- Safety & Recalls
- Regulatory Updates
- Drug Coverage
- COPD
- Cardiovascular
- Obstetrics-Gynecology & Women's Health
- Ophthalmology
- Clinical Pharmacology
- Pediatrics
- Urology
- Pharmacy
- Idiopathic Pulmonary Fibrosis
- Diabetes and Endocrinology
- Allergy, Immunology, and ENT
- Musculoskeletal/Rheumatology
- Respiratory
- Psychiatry and Behavioral Health
- Dermatology
- Oncology
Rifaximin: A nonabsorbable, broad-spectrum antibiotic for reduction in the risk for recurrence of overt hepatic encephalopathy
A recent landmark phase 3 trial in patients in remission from recurrent hepatic encephalopathy demonstrated that rifaximin at a dose of 550 mg twice daily is significantly more effective than placebo in maintaining remission and reducing the risk of hospitalization. Rifaximin's approval by FDA in March 2010, offers clinicians the first pharmacologic treatment for hepatic encephalopathy in more than 30 years.

Key Points
Abstract
Rifaximin is a broad-spectrum antibiotic that has been approved by FDA for reduction in the risk for recurrence of overt hepatic encephalopathy, a debilitating disorder caused by the inability of the liver to remove bacterial-derived toxic by-products, specifically ammonia. It is a nonabsorbable antibiotic that provides activity locally in the gut due to its negligible systemic absorption. Rifaximin has a favorable side-effect profile and a low potential for drug interactions. A recent landmark phase 3 trial in patients in remission from recurrent hepatic encephalopathy demonstrated that rifaximin at a dose of 550 mg twice daily is significantly more effective than placebo in maintaining remission and reducing the risk of hospitalization. Rifaximin's approval by FDA in March 2010, offers clinicians the first pharmacologic treatment for hepatic encephalopathy in more than 30 years. (Formulary. 2010; 45:210–216.)
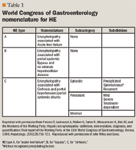

Rifaximin (pronounced riff-ax-ih-min) is an oral nonsystemic antibiotic that is FDA approved for the treatment of traveler's diarrhea caused by noninvasive strains of Escherichia coli for patients 12 years of age and older. It was designated an orphan drug by FDA in 1998 for the treatment of HE and is currently approved in 11 countries for this indication.4 In March 2010, rifaximin 550-mg tablets were granted FDA approval for the reduction in risk for overt HE recurrence in patients 18 years of age and older.5
CHEMISTRY AND PHARMACOLOGY
Currently, therapy for HE focuses on reducing production of neurotoxins derived from the gut, mainly ammonia (NH3).6 Ammonia is a by-product of intestinal flora metabolism in the colon. Its production results in a high portal concentration because it is absorbed systemically. In individuals with normal hepatic function, 90% of portal NH3 is effectively extracted by the liver.7 The NH3 extraction rate declines as hepatic dysfunction and portosystemic shunting progresses.8
Rifaximin targets the gastrointestinal neurotoxin-producing bacteria implicated in the development of HE. In theory, reduced NH3 is directly correlated with a positive effect on neurotransmission.9 This includes preventing morphologic changes of astrocytes (whose main function is to assist in providing nutrients and neurotransmitter precursors to neurons) reducing cerebral energy failure, and antagonizing GABAergic transmission.4,7,10
Rifaximin (4-deoxy-4'-methylpyrido[1',2'-1,2]imidazo-[5,4-c]-rifamycin SV) is a semi-synthetic antibiotic comparable to rifampin in its structure and mechanism of action. Unlike other rifampin derivatives, it contains an additional pyridoimidazole ring, which minimizes absorption.6 Rifaximin binds to the -subunit of bacterial DNA-dependent RNA polymerase and prevents catalysis of polymerization of deoxyribonucleotides into a DNA strand. As a result, bacterial RNA synthesis is inhibited.4,6 In vitro studies of rifaximin have demonstrated broad-spectrum coverage including Gram-positive, Gram-negative, and anaerobic bacteria as well as a limited risk of bacterial resistance.4,6 Furthermore, rifaximin does not bind to RNA polymerase in eukaryotic cells, thus human cell production is not affected. Compared with other antibiotics, rifaximin has a lower rate of fecal pathogenic eradication, so depletion of normal gastrointestinal flora is reduced.4
PHARMACOKINETICS
Rifaximin has a minimal absorption of <1% of its oral dose due to low aqueous solubility, low permeability, and P-glycoprotein efflux.4,6 This results in the majority of the drug residing in the gastrointestinal tract, with more than 96% excreted in the feces, almost entirely as unchanged drug. Approximately 0.32% of rifaximin is excreted in the urine and, of the dose excreted in the urine, 0.025% is rifaximin and <0.01% is 25-desacetylrifaximin, the only known metabolite of rifaximin. Rifaximin exhibits moderate plasma protein binding, with approximately 68% in healthy individuals and 62% in those with liver impairment.4
A phase 1 pharmacokinetic study was conducted in 14 patients who received a single oral 400-mg dose given as 2 tablets of 200 mg under both fed and fasting conditions.4 Mean areas under the plasma concentration time curve (AUC) were 18.4 ng·h/mL and 34.7 ng·h/mL, respectively. Additionally, single-dose oral administration of 550 mg was conducted in fasted and fed healthy subjects with AUCtau (area under the curve from time 0 to the end of the dosing interval) values of 11.1 ng·h/mL and 22.5 ng·h/mL, respectively. These results indicate that rifaximin absorption follows nonlinear pharmacokinetics in healthy patients.4

CLINICAL TRIALS
Rifaximin has been primarily studied for the acute treatment of HE due to its orphan drug status since 1998. Most studies compared rifaximin treatment to the current standard-the nonabsorbable disaccharide, lactulose, and the most commonly used antibiotic, neomycin.10,11 Frequently, primary end points included serum ammonia levels, asterixis, portal-systemic encephalopathy (PSE) index, West Haven criteria (Conn score), and hospital duration. Asterixis is an abnormal tremor, especially in the hands, commonly associated with HE. The PSE index was introduced in 1977 primarily for use in clinical trials to quantify the severity of HE.2 Scoring criteria of the PSE index includes arterial ammonia levels, degree of asterixis, electroencephalographic findings, and a number connection test. It is important to mention that to date it has not been prospectively validated in the United States and is subject to arbitrary results.2,7 Therefore, the West Haven criteria have recently become the most commonly used grading criteria for patients with overt HE and are recommended in HE clinical trials by the Working Party on Hepatic Encephalopathy.2,8
Rifaximin was equally effective and in some studies showed superior efficacy to lactulose and antimicrobials in patients with cirrhosis and mild-to-moderately severe HE.10,11 Response rates varied among clinical trials, but responses to rifaximin ranged from 80% to 90%, with a faster onset of action by up to 3 days.12 Commonly used end points in these studies included changes in cognition, HE grade, PSE index, blood ammonia levels, and hospitalization rates. Additionally, rifaximin had a better tolerability profile overall compared to lactulose and systemic antibiotics. Factors limiting rifaximin's use in treatment of acute HE included irregular reporting with results, lack of larger, better designed randomized controlled trials, and cost comparison to other treatments.10,11
Neff et al conducted a retrospective medical chart review of patients (n=203) who received rifaximin for maintenance therapy for HE between January 2004 and May 2009.12 Patients had a mean Model for End-stage Liver Disease (MELD) score of 11.5. MELD scores can range from 6 to 40, with higher scores indicating more severe disease.8 Patients received doses of rifaximin from 400 to 1,600 mg/day, while 54 patients also received lactulose. Overt HE developed in 26% of patients (n=38) taking rifaximin, with a mean MELD score of 19, and 39% of those patients requiring hospitalization. The authors concluded that rifaximin is effective in the management of HE in patients with cirrhosis, particularly those with a MELD score of ≤20, but that additional studies are needed to determine the association between rifaximin's efficacy and the MELD score.
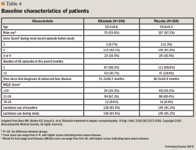
The primary end point was time to first breakthrough-episode of HE. This was further defined as the time from the first dose of the study drug in which baseline Conn score of 0 or 1 increased to 2 or more, or from a baseline Conn score of 0 to a score of 1 plus a 1-unit increase in the asterixis grade. The key secondary end point was time to first hospitalization involving HE.
Breakthrough episodes of HE occurred in 22.1% of patients in the rifaximin group compared with 45.9% of patients in the placebo group. The hazard ratio for the risk of a breakthrough episode was 0.42 in the rifaximin group, as compared with the placebo group, reflecting a relative risk reduction of 58% during the 6-month period (95% CI, 0.28 to 0.64; P<.001). This translates to 4 patients needing to be treated with rifaximin over 6 months to prevent 1 episode of HE. This risk reduction was seen across all subgroups studied. Furthermore, hospitalizations involving HE were reported in 13.6% of rifaximin-treated patients compared with 22.6% in the placebo group. It is important to mention that concomitant lactulose was administered in 91.2% of the placebo group and 91.4% of the rifaximin group.
The incidence of adverse events were similar in the rifaximin (80.0%) and placebo (79.9%.) groups. Two cases of Clostridium difficile occurred in the intervention group, although both patients had multiple risk factors and following treatment made a full recovery without discontinuing rifaximin. Death rates were also similar between the 2 groups and were mostly associated with liver disease progression. Rates of drug discontinuation were not reported.
While Bass et al presented a strong case for rifaximin, this study did have some limitations. A high concomitant use of lactulose hinders accurate data on rifaximin efficacy and safety as monotherapy for prevention.8 The authors also did not assess clinical criteria to determine the exact mechanism of action of rifaximin in preventing HE, although its efficacy is widely believed to correlate with reduction of NH3-producing bacteria. Lastly, the study population failed to assess the most severe forms of liver disease such as MELD scores >25, and only 8.6% of patients in the intervention group had MELD scores over 19.5
ADVERSE EFFECTS
According to phase 3 trial data reported by Bass et al, rifaximin is well tolerated. The most common adverse events occurring in >8% of patients include peripheral edema (15%), nausea (14.3%), diarrhea (10.7%), fatigue (12.1%), dizziness (12.9%), ascites (11.4%), headache (10%), muscle spasms (9.3%), pruritus (9.3%), and abdominal pain (8.6%).8 The same study population who completed the randomized controlled trial was also studied in a long-term, ongoing open-label maintenance study. Long-term treatment and increased exposure to rifaximin did not adversely affect its safety profile. Adverse events, discontinuation rates, and deaths were all lower during long-term therapy compared to that in the rifaximin group in the randomized controlled trial. Laboratory results were consistent to those with cirrhosis, and no hepatotoxicity or nephrotoxicity was observed.13
The development of C difficile infection may also be a small concern in patients taking rifaximin. Two patients reported C difficile infection, compared with none in the placebo group in the trial by Bass et al, although both affected patients had several risk factors predisposing them to the infection. Rifaximin is also being studied for its treatment in C difficile infections.14
DRUG INTERACTIONS
Overall, rifaximin has a minimal risk of drug interactions due to its limited systemic exposure; however, antibiotics related to rifaximin, such as rifampin, are known inducers of the P450 enzymes. An in vitro study did show a slight induction potential for CYP3A4 with rifaximin; however, the clinical relevance is thought to be minimal, although the effects on 3A4 are still unknown in patients with hepatic dysfunction.11 Two drug-drug interaction studies have been conducted with rifaximin.15,16
An open-label crossover study evaluated rifaximin's propensity to induce CYP3A4 enzymes by examining ethinyl estradiol (0.07 mg) and norgestimate (0.50 mg) in 28 healthy female patients. There is a well-documented interaction between rifampin and this oral contraceptive combination. Administration of rifaximin at a dose of 200 mg 3 times daily for 3 days did not significantly alter the pharmacokinetics of the ethinyl estradiol or norgestimate.15
Rifaximin at a dose of 200 mg 3 times daily also did not alter the pharmacokinetic profile of oral or intravenous midazolam, another medication with a known interaction with potent inducers of CYP3A4.16 Rifaximin at a dose of 550 mg 3 times daily for 7 or 14 days did result in a slightly reduced AUC for a single 2-mg dose of oral midazolam, 3.8% and 8.8%, respectively.17
Rifaximin has not been found to inhibit any major cytochrome P450 isoenzymes, and no dosage adjustments are recommended at this time when drugs are co-administered with rifaximin.17
DOSING AND ADMINISTRATION
Rifaximin is a tablet that is to be taken twice daily with or without food. Based on the recent large phase 3 clinical trial evaluating both efficacy and tolerability, dosage of rifaximin for HE will be 550 mg twice daily, although dose-ranging studies have been conducted in doses up to 2,400 mg/day with no significant adverse effects. This differs from its dose for traveler's diarrhea, which is 200 mg three times daily. Dosage adjustments are not required for patients with liver dysfunction because of the drug's localized action in the gut, although the AUC of rifaximin is higher in those with more advanced hepatic impairment. Rifaximin has not been studied in patients with renal dysfunction.11
FORMULARY CONSIDERATIONS
HE is a reversible neuropsychiatric syndrome associated with both acute and chronic liver disease that continues to be a major clinical problem. It is characterized by cognitive, psychiatric, and motor impairment. Currently more than 600,000 patients are diagnosed with cirrhosis each year, and there are up to 200,000 patients who suffer from HE. Current treatment primarily involves the acute treatment of overt episodes, with no FDA-approved agents for the prevention of HE. The current standard is the nonabsorbable disaccharides, primarily lactulose (or lactitol). They decrease the absorption of ammonia through their cathartic effects and alteration of the colonic pH. While effective in both acute treatment and prevention of overt HE, the side effects of lactulose therapy, including significant gastrointestinal effects, result in poor tolerability and frequent noncompliance. Alternative treatments include oral systemic antibiotics such as neomycin, vancomycin, and metronidazole. These agents are all associated with significant adverse events when used long term, specifically nephrotoxicity, ototoxicity, and neurotoxicity.
Rifaximin is a minimally absorbed broad-spectrum antibiotic that provides an appealing alternative for patients with HE. Rifaximin was recently approved by FDA in March 2010 for the reduction in risk of recurrence of overt HE in patients 18 years of age or older. Rifaximin has also been used as an orphan drug for the treatment of HE since 1998. Data from several trials have proven that rifaximin is equally effective or superior to both the nonabsorbable disaccharide and antibiotics in relieving the signs and symptoms of acute episodes of HE and is better tolerated.
Data from a phase 3, randomized, double-blind, placebo-controlled trial in 299 patients in remission from recurrent hepatic HE provided the strongest evidence for its swift FDA approval. The trial differed from previous studies in that it assessed the protective effects of rifaximin against breakthrough recurrences of HE versus the treatment of acute symptoms. Patients taking 550 mg of rifaximin twice daily for 6 months had a significant reduction in their risk of an episode of HE. The primary end point, the time until experiencing a breakthrough overt HE episode, was reduced by 58% over a 6-month period in the rifaximin group compared with the placebo group. Breakthrough episodes occurred in only 22.1% of patients compared with 45.9% taking placebo. Hospitalizations also had a significant decrease in patients taking rifaximin (13.6% vs 22.6%). It should be noted that 90% of patients were taking lactulose therapy concurrently, and those with severe hepatic dysfunction were not included in the trial.
Rifaximin has a favorable adverse effect profile with similar adverse event rates compared with placebo in the phase 3 trial. The most frequent adverse events seen in more than 10% of patients included peripheral edema, nausea, fatigue, dizziness, ascites, and diarrhea. Serious adverse events were also similar to placebo; however, there were 2 cases of C difficile infection in the rifaximin group compared with none in the placebo group. Continuous monitoring of adverse events is still needed to truly assess the long-term safety of rifaximin in HE, especially in those with more severe hepatic dysfunction. Rifaximin also exhibits an extremely favorable drug interaction profile. No dosage adjustments are necessary when rifaximin is coadministered with other medications. One of the major issues with rifaximin is its high acquisition cost. Hopefully, this cost will be offset by preventing hospitalizations and minimizing stays in patients with HE.
Rifaximin also has an indication for the treatment of traveler's diarrhea caused by noninvasive strands of E Coli at a dosage of 200 mg 3 times a day. In addition to its role in HE and traveler's diarrhea, rifaximin is also being studied for a variety of gastrointestinal conditions including irritable bowel syndrome and C difficile-associated diarrhea.
Rifaximin represents a novel alternative for both the treatment and the maintenance of remission for patients with hepatic encephalopathy. Despite its high cost, rifaximin should be strongly considered for formulary inclusion based on its efficacy and tolerability in patients with HE, especially given its FDA indication in the prevention of recurrent episodes.
Dr Feret is clinical associate professor at University of Rhode Island College of Pharmacy, Kingston, R.I.
Dr Barner is currently a pharmacist for Wal-Mart, North Kingston, R.I.
Disclosure Information: The authors report no financial disclosures as related to products discussed in this article.
In each issue, the "Focus on" feature reviews a newly approved or investigational drug of interest to pharmacy and therapeutics committee members. The column is coordinated by Robert A. Quercia, MS, RPh, clinical manager, Department of Pharmacy Services, Hartford Hospital, Hartford, Conn, and adjunct associate professor, University of Connecticut School of Pharmacy, Storrs, Conn; and by Craig I. Coleman, PharmD, associate professor of pharmacy practice, University of Connecticut School of Pharmacy, and director, Pharmacoeconomics and Outcomes Studies Group, Hartford Hospital.
EDITORS' NOTE: The clinical information provided in "Focus on" articles is as current as possible. Due to regularly emerging data on developmental or newly approved drug therapies, articles include information published or presented and available to the author up until the time of the manuscript submission.
REFERENCES
1. Al Sibae MR, McGuire BM. Current trends in the treatment of hepatic encephalopathy. Ther Clin Risk Manag. 2009;5:617–626.
2. Ferenci P, Lockwood A, Mullen K, Tarter R, Weissenborn K, Blei AT; and the Members of the Working Party. Hepatic encephalopathy-definition, nomenclature, diagnosis, and quantification: final report of the Working Party at the 11th World Congress of Gastroenterology, Vienna, 1998. Hepatology. 2002;35:716–721.
3. Bajaj JS. Review article: the modern management of hepatic encephalopathy. Aliment Pharmacol Ther. 2010;31:537–547.
4. Salix Pharmaceuticals Inc. Xifaxan (rifaximin) tablets, 550 mg NDA 22-554: Briefing document for the Gastrointestinal Drugs Advisory Committee Meeting 23 Feb. 2010. Available at: http://www.fda.gov/downloads/AdvisoryCommittees/CommitteesMeetingMaterials/Drugs/GastrointestinalDrugsAdvisoryCommittee/UCM201081.pdf. Accessed June 16, 2010.
5. FDA approves Xifaxan 550 mg tablets for reduction in risk of overt hepatic encephalopathy (HE) recurrence. Available at: http://www.salix.com/news/stories/20100324.aspx. Accessed June 16, 2010.
6. Huang DB, Dupont HL. Rifaximin a novel antimicrobial for enteric infections. J Infect. 2005;50:97–106.
7. Mas A. Hepatic encephalopathy: from pathophysiology to treatment. Digestion. 2006;73(suppl 1):86–93.
8. Bass NM, Mullen KD, Sanyal A, et al. Rifaximin treatment in hepatic encephalopathy. N Engl J Med. 2010;362:1071–1081.
9. Maclayton DO, Eaton-Maxwell A. Rifaximin for treatment of hepatic encephalopathy. Ann Pharmacother. 2009;43:77–84.
10. Hazell A, Butterworth R. Hepatic encephalopathy: an update of pathophysiologic mechanisms. Proc Soc Exp Biol Med. 1999;222:99–112.
11. Lawrence KR, Klee JA. Rifaximin for the treatment of hepatic encephalopathy. Pharmacotherapy. 2008; 28:1019–1032.
12. Neff G, Kemmer N, Gaddis A, Broda T, Kaiser T. Efficacy of rifaximin in maintenance of remission in patients with hepatic encephalopathy. Am J Gastroenterol. 2009;104(suppl 3):A415.
13. Sheikh M, Bass N, Sanyal A, et al. Rifaximin has a favorable long-term safety profile for maintenance of remission from overt hepatic encephalopathy. Am J Gastroenterol. 2009;104(suppl 3):A360.
14. Halsey J. Current and future treatment modalities for Clostridium difficile-associated disease. Am J Health Syst Pharm. 2008;65:705-715.
15. Trapnell CB, Connolly M, Pentikis H, Forbes WP, Bettenhausen DK. Absence of effect of oral rifaximin on the pharmacokinetics of ethinyl estradiol/norgestimate in healthy females. Ann Pharmacother. 2007;41:222–228.
16. Pentikis HS, Connolly M, Trapnell CB, Forbes WP, Bettenhausen DK. The effect of multiple-dose, oral rifaximin on the pharmacokinetics of intravenous and oral midazolam in healthy volunteers. Pharmacotherapy. 2007;27:1361–1369.
17. Xifaxan [package inset]. Morrisville, NJ: Salix Pharmaceuticals, Inc.; March 2010.
Coalition promotes important acetaminophen dosing reminders
November 18th 2014It may come as a surprise that each year Americans catch approximately 1 billion colds, and the Centers for Disease Control and Prevention estimates that as many as 20% get the flu. This cold and flu season, 7 in 10 patients will reach for an over-the-counter (OTC) medicine to treat their coughs, stuffy noses, and sniffles. It’s an important time of the year to remind patients to double check their medicine labels so they don’t double up on medicines containing acetaminophen.
Support consumer access to specialty medications through value-based insurance design
June 30th 2014The driving force behind consumer cost-sharing provisions for specialty medications is the acquisition cost and not clinical value. This appears to be true for almost all public and private health plans, says a new report from researchers at the University of Michigan Center for Value-Based Insurance Design (V-BID Center) and the National Pharmaceutical Council (NPC).
Management of antipsychotic medication polypharmacy
June 13th 2013Within our healthcare-driven society, the increase in the identification and diagnosis of mental illnesses has led to a proportional increase in the prescribing of psychotropic medications. The prevalence of mental illnesses and subsequent treatment approaches may employ monotherapy as first-line treatment, but in many cases the use of combination of therapy can occur, leading to polypharmacy.1 Polypharmacy can be defined in several ways but it generally recognized as the use of multiple medications by one patient and the most common definition is the concurrent use of five more medications. The presence of polyharmacy has the potential to contribute to non-compliance, drug-drug interactions, medication errors, adverse events, or poor quality of life.
Medical innovation improves outcomes
June 12th 2013I have been diagnosed with stage 4 cancer of the pancreas, a disease that’s long been considered not just incurable, but almost impossible to treat-a recalcitrant disease that some practitioners feel has given oncology a bad name. I was told my life would be measured in weeks.
