are highest in black patients, adolescent patients, young adults, and MSM, including HIV-infected MSM. Each year, an estimated 600,000 new cases of gonorrhea occur.4 Infected men frequently have symptoms, allowing them to seek treatment before complications occur. In women, however, symptoms may not appear until complications such as PID or ectopic pregnancy occur.
Currently, the United States Preventative Services Task Force (USPSTF) recommends screening all pregnant and nonpregnant women aged <25 years if they are at high risk.18 USPSTF does not recommend screening men even if they are at high risk. The morbidity associated with undiagnosed gonorrhea in men is much lower than in women, as men will present with symptoms more often than women, which leads to earlier treatment and cure rates. The task force does not recommend screening women aged >25 years unless they are at high risk for infection. A Gram stain of male urethral sample is specific and sensitive for diagnosis.4 Diagnosis of persistent gonorrhea infection after treatment is confirmed with cultures and antimicrobial susceptibility testing.
Frequently, co-infection with C trachomatis occurs in patients with gonorrhea. If co-infection is suspected, the patient should be treated for both infections.
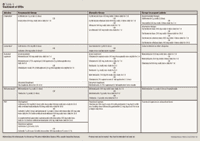
In May 2007, the CDC updated the treatment guidelines for gonorrhea because of outbreaks of resistance to fluoroquinolones reported in Asia, Hawaii, and California.17,19 This resistance limits the options available for treatment (see Table 1 for treatment recommendations). Until recently, cefixime 400-mg tablets were not commercially available, so a suspension formulation was the only option available for oral dosing.20 In April 2008, the oral tablet was once again made available, offering a more convenient dosage form to treat gonorrhea. If a patient is co-infected with chlamydia, a 1-time dose of azithromycin 2 g may be used.17 Azithromycin is not routinely recommended for the treatment of gonorrhea without chlamydia because of concerns about resistance.
Syphilis. Syphilis is caused by the bacterium Treponema pallidum.3 Although syphilis incidence had decreased by 2000, the incidence of the disease has since increased; in 2007, a total of 11,466 cases were reported, an increase of 15% from 2006.3 The population most affected is MSM.
Syphilis is classified into various stages based on the presentation of the patient. Initiation of therapy is critical in primary and secondary syphilis, which are the infectious stages. Primary syphilis occurs approximately 21 days after the exposure and typically presents as a chancre, usually nonpainful, at the site of exposure.21 If left untreated, the chancre will heal on its own. Secondary syphilis presents approximately 4 to 10 weeks after the chancre; in 70% of patients, rash is the presenting symptom of patients seeking treatment. If untreated during these stages, the symptoms will self-resolve but the patient may then experience early latent or late latent syphilis. Differentiation between early latent and late latent syphilis is based on when the patient's symptoms occur; early latent syphilis occurs within 1 year of infection, whereas late latent syphilis occurs later. Patients that have any central nervous system (CNS) involvement are considered to have neurosyphilis.
Screening for syphilis is typically performed when the patient presents with symptoms, has had sexual intercourse with a known infected partner, has been diagnosed with other STDs, or has engaged in other high-risk behaviors. In pregnant women, screening is typically performed at the patient's first prenatal visit.22 Diagnosis of syphilis is very complex and is not within the scope of this article. The 2 types of tests used are nontreponemal and treponemal, and both are commonly used for diagnosis and follow-up.

Bacterial vaginosis. Bacterial vaginosis (BV) is a polymicrobial syndrome that causes 10% to 30% of infectious vaginitis cases in women of childbearing age.24 Bacterial vaginosis is the result of an overgrowth of Gardnerella vaginalis, Mycoplasma hominis, Mobiluncus species, Bacteroides species (excluding Bacteroides fragilis), and Peptostreptococcus species. Although 50% of women with BV are asymptomatic, some may complain of a malodorous vaginal discharge, and approximately half of those women may have itching, burning, and frictional dyspareunia.25 The cause of BV is unclear, and it is not known if there is a link to a sexually transmitted pathogen. However, BV is not commonly found in women who are not sexually active. BV tends to be associated with multiple sex partners or a new sex partner, douching, and a lack of vaginal lactobacilli.
BV is diagnosed using specific clinical criteria known as the Amsel criteria. The 4 criteria are thin, homogenous, white discharge coating vaginal walls; presence of clue cells on microscopic evaluation; pH of vaginal fluid >4.5; and fishy odor of vaginal discharge before or after application of 10% KOH (ie, the whiff test). Patients having 3 of the 4 criteria have a 90% likelihood of having BV.24 Although it is usually impractical in a clinic setting, a Gram stain of vaginal discharge can also be predictive of BV.
BV has been associated with PID and many other complications; therefore, treatment of women who are symptomatic is required (see Table 1 for treatment recommendations).
Commonly, women will experience recurrent BV. These women can be treated with the same recommended or alternative regimens (metronidazole and clindamycin) but for longer periods of time (ie, 10–14 d).24 Changing the treatment regimen for a recurrence of BV can also be considered.4 Women with multiple recurrences should be managed by a specialist. One study demonstrated that women with recurrent BV who were treated with twice-weekly metronidazole 0.75% vaginal gel for 16 weeks successfully maintained a clinical cure for 12 weeks.26 Other studies have assessed oral or vaginal replacement of lactobacillus, but no evidence of a decrease in recurrence rates has been demonstrated.24,27
Trichomoniasis. There are >170 million cases of trichomoniasis annually worldwide, including >7.4 million new cases annually in North America.4,28 Trichomoniasis vaginalis is the protozoan that causes trichomoniasis. Men who are infected may be asymptomatic or may have nongonococcal urethritis. Women who are infected may have no or minimal symptoms. Women who are symptomatic have a diffuse, malodorous, yellow-green discharge along with vulvar irritation.4
Microscopy of vaginal secretions is used most frequently as a diagnostic tool, although this method is only 60% to 70% sensitive for trichomoniasis. Many point-of-care tests are available for screening, but these tests often result in false positives, particularly in low-prevalence populations. For women, a culture of vaginal secretions is the most sensitive and accessible method for diagnosis when microscopy is not conclusive. For men, wet prep microscopy is not useful. A culture for T vaginalis from urethral swab, urine, and semen has the highest sensitivity.4
Only the nitroimidazoles metronidazole and tinidazole are effective in treating trichomoniasis. Studies have demonstrated that tinidazole 2 g is equivalent or superior to metronidazole 2 g. 4 Tinidazole has been demonstrated to have a longer half-life than metronidazole and can achieve higher concentrations in genitourinary tissues.4,29 Metronidazole vaginal gel is not as effective as oral metronidazole, as it does not reach therapeutic levels in the urethra and vaginal glands.4 See Table 1 for treatment recommendations.
Although it has been demonstrated that trichomoniasis is associated with adverse outcomes in pregnancy, there are no data supporting the hypothesis that treatment decreases the chances of morbidity. At the very least, treatment will help to alleviate symptoms in pregnant women, reduce the risk of further sexual transmission, and may prevent infection in the newborn.4 Patients being treated for trichomoniasis should ensure that all of their sexual partners are also treated to avoid further infection and transmission. Patients should also be educated to avoid sex until both they and their partners have completed therapy and are asymptomatic.
PID. PID consists of inflammatory disorders of the upper female genital tract, including endometritis, salpingitis, tubo-ovarian abscesses, and pelvic peritonitis.4 Both N gonorrhoeae and C trachomatis have been demonstrated to cause many cases of PID. On the other hand, many organisms that are part of the normal vaginal flora, such as G vaginalis, Haemophilus influenzae, enteric gram-negative rods, and Streptococcus agalactiae, can also cause PID. In some cases, cytomegalovirus, M hominis, Ureaplasma urealyticum, and Mycoplasma genitalium can be responsible for PID. It is recommended that all women with a diagnosis of acute PID should be tested for N gonorrhoeae, C trachomatis, and HIV.
Infection with PID can involve many signs and symptoms, so diagnosis can be difficult. However, timely diagnosis and treatment are very important, as a delay can result in inflammatory sequelae in the upper reproductive tract. Laparoscopy is very effective in diagnosing salpingitis and specific bacterial infections.4 However, it is not rational to use laparoscopy when the patient's symptoms are vague, and laparoscopy is not readily available in every clinic. Diagnosis based on clinical findings is not precise, as there is not a single key historical, physical, or laboratory finding that is specific for PID, and combinations of these might exclude actual cases of PID. Screening for PID should therefore include the ability to recognize when the disease should be suspected; other diagnostic techniques can then be used.
In general, all treatments for acute PID should cover N gonorrhoeae and C trachomatis whether these organisms are detected or not, since they may be present in the upper reproductive tract. Eradication of anaerobes may prevent destruction of tubal and epithelial cells, and so antianaerobic therapy should be added to treatment regimens.4 Also, BV is frequently present along with PID, so treatment regimens should include metronidazole if appropriate. Azithromycin has been studied recently for this indication; clinical cure rates were demonstrated to be the same as the rates observed with other treatment regimens, and azithromycin was associated with better compliance than multidrug regimens.30 Although women with mild-to-moderate acute PID benefit from either oral or parenteral treatment, the CDC suggests that women be hospitalized for parenteral treatment if any of the following criteria are present: a surgical emergency (eg, appendicitis) cannot be ruled out; the patient is pregnant; the patient has no clinical response to oral antibiotics; the patient is unable to follow or tolerate an outpatient oral regimen; the patient is severely ill with nausea, vomiting, or high fever; or the patient has tubo-ovarian abscess. The CDC's recommended treatment regimens for PID have been demonstrated to have a 90% overall cure rate.30
The CDC's treatment recommendations previously included the use of fluoroquinolones such as levofloxacin and ofloxacin. However, in April 2007, the CDC eliminated these drugs from the treatment recommendations because of the increasing prevalence of quinolone-resistant N gonorrhoeae.31
Because of the infectious nature of the causative agents of PID, male sex partners should be treated if they have had sexual contact within the 60 days preceding the woman's onset of symptoms. Although they may be asymptomatic, sex partners are likely to have a urethral gonococcal or chlamydial infection. Therefore, treatment regimens for the partner should be effective against N gonorrhoeae and C trachomatis, as well.4
CONCLUSION
Although many efforts have been made to improve patients' awareness about STDs, reports of STDs have continued to increase in the United States. Patients who are at high risk for STDs should be counseled frequently and made aware of lifestyle activities that put them at greater risk. Treatment of STDs should also include treatment of patients' partners. Overall, patients, especially the younger population, should be screened regularly and counseled to ensure early diagnosis and treatment.
Dr Wasik is a clinical assistant professor, University of Illinois at Chicago, College of Pharmacy, and a clinical pharmacist, Center for Women’s Health, Chicago. Dr Djuric Kachlic is a clinical assistant professor, University of Illinois at Chicago, College of Pharmacy, and a clinical pharmacist, Family Medicine Center, Chicago.
Disclosure Information: The authors report no financial disclosures as related to products discussed in this article.
REFERENCES
1. Weinstock H, Berman S, Cates W. Sexually transmitted diseases among American youth: Incidence and prevalence estimates, 2000. Perspect Sex Reprod Health. 2004;36:6–10.
2. Centers for Disease Control and Prevention. Sexually transmitted disease surveillance, 2007. Atlanta, GA: U.S. Department of Health and Human Services; December 2008.
3. Centers for Disease Control and Prevention. Trends in reportable sexually transmitted diseases in the United States, 2006; national surveillance data for chlamydia, gonorrhea, and syphilis. http://www.cdc.gov/std/stats/trends2006.htm. Update available at http://www.cdc.gov/STD/stats07/trends.htm. Accessed February 3, 2009.
4. Centers for Disease Control and Prevention. Sexually transmitted diseases treatment guidelines 2006 [erratum in MMWR Recomm Rep. 2006;55:997]. Workowski K, Berman S. MMWR Recomm Rep. 2006;55:1–94.
5. Majeroni, B, Ukkadam, S. Screening and treatment for sexually transmitted infections in pregnancy. Am Fam Physician 2007; 76:265–270.
6. Bodley-Tickell A, Olowokure B, Bhaduri S, et al, for the West Midlands STI Surveillance Project. Trends in sexually transmitted infections (other than HIV) in older people: Analysis of data from an enhanced surveillance system. Sex Transm Infect 2008; 84:312–317.
7. Miller K. Diagnosis and treatment of Chlamydia trachomatis infection [erratum in Am Fam Physician. 2008;77:920]. Am Fam Physician 2006;73: 1411–1416.
8. U.S. Preventive Services Task Force. Screening for Chlamydial Infection. June 2007. Agency for Healthcare Research and Quality, Rockville, MD. http://www.ahrq.gov/clinic/uspstf/uspschlm.htm. Accessed February 3, 2009.
9. Trigg B, Kerndt P, Aynalem G. Sexually transmitted infections in pelvic inflammatory disease in women. Med Clin N Am 2008;92:1083–1113.
10. Gupta R, Warren T, Wald A. Genital herpes. Lancet. 2007;370:2127–2137.
11. Patel R, Rompalo A. Managing patients with genital herpes and their sexual partners. Infect Dis Clin N Am. 2005;19:427–438.
12. American College of Obstetricians and Gynecologists. ACOG Committee Opinion No. 344: Human papillomavirus vaccination. Obstet Gynecol 2006;108:699–705.
13. American Cancer Society. Cancer facts and figures 2008. Atlanta: ACS: 2008. http://www.cancer.org/docroot/STT/content/STT_1x_Cancer_Facts_and_Figures_2008.asp?from=fast/. Accessed February 3, 2009.
14. Markowitz L, Dunne E, Saraiya M, Lawson H, Chesson H, Unger E. Quadrivalent Human Papillomavirus Vaccine: Recommendations of the Advisory Committee on Immunization Practices (ACIP). MMWR. 2007;56:1–24.
15. Zimmerman R. HPV Vaccine and its recommendations, 2007. J Fam Pract. 2007;56:S1–5.
16. Gardasil [package insert]. Whitehouse Station, NJ: Merck; 2006.
17. Workowski K, Berman S, Douglas J. Emerging antimicrobial resistance in Neisseria gonorrhoeae: Urgent need to strengthen prevention strategies [erratum in Ann Intern Med. 2008;148:888]. Ann Intern Med. 2008;148:606–613.
18. US Preventive Services Task Force. Screening for gonorrhea. Revised January 2006. Agency for Healthcare Research and Quality, Rockville, MD. http://www.ahrq.gov/clinic/uspstf/uspsgono.htm. Accessed February 3, 2009.
19. Centers for Disease Control and Prevention. Update to CDC's 2006 sexually transmitted diseases treatment guidelines, 2006: fluoroquinolones no longer recommended for treatment of gonococcal infections. MMWR. 2007;56:332–336.
20 Centers for Disease Control and Prevention. Availability of cefixime 400 mg tablets–United States, April 2008. MMWR. 2008;57:435.
21. Golden M, Marra C, Holmes K. Update on syphilis: Resurgence of an old problem. JAMA. 2003;290:1510–1514.
22. Catalonge, N; U.S. Preventive Services Task Force. Screening for syphilis infection: Recommendation statement [erratum in Ann Fam Med. 2004;2:517]. Ann Fam Med. 2004;2:362–365.
23. Centers for Disease Control and Prevention. Drugs for sexually transmitted infections. Treat Guidel Med Letter. 2007;5:81–88.
24. Owen M, Clenney T. Management of vaginitis. Am Fam Physician. 2004;70:2125–2132.
25. Livengood C, Soper D, Sheehan K, et al. Comparison of once-daily and twice-daily dosing of 0.75% metronidazole gel in the treatment of bacterial vaginosis. Sex Transmit Dis. 1999;26:137–142.
26. Sobel J, Ferris D, Schwebke J, et al. Suppressive antibacterial therapy with 0.75% metronidazole vaginal gel to prevent recurrent bacterial vaginosis. Am J Obstet Gynecol. 2006;194:1283–1289.
27. Van Kessel K, Assefi N, Marrazzo J, Eckert L. Common complementary and alternative therapies for yeast vaginitis and bacterial vaginosis: A systematic review. Obstet Gynecol Surv. 2003;58:351–358.
28. Schmid G. Trichomoniasis treatment in women: RHL commentary (last revised: 28 July 2003). The WHO Reproductive Health Library; Geneva: World Health Organization. Available at http://www.who.int/rhl/rti_sti/gscom/en/index.html. Accessed February 3, 2009.
29. Fung H, Doan T-L. Tinidazole: A nitroimidazole antiprotozoal agent. Clin Ther. 2005;27:1859–1884.
30. Lareau S, Beigi R. Pelvic inflammatory disease and tubo-ovarian abscess. Infect Dis Clin N Am. 2008;22:693–708.
31. Updated recommended treatment regimens for gonoccocal infections and associated conditions–United States, April 2007. http://www.cdc.gov/STD/treatment/2006/updated-regimens.htm. Accessed February 3, 2009.
32. Tindamax [package insert]. Arlington Heights, IL: Presutti; 2004.
33. Clindesse [package insert]. St. Louis, MO: Ther-Rx; 2004.
Employers Face Barriers With Adopting Biosimilars
March 1st 2022Despite the promise of savings billions of dollars in the United States, adoption of biosimilars has been slow. A roundtable discussion among employers highlighted some of the barriers, including formulary design and drug pricing and rebates.
