- Safety & Recalls
- Regulatory Updates
- Drug Coverage
- COPD
- Cardiovascular
- Obstetrics-Gynecology & Women's Health
- Ophthalmology
- Clinical Pharmacology
- Pediatrics
- Urology
- Pharmacy
- Idiopathic Pulmonary Fibrosis
- Diabetes and Endocrinology
- Allergy, Immunology, and ENT
- Musculoskeletal/Rheumatology
- Respiratory
- Psychiatry and Behavioral Health
- Dermatology
- Oncology
Review of inhaled corticosteroid/long-acting beta2-agonist combinations
In an effort to simplify therapy for patients with asthma or COPD, products have been designed that combine medications from 2 separate therapeutic classes.

Key Points
- Abstract
Treatment guidelines for patients with the respiratory conditions asthma and chronic obstructive pulmonary disease (COPD) call for complex drug regimens, including multiple inhaled medications from different classes. In an effort to simplify therapy for these patients, products have been designed that combine medications from 2 separate therapeutic classes. This review focuses on inhaled corticosteroid/long-acting beta2-agonist combinations that are available for use for these disease states. The evidence supporting their use in each of these disease states is reviewed, as well as how the agents fit into accepted treatment guidelines. The genesis of the boxed warnings carried by these medications is also described. (Formulary. 2008;43:204–219.)
Both long-acting beta2-agonists (LABAs) and inhaled corticosteroids (ICSs) are commonly used to treat respiratory conditions, primarily asthma and chronic obstructive pulmonary disease (COPD). Patients with either of these disease states are also often treated with medications from other therapeutic classes to alleviate and treat symptoms. Inhaled short-acting beta2-agonists (SABAs) should be prescribed as rescue medications for all patients with asthma or COPD who tolerate them. Patients with COPD are often prescribed an inhaled short-acting or long-acting anticholinergic agent. Other therapeutic classes used to treat asthmatic patients include inhaled mast-cell stabilizers, oral leukotriene antagonists, and monoclonal antibodies. Theophylline is also available, but this agent is rarely used today. It is common for a patient to be taking medications from several of these classes, including a cocktail of ≥2 inhaled medications.

Both of the available ICS/LABA combinations are approved by FDA for the treatment of asthma; only the fluticasone/salmeterol 250 mcg/50 mcg formulation is approved for the treatment of COPD associated with chronic bronchitis. There is also evidence that a combination product containing the SABA albuterol and the ICS beclomethasone is effective for rescue use in patients with mild asthma, but this combination is not yet available in the United States or abroad.1
Pharmacology
Asthma is a chronic inflammatory disorder of the airways in which many cells and cellular elements play a role, including mast cells, eosinophils, T lymphocytes, neutrophils, and epithelial cells.2 Corticosteroids have been demonstrated to inhibit these multiple types of cells and other key factors involved in asthma (ie, histamine, eicosanoids, leukotrienes, and cytokines).
Fluticasone is a synthetic trifluorinated corticosteroid with potent anti-inflammatory activity.3 Fluticasone has been established as a human glucocorticoid receptor agonist with 3 times the potency of budesonide.3 Budesonide is an anti-inflammatory corticosteroid that exhibits potent glucocorticoid activity and weak mineralocorticoid activity.4
Salmeterol and formoterol are both LABAs. LABAs work on the bronchoconstrictive component of asthma or COPD. The pharmacologic effects of these drugs are at least in part attributable to stimulation of intracellular adenyl cyclase, the enzyme that catalyzes the conversion of adenosine triphosphate (ATP) to cyclic-3',5'-adenosine monophosphate (AMP).3 Increased cyclic AMP levels cause relaxation of bronchial smooth muscle and inhibition of release of mediators of immediate hypersensitivity from cells, especially from mast cells.3
The time to a significant sustained improvement in lung function in patients with asthma is 1 week for fluticasone/salmeterol and 2 to 4 weeks for budesonide/formoterol.5,6 However, there is an initial response of bronchodilation within 3 minutes after administration of budesonide/formoterol and 28 minutes for fluticasone/salmeterol.7–9 This quicker onset of bronchodilation has led to the use of formoterol as a rescue medication; this use is not approved in the United States but is approved in Canada and Europe.
EVIDENCE-BASED INDICATIONS
Asthma. The evidence supporting the use of ICS/LABA combinations in patients with asthma is well established. The combinations have been demonstrated to improve symptom scores, decrease nocturnal asthma, improve lung function, decrease the use of SABAs, reduce the number of exacerbations, and achieve clinical control of asthma in more patients, more rapidly, and at a lower dose of ICS than ICS administered alone.2
The Formoterol and Corticosteroids Establishing Therapy (FACET) trial was a randomized, double-blind, parallel-group study in 852 patients aged 18 to 70 years.10 Included patients had asthma for ≥6 months and had been treated with an inhaled glucocorticoid for ≥3 months. All patients were required to have a forced expiratory volume in 1 second (FEV1) <50% of predicted value with an increase of ≥15% in FEV1 from baseline after the inhalation of terbutaline 1 mg. The study had a 4-week run-in period followed by 12 months of treatment. The treatment groups included budesonide 200 mcg/d plus placebo (n=213), budesonide 200 mcg/d plus formoterol 9 mcg (n=210), budesonide 800 mcg/d plus placebo (n=214), and budesonide 800 mcg/d plus formoterol 9 mcg (n=215). It should be noted that the delivery device used in this study was a dry powder inhaler (DPI), not the metered-dose inhaler (MDI) that is available in the United States.
The primary end point was the rate of severe and mild exacerbations of asthma per patient per year. Secondary outcomes included lung function, symptoms, and the need for rescue therapy with terbutaline. The lowest rates of exacerbations were observed among patients who received the higher dose of budesonide plus formoterol. The rate of severe exacerbations was reduced by 26% when formoterol was added to the lower dose of budesonide. The combination of formoterol and the higher dose of budesonide reduced the estimated rate of severe exacerbations by 63%, from 0.91 to 0.34 per year per patient (P<.001), and mild exacerbations by 62%, from 35.4 to 13.4 per year per patient (P<.001). Throughout the study, 80.8% of the patients receiving both formoterol and the higher dose of budesonide were free of severe exacerbations. Pulmonary function and symptoms also improved during therapy with these regimens, with more noticeable improvements with the addition of formoterol rather than with an increased dose of budesonide alone.10
Noonan et al6 conducted a 12-week, randomized, double-blind, double-dummy, placebo-controlled study in 596 patients ≥12 years of age. Patients had moderate-to-severe asthma and had previously been treated with an ICS. Included patients had an FEV1 of 45% to 85% of predicted value, with a postbronchodilator reversibility of ≥12%. Patients were also required to have used moderate-to-high ICS doses consistently for >4 weeks before enrollment. During a 2-week run-in period, patients were to discontinue all of their asthma medications and received budesonide 80 mcg per inhalation (2 inhalations twice daily) and as-needed salbutamol. Patients were randomized to 1 of 5 groups: budesonide/formoterol 320 mcg/9 mcg twice daily, budesonide 320 mcg twice daily, formoterol 9 mcg twice daily, budesonide 320 mcg plus formoterol 9 mcg twice daily (in separate inhalers), and placebo.
The 2 primary efficacy variables were mean change from baseline in morning predose FEV1 obtained approximately 12 hours after the most recent administration of study medication and mean change from baseline FEV1 assessed as the average change in FEV1 from serial spirometry over the 12-hour period after administration of the morning dose of study medication.6
Mean changes from baseline in morning predose FEV1 at end of treatment were greater (P≤.049) with budesonide/formoterol (0.19 L) versus budesonide (0.10 L), formoterol (–0.12 L), and placebo (–0.17 L). Mean changes from baseline in 12-hour FEV1 were greater (P≤.001) with budesonide/formoterol after 1 day (0.37 L), 2 weeks (0.34 L), and at end of treatment (0.37 L) versus budesonide (0.11, 0.15, and 0.15 L, respectively) and placebo (0.09, –0.03, and –0.03 L, respectively) and after 2 weeks and at end of treatment versus formoterol (0.19 and 0.17 L, respectively). Fewer patients receiving budesonide/formoterol versus single products or placebo met worsening asthma criteria (P≤.025). Results were similar in the budesonide/formoterol group and the budesonide plus formoterol group on all measures. All treatments were well tolerated with similar safety profiles.6
The American Lung Association Asthma Clinical Research Centers conducted a 16-week double-blind trial in 500 patients with asthma that was well controlled by inhaled fluticasone 100 mcg twice daily. The investigators' hypothesis was that these patients would do as well with either montelukast or fluticasone/salmeterol once daily. Patients were randomized to fluticasone 100 mcg twice daily (n=169), montelukast 5 or 10 mg every night (n=166), or fluticasone 100 mcg plus salmeterol 50 mcg every night (n=165).11
The primary outcome measure was the time to treatment failure. Secondary outcomes included measures of peak expiratory flow (PEF) from diary cards and FEV1, measures of asthma symptoms and medication use from the patients' daily diary cards, number of days on which patients were free of asthma symptoms, and scores related to quality of life. The rates of treatment failure were 20.2% and 20.4% in the fluticasone group and fluticasone/salmeterol group, respectively, and 30.3% in the montelukast group, which represented an approximately 60% higher rate in the montelukast group as compared with the other 2 groups (HR=1.6; 95% CI, 1.1–2.6; P=.03 for both comparisons). Decrease in FEV1 by ≥20% from baseline was the most common reason for treatment failure (47.8%). Mean bronchodilator FEV1 values were higher in the fluticasone group (91.1% of predicted value) and in the fluticasone/salmeterol group (91.8% of predicted value) than in the montelukast group (88.8% of predicted value) (P=.002 and P<.001, respectively). The high percentages of symptom-free days in all 3 treatment groups (montelukast, 78.7%; fluticasone, 85.8%; fluticasone/salmeterol, 82.7%) suggest that most patients fared well.11
COPD. In late 2003, the FDA approved fluticasone/salmeterol 250 mcg/50 mcg for use in COPD. Several trials have indicated that a LABA plus an ICS is superior to placebo, LABA alone, or ICS alone.12–14 However, the role of combination therapy is still being established.
Combined ICS/LABA use in COPD was assessed by Hanania et al.12 A randomized, double-blind, placebo-controlled, parallel-group, multicenter trial evaluated 723 patients with COPD and with symptoms of chronic bronchitis and moderate dyspnea. Patients were randomized to receive salmeterol 50 mcg (n=177), fluticasone 250 mcg (n=183), the combination of fluticasone 250 mcg plus salmeterol 50 mcg (n=178), or placebo (n=185) for 24 weeks. The combination was demonstrated to be superior to salmeterol and placebo but not fluticasone in improving the morning predose FEV1; the combination was demonstrated to be superior to salmeterol, fluticasone, and placebo in improving 2-hour postdose FEV1. However, the combination was not demonstrated to be superior to fluticasone or salmeterol alone in improving health status or reducing exacerbation rates.
Although this study did not lead to FDA approval of this combination therapy for the treatment of COPD, the trial did establish the combination's potential efficacy in special populations, specifically in patients who experience dyspnea despite monotherapy with LABAs.15 The authors encouraged clinicians to assess patients within 4 weeks of treatment initiation for improvements in symptoms, activities of daily living, and overall health status. They suggested that therapy should be discontinued if no benefit is observed.15
In an effort to determine if the use of ICS/LABAs affected mortality in patients with COPD, the Towards a Revolution in COPD Health (TORCH) trial was designed.13 The TORCH trial was a randomized, double-blind trial comparing treatment with fluticasone/salmeterol 500 mcg/50 mcg twice daily (n=1,533), salmeterol alone (n=1,521), fluticasone alone (n=1,534), or placebo (n=1,524), for 3 years. Included patients had a ≥10-pack-year history of smoking, mean age of 65 years (range, 40–80 y), diagnosis of COPD with prebronchodilator FEV1 <60%, an increase of FEV1 <10% of predicted value for that patient with the use of albuterol 400 mcg, and a ratio of prebronchodilator FEV1 to forced vital capacity (FVC) ≤0.7. Before the 2-week run-in period, all use of corticosteroids and inhaled LABAs was stopped, but patients could continue their other COPD medications.
The primary end point was the time to death from any cause at 3 years. An independent blinded clinical end point committee was responsible for determining whether deaths were related to COPD. Secondary end points were the frequency of exacerbations (requiring treatment with antibiotics, systemic corticosteroids, hospitalization, or a combination of these) and health status using the Saint George's Respiratory Questionnaire (SGRQ).13
The results demonstrated that the probability of death from any cause at 3 years was 12.6% in the combination group, 13.5% in the salmeterol group, 16% in the fluticasone group, and 15.2% in the placebo group. The HR for the combination group compared with placebo for death from any cause was 0.825 (95% CI, 0.681–1.002; P=.052) and 0.78 (95% CI, 0.57–1.06; P=.11) for death from COPD. The probability of death from COPD at 3 years was 4.7% in the combination group, 6.1% in the salmeterol group, 6.9% in the fluticasone group, and 6% in the placebo group. There was an increased probability of pneumonia during the 3 years in any treatment group containing fluticasone (combination, 19.6% and fluticasone, 18.3% vs salmeterol, 13.3% and placebo, 12.3%). The annual rate of exacerbations was lower in the combination group (0.85; 95% CI, 0.8–0.9) than in the placebo group (1.13; 95% CI, 1.07–1.2), and annual hospital admission rate was 17% lower (P<.03) in the combination group compared with the placebo group.13
In an editorial, Dr Rabe, chair of the science committee for the Global Initiative for Obstructive Lung Disease (GOLD), addressed the findings of the TORCH trial. He reiterated the point that the TORCH trial did not demonstrate a statistically significant improvement with ICS/LABA therapy compared with placebo in its primary end point, death from any cause. However, the trial did demonstrate that ICS/LABAs provided a statistically significant advantage in health status, frequency of exacerbations, use of oral steroids, and protection against a decline in lung function. He stated that these results were important and warranted further investigation.16
The most significant benefits of ICS/LABA combinations for patients with COPD are observed in patients with severe disease with repeated exacerbations. Kardos et al14 conducted a randomized, multicenter, double-blind clinical trial that investigated the combination of salmeterol and fluticasone (n=507) versus salmeterol alone (n=487) on exacerbation rates in COPD patients.
In this trial, patients with severe COPD (postbronchodilator FEV1<50% predicted), a ≥10-pack-year history of smoking, and ≥2 moderate-to-severe exacerbations in the prior year were required to complete a 4-week run-in period with stable medications and no exacerbations. Patients were then treated with the combination of salmeterol 50 mcg and fluticasone 500 mcg or salmeterol 50 mcg alone. During the run-in and treatment periods, inhaled albuterol was used as rescue medication, and regular treatment with short-acting bronchodilators, antioxidants/mucolytics, SABAs, and theophylline was permitted. All non-COPD medications, including preexisting selective beta-blocker therapy, could be continued if the dose remained constant. Acute moderate and severe exacerbations were treated with systemic corticosteroids and/or antibiotics. Long-acting bronchodilators (including tiotropium) and systemic steroids (except as treatment for exacerbations) were not permitted during the study.14
The primary end point was the number of moderate (ie, worsening symptoms that require a change in medication and medical assistance) and severe (ie, hospitalization or emergency room treatment) exacerbations in each treatment group. Secondary end points included time to first exacerbation, PEF, postbronchodilator FEV1, and disease-specific quality of life as assessed by the SGRQ.14
The study results demonstrated that the number of moderate/severe exacerbations in the combination group was 334 compared with 464 in the salmeterol group (P<.0001), corresponding to annualized rates of 0.92 per patient in the combination group and 1.4 per patient in the salmeterol group (P<.0001). The combination group demonstrated significantly improved mean time to first exacerbation (128 d) versus the salmeterol group (93 d; HR=1.47; 95% CI, 1.21–1.79). The study results demonstrated that in order to prevent 1 moderate/severe exacerbation per year, the number of patients needed to treat with combination therapy versus salmeterol is 2.08. The combination group also demonstrated significantly improved PEF (18 L/min) compared with the salmeterol group (4.4 L/min; P<.0001). The number of patients with clinically meaningful improvement of SGRQ was significantly higher in the combination group than in the salmeterol group (41.7% vs 30%; P=.0019). Adverse events were similar in both treatment groups; oropharyngeal candidiasis was the most frequent drug-related adverse event (n=8). Suspected pneumonia occurred more frequently in the combination group than in the salmeterol group (23 cases vs 7 cases).14
Fluticasone/salmeterol vs budesonide/formoterol. The Exacerbation Control Evaluated in a 6 month Long (EXCEL) study compared fluticasone/salmeterol and budesonide/formoterol in patients with persistent asthma.17 This randomized, multicenter, double-dummy, parallel-group trial assessed the effects of these combinations on the rate of exacerbations. Patients were treated with either fluticasone/salmeterol 250 mcg/50 mcg (1 inhalation twice daily) (n=694) or budesonide/formoterol 200 mcg/6 mcg (2 inhalations twice daily) (n=697).
The 2 groups demonstrated similar exacerbation rates over 24 weeks (fluticasone/salmeterol, 2.69; budesonide/formoterol, 2.79; HR=0.96; 95% CI, 0.84–1.10; P=.571).17
Safety was also assessed and was observed to be similar in the 2 groups. The most common medication-related adverse events were hoarseness (fluticasone/salmeterol, 2%; budesonide/formoterol, 2%), oral candidiasis (fluticasone/salmeterol, 2%; budesonide/formoterol, 1%), and headache (fluticasone/salmeterol, 1%; budesonide/formoterol, 2%).17
Formoterol's faster onset of bronchodilation compared with salmeterol has led to the strategy of further simplifying medication regimens by using formoterol (or budesonide/formoterol) as both maintenance and rescue therapy. Kuna et al18 conducted a 6-month randomized, double-blind study that enrolled 3,335 patients who were aged ≥12 years, had been diagnosed with asthma ≥6 months before the start of the study, and who had been using ICS for >3 months. There were 3 treatment arms in the study: budesonide/formoterol 160 mcg/4.5 mcg (1 inhalation twice daily) plus rescue inhalations as needed (n=1,107), salmeterol/fluticasone 25 mcg/125 mcg (2 inhalations twice daily) plus terbutaline for rescue (n=1,123), or budesonide/formoterol 320 mcg/9 mcg (1 inhalation twice daily) plus terbutaline for rescue (n=1,105).
The primary end point of the study was the time to first severe exacerbation (ie, need for hospitalization, emergency room treatment, or oral corticosteroids for ≥3 d). Secondary outcomes included measures of day-to-day asthma control, including lung function, symptom-free days, and asthma-related quality of life. The budesonide/formoterol maintenance and reliever therapy 160 mcg/4.5 mcg prolonged the time to first exacerbation compared with patients receiving salmeterol/fluticasone plus terbutaline (P=.0034) and patients receiving budesonide/formoterol 320 mcg/9 mcg plus terbutaline (P=.023). Patients in the budesonide/formoterol 160 mcg/4.5 mcg group also experienced fewer total exacerbations compared with patients receiving salmeterol/fluticasone plus terbutaline (RR=0.61; 95% CI, 0.49–0.76; P<.001) and compared with those receiving budesonide/formoterol 320 mcg/9 mcg plus terbutaline (RR=0.72; 95% CI, 0.57–0.90; P=.0048). Day-to-day control of asthma was similar among treatment groups; all treatments demonstrated similar improvements in lung function, asthma control days, and asthma-related quality of life.
The investigators concluded that the combination of increased efficacy and simplicity associated with budesonide/formoterol for maintenance and relief represents a significant improvement over fixed, twice-daily combinations of higher-dose ICS/LABA, which until now have been regarded as the most effective way to manage moderate-to-severe persistent asthma. The relatively low cost of SABAs compared with this combination product, however, may reduce the attractiveness of this strategy to healthcare systems.
Multiple delivery devices. Fluticasone/salmeterol is available as both a DPI and an MDI (see Table 1). Bateman et al19 conducted a randomized, multicenter, double-blind, double-dummy, parallel-group study to investigate the hypothesis of equivalent efficacy and comparable safety of salmeterol/fluticasone MDI versus DPI. The study evaluated 497 patients aged 11 to 79 years who had mild-to-moderate asthma. Patients entered a 2-week run-in period of ICS therapy, after which they were assigned to 12 weeks of salmeterol/fluticasone 25 mcg/50 mcg (2 inhalations twice daily) by MDI (n=165), salmeterol/fluticasone 50 mcg/100 mcg (1 inhalation twice daily) by DPI (n=167), or fluticasone 50 mcg (2 inhalations twice daily) by MDI (n=165).
The primary end point was increase in mean morning PEF from Weeks 1 to 12. The trial demonstrated that the ICS/LABA combination was significantly more effective than ICS monotherapy in producing an improvement in morning PEF (adjusted mean treatment difference, Weeks 1–12, –19 L/min; 95% CI, –28 to –11; P<.001). FEV1 measurements improved from baseline among all 3 groups; there was no statistically significant difference among the groups. Patients in the salmeterol/fluticasone MDI group had significantly more symptom-free days than patients receiving fluticasone (55% vs 25%; P=.001).19 The results of the trial demonstrated that the single-inhaler combination of salmeterol/fluticasone was equally effective for treatment of mild-to-moderate asthma when delivered by either DPI or MDI.
GUIDELINE-BASED USE
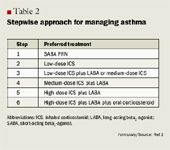
For patients with asthma whose symptoms are not controlled by low-dose ICS alone (and require Step 3 of therapy), providers have the option of prescribing either low-dose ICS plus a LABA combination or medium-dose ICS alone (see Table 2).2 This is a departure from the 2002 update of the previous guideline (NAEPP EPR2), which recommended only low- to medium-dose ICS plus a LABA.21 All patients with severe persistent asthma (see Table 3) should receive a medium-dose ICS plus a LABA (Step 4) or a high-dose ICS plus a LABA (Steps 5 and 6).2 LABAs are not to be used for monotherapy in asthma but only as add-on therapy to ICS when the ICS alone is insufficient.2
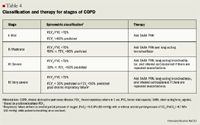
For the treatment of COPD, ICS/LABA combinations should be reserved for patients with Stage III or IV (severe or very severe) disease associated with chronic bronchitis who have repeated exacerbations of their disease (see Table 4).20
Dose Selection

For the treatment of asthma, the stepwise approach to therapy should be used (see Table 2).2 Step 3 of this approach recommends a low-dose ICS combination plus a LABA. As asthma severity increases, so should the ICS potency. Therefore, for Step 6 (severe asthma), the recommendation includes high-dose ICS, LABA, and oral corticosteroid. The categorization of dosing strength is addressed in Table 1.
For the treatment of COPD, fluticasone/salmeterol 250 mcg/50 mcg is the appropriate and approved dose administered as 1 inhalation twice daily; when using an MDI, the 115 mcg/21 mcg dose administered as 2 inhalations twice daily is recommended.
SAFETY
All LABAs, including those that are components of a combination product, are contraindicated in the United States for acute episodes of shortness of breath associated with COPD or asthma or in instances of status asthmaticus.3,4 SABAs such as albuterol are the preferred agents for acute dyspnea. Of note, budesonide/formoterol is approved for acute shortness of breath in Canada and Europe; this is because of formoterol's onset of bronchodilation of 1 to 3 minutes.4
The labeling for both of these products (fluticasone/salmeterol and budesonide/formoterol) carries a boxed warning.3,4 Labeling for all LABAs in the United States must contain the warning that these agents may increase the risk of asthma- related death. This presents a dilemma to practitioners and pharmacists, as LABAs are approved by FDA for the treatment of asthma and are a part of all treatment guidelines for asthma. It is important for pharmacists to understand the genesis of this warning.
The Salmeterol Multicenter Asthma Research Trial (SMART) assessed 26,355 patients with asthma to determine the safety of salmeterol when added to usual asthma therapy.23 The study was prematurely stopped after a planned interim analysis was performed and investigators observed that patients receiving salmeterol were at increased risk for fatal asthma events compared with patients treated with placebo (0.10% vs 0.02%; RR=4.37; 95% CI, 1.25–15.34). Post-hoc subpopulation analyses were then performed. Investigators observed that in Caucasians, asthma-related death occurred at a higher rate in patients treated with salmeterol compared with patients treated with placebo (0.07% vs 0.01%; RR=5.82; 95% CI, 0.70–48.37). Also, in African Americans, asthma-related death occurred at a higher rate in patients treated with salmeterol compared with those treated with placebo (0.31% vs 0.04%; RR=7.26; 95% CI, 0.89–58.94).23 Overall, of the 26,355 patients enrolled, there were 16 asthma-related deaths; 13 were in the salmeterol group and 3 were in the placebo group. Of the 13 in the salmeterol group, 6 were Caucasians (out of 9,281) and 7 were African Americans (out of 2,319). Among placebo-treated patients, there was 1 death in the Caucasian group and 1 death in the African American group. The race of the third patient in the placebo group who experienced an asthma-related death was unknown.
As pointed out by Cazzola and Matera,24 chronic use of anti-inflammatory agents (such as ICS) was not required in the SMART trial. Only 47% of the total patient population in this trial received ICS at baseline, and in the subgroup that experienced the most deaths (African American patients), only 37% of patients were using ICS at baseline.23 Another possible confounder was that adherence to ICS was not assessed throughout the study. LABAs are intended as add-on therapies; anti-inflammatory agents, particularly ICS, are the cornerstone for all asthma treatments (see Table 2). It is possible that the low rate of ICS use in SMART, particularly among African American patients, had more of an effect on asthma-related deaths than the salmeterol use. Monotherapy with LABA agents for asthma is not appropriate; using ICS/LABA combinations will ensure that patients with asthma receive the needed anti-inflammatory component of therapy.
The controversy over the safety of LABAs in patients with asthma was debated among asthma specialists at the 2006 Eastern Allergy Conference.25 The consensus reached was that a low-dose ICS plus a LABA is preferred to a medium-dose ICS for a patient requiring step-up therapy from a low-dose ICS, regardless of race.
The FACET trial assessed the effect of adding a LABA, formoterol, for patients with asthma who were already receiving an ICS.10 Patients were required to have been taking an ICS for ≥3 months to be included; before randomization, there was a 4-month run-in period during which patients received inhaled budesonide 800 mcg twice daily. After the run-in period, patients received budesonide 100 mcg twice-daily plus placebo (n=213), budesonide 100 mcg plus formoterol 12 mcg (n=210), budesonide 400 mcg plus placebo (n=214), or budesonide 400 mcg plus formoterol 12 mcg (n=215). In this study (in which all patients received ICS), there were no asthma-related deaths, and the number of severe asthma exacerbations per patient per year was lower for formoterol than for placebo when combined with lower-dose ICS (0.67 vs 0.91; P=.01) and with higher-dose ICS (0.34 vs 0.46; P=.01).
It is important for pharmacists to remember that monotherapy with LABAs in patients with asthma increases the risk for severe asthma exacerbations and asthma-related death. Patients who are prescribed an ICS and a LABA for their asthma must be counseled on the importance of using their ICS as prescribed on a daily basis. Also, providers who prescribe LABAs for purposes other than add-on therapy in patients with asthma should be reminded of this risk. Using combination ICS/LABA products reduces the risk of patients only adhering to the LABA component of therapy and should be encouraged for patients with asthma whose symptoms are not controlled with ICS monotherapy.
INAPPROPRIATE PRESCRIBING
There is significant evidence demonstrating that ICS/LABA combinations are effective for treating patients with asthma. Evidence also exists demonstrating an improved rate of exacerbations for patients with severe COPD with these combinations.13,14 However, it must be remembered that neither of the combinations is a first-line drug for patients with intermittent or mild persistent asthma or with mild-to-moderate COPD and should not be prescribed at the first complaint of dyspnea or wheezing.
For patients with intermittent asthma (see Table 3), a SABA dosed on an as-needed basis should be the initial treatment. Additionally, mild asthma is often well controlled with an ICS alone with as-needed SABA.
There is no role for ICS/LABA combinations in the treatment of acute pulmonary conditions such as the flu, acute bronchitis, pneumonia, or other respiratory infections in patients who do not have COPD or asthma.
CONCLUSION
Combining medications from different classes into a single product is a safe and efficacious strategy, when used appropriately, to simplify complex therapeutic plans. In addition to convenience and improved medication compliance, patients who have co-pays for each prescription filled will be able to save money. This can also reduce the burden on health-system pharmacies by reducing the number of prescriptions processed.
Asthma and COPD are disease states that often require complex medication regimens. Combining ICS with LABAs is an effective strategy to simplify these regimens. Budesonide/formoterol and fluticasone/salmeterol are now available, and more combinations are likely to be available in the future.
Combination products are available in a range of doses, providing flexibility to practitioners to ensure that convenience does not compromise appropriateness of dose.
Dr Roefaro is a clinical pharmacy specialist and the PGY-1 pharmacy residency director, VA Boston Healthcare System, Boston, Massachusetts. He can be reached at john.roefaro@va.gov
. Dr Daryanani is a clinical pharmacy specialist, VA Boston Healthcare System.
Disclosure Information: The authors report no financial disclosures as related to products discussed in this article.
REFERENCES
1. Papi A, Canonica GW, Maestrelli P, et al; BEST Study Group. Rescue use of beclomethasone and albuterol in a single inhaler for mild asthma. N Engl J Med. 2007;356:2040–2052.
2. Expert Panel Report 3 (EPR3): Guidelines for the Diagnosis and Management of Asthma. National Institutes of Health. National Heart, Lung, and Blood Institute. Publication No. 08-4051, 2007. http:// http://www.nhlbi.nih.gov/guidelines/asthma/ . Accessed May 15, 2008.
3. Advair [package insert]. Research Triangle Park, NC: GlaxoSmithKline; 2006.
4. Symbicort [package insert]. Dunkerque, France: AstraZeneca; 2007.
5. Kavuru M, Melamed J, Gross G, et al. Salmeterol and fluticasone propionate combined in a new powder inhalation device for the treatment of asthma: A randomized, double-blind, placebo-controlled trial. J Allergy Clin Immunol. 2000;105;6(part 1):1108–1116.
6. Noonan M, Rosenwasser LJ, Martin P, O'Brien CD, O'Dowd L. Efficacy and safety of budesonide and formoterol in one pressurised metered-dose inhaler in adults and adolescents with moderate to severe asthma: A randomised clinical trial. Drugs. 2006;66:2235–2254.
7. Bateman ED, Fairall L, Lombardi DM, English R. Budesonide/formoterol and formoterol provide similar rapid relief in patients with acute asthma showing refractoriness to salbutamol. Respir Res. 2006;7:13–21.
8. Palmqvist M, Arvidsson P, Beckman O, Peterson S, Lotvall J. Onset of bronchodilation of budesonide/formoterol vs. salmeterol/fluticasone in single inhalers. Pulm Pharmacol Ther. 2001;14:29–34.
9. Spencer CM, Jarvis B. Salmeterol/fluticasone propionate combination. Drugs. 1999;57:933–940.
10. Pauwels RA, Lofdahl CG, Postma DS, et al. Effect of inhaled formoterol and budesonide on exacerbations of asthma. Formoterol and Corticosteroids Establishing Therapy (FACET) International Study Group. N Engl J Med. 1997;337:1405–1411.
11. American Lung Association Asthma Clinical Research Centers, Peters SP, Anthonisen N, Castro M, et al. Randomized comparison of strategies for reducing treatment in mild persistent asthma. N Engl J Med. 2007;356:2027–2039.
12. Hanania NA, Darken P, Horstman D, et al. The efficacy and safety of fluticasone propionate (250 microg)/salmeterol (50 microg) combined in the Diskus inhaler for the treatment of COPD. Chest. 2003;124:834–843.
13. Calverley PM, Anderson JA, Celli B, et al; TORCH investigators. Salmeterol and fluticasone propionate and survival in chronic obstructive pulmonary disease. N Engl J Med. 2007;356:775–789.
14. Kardos P, Wencker M, Glaab T, Vogelmeier C. Impact of salmeterol/fluticasone propionate versus salmeterol on exacerbations in severe chronic obstructive pulmonary disease. Am J Respir Crit Care Med. 2007;175:144–149.
15. National Collaborating Centre for Chronic Conditions. Chronic obstructive pulmonary disease. National clinical guideline on management of chronic obstructive pulmonary disease in adults in primary and secondary care. Thorax. 2004;59(suppl 1):1–232.
16. Rabe KF. Treating COPD–The TORCH trial, p values, and the dodo. N Engl J Med. 2007;356:851–854.
17. Dahl R, Chuchalin A, Gor D, Yoxall S, Sharma R. EXCEL: A randomised trial comparing salmeterol/fluticasone propionate and formoterol/budesonide combinations in adults with persistent asthma. Respir Med. 2006:100;1152–1162.
18. Kuna P, Peters MJ, Manjra AI, et al. Effect of budesonide/formoterol maintenance and reliever therapy on asthma exacerbations. Intl J Clin Pract. 2007;61:725–736.
19. Bateman ED, Silins V, Bogolubov M. Clinical equivalence of salmeterol/fluticasone propionate in combination (50/100 microg twice daily) when administered via a chlorofluorocarbon-free metered dose inhaler or dry powder inhaler to patients with mild-to-moderate asthma. Respir Med. 2001;95:136–146.
20. Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease. Executive summary. Updated 2007. http:// http://www.goldcopd.com/guidelineitem.asp?l1=2&l2=1&intId=996. Accessed May 15, 2008.
21. Expert Panel Report: Guidelines for the Diagnosis and Management of Asthma. Update on Selected Topics 2002. http:// http://www.nhlbi.nih.gov/guidelines/asthma/. Accessed May 15, 2008.
22. A 6-month double-blind, double-dummy, randomized, parallel group, multicenter efficacy & safety study of Symbicort pMDI 2 x 160/4.5 mcg & 80/4.5 mcg bid compared to formoterol TBH, budesonide pMDI (& the combination) & placebo in COPD patients. Unpublished data provided by AstraZeneca. Accessed April 17, 2008.
23. Nelson HS, Weiss ST, Bleecker ER, Yancey SW, Dorinsky PM; SMART Study Group. The Salmeterol Multicenter Asthma Research Trial: A comparison of usual pharmacotherapy for asthma or usual pharmacotherapy plus salmeterol. Chest. 2006;129:15–26.
24. Cazzola M, Matera MG. Review: Safety of long-acting beta2-agonists in the treatment of asthma. Therapeutic Advances in Respiratory Disease. 2007;1:35–46.
25. Lang DM, Davis RS. The long-acting beta-agonist controversy: A clinical dilemma. Allergy Asthma Proc. 2007;28:136–144.
Payers Recognize the Benefits, but Still See Weight Loss Drugs through a Cost Lens
April 12th 2024Jeffrey Casberg, M.S., R.Ph., a senior vice president of clinical pharmacy at IPD Analytics LLC, a drug intelligence firm that advises payers and pharmaceutical companies, talks about how payers are thinking about weight-loss drugs.
