- Safety & Recalls
- Regulatory Updates
- Drug Coverage
- COPD
- Cardiovascular
- Obstetrics-Gynecology & Women's Health
- Ophthalmology
- Clinical Pharmacology
- Pediatrics
- Urology
- Pharmacy
- Idiopathic Pulmonary Fibrosis
- Diabetes and Endocrinology
- Allergy, Immunology, and ENT
- Musculoskeletal/Rheumatology
- Respiratory
- Psychiatry and Behavioral Health
- Dermatology
- Oncology
Roflumilast: A new phosphodiesterase 4 inhibitor for chronic obstructive pulmonary disease
Roflumilast is a novel, orally active phosphodiesterase 4 inhibitor currently under review by FDA for the treatment of patients with moderate-to-severe COPD.

Key Points
Abstract
Chronic obstructive pulmonary disease (COPD) is a common cause of morbidity and mortality in adults worldwide affecting approximately 4% to 10% of the population in the United States. Studies have shown that inflammation is a likely physiologic cause of the progressive airflow limitation seen in COPD patients. Although various anti-inflammatory agents are currently available for treating these patients, they do not reduce the likelihood of disease decline. Roflumilast is a novel, orally active phosphodiesterase 4 inhibitor currently under review by FDA for the treatment of patients with moderate-to-severe COPD. During clinical trials, roflumilast was shown to improve markers of lung function including forced expiratory volume in 1 second and forced vital capacity as well as reduce the incidence of COPD exacerbations, particularly in patients with more severe disease. These benefits are seen with monotherapy and when combined with long-acting bronchodilators including anticholinergics (tiotropium) and beta-2 agonists (salmeterol), although they appear to decrease over time. Roflumilast is generally well tolerated; the most common adverse events include nausea, vomiting, weight loss, and headache. Adverse cardiovascular events have not been shown to be of concern with roflumilast use up to 1 year in duration. (Formulary.2010;45;6-13.)
Chronic obstructive pulmonary disease (COPD) is one of the most common airway diseases worldwide.1 Studies have suggested an overall prevalence between 4% and 10%.2 COPD is a chronic condition characterized by progressive airflow limitation that is not fully reversible. This airflow restriction is commonly associated with an abnormal inflammatory response characterized by increases in neutrophils, macrophages, and CD8+ T lymphocytes.1 Phosphodiesterase (PDE) 4 is the predominant PDE expressed in these inflammatory cells.3 Current management guidelines recommend the use of short- or long-acting bronchodilators and various anti-inflammatory agents including inhaled and systemic corticosteroids.1,4,5 Systematic reviews have suggested that long-acting inhaled therapies with either beta agonists or corticosteroids reduce COPD exacerbations by 13% to 25% as compared with placebo.5 Many clinicians, however, believe that medications delivered by the oral route improve adherence to therapy although data supporting this theory are scant.6
Roflumilast (Daxas, Nycomed) is a new orally administered PDE inhibitor being reviewed for the treatment of patients with COPD. Unlike theophylline, roflumilast is a selective PDE4 inhibitor thus theoretically reducing the risk for untoward adverse events. An NDA for roflumilast was submitted by the manufacturer to FDA in July 2009 for the treatment of patients with symptomatic COPD. A Marketing Authorization Application (MAA) was submitted to the European Medicines Agency (EMEA) in May 2009 for the same indication.
PHARMACOLOGY

PHARMACOKINETICS
The main pharmacokinetic parameters of roflumilast are summarized in Table 1. With oral administration, roflumilast demonstrates rapid absorption with an absolute bioavailability of 79%.13 When given with a high-fat meal, the extent of exposure to roflumilast was not altered although the rate of absorption was modestly affected.14,15 The volume of distribution was estimated at 2.9 L/kg, suggesting pronounced tissue of the drug with high protein binding (>96%).13,16 Dose proportional and linear increases in mean peak plasma concentrations (Cmax) and area under the curve (AUC) were demonstrated for roflumilast and its active metabolite with no differences between single- and multiple-dose regimens.16 Roflumilast is extensively metabolized by cytochrome-P 450 (CYP) 3A4 and CYP1A2 to its active metabolite roflumilast N-oxide which has an in vivo potency profile similar to roflumilast.13,16 A terminal half-life of 14 to 16 hours and 19 to 21 hours has been demonstrated for roflumilast and roflumilast N-oxide, respectively, supporting the use of a once-daily dosing regimen.13,16-18 Only a small portion of roflumilast and roflumilast N-oxide is excreted unchanged in the urine (<1% for each).19 Not surprisingly, pharmacokinetic parameters of roflumilast are unchanged in patients with severe renal insufficiency.20 Patients with mild-to-moderate hepatic impairment have demonstrated modest increases in both the AUC and Cmax of roflumilast and roflumilast N-oxide, although no safety or tolerability concerns arose suggesting that dosing does not need adjustment in this population.19
CLINICAL TRIALS
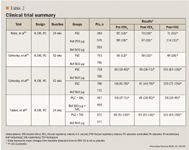
A multicenter, double-blind, randomized, placebo-controlled, phase 3 trial including 1,411 patients was undertaken in an outpatient setting.21 Study participants had moderate-to-severe COPD for at least 12 months, defined as a greater than 10 pack-year smoking history, postbronchodilator forced expiratory volume in 1 second (FEV1) of 30% to 80% of predicted, and a FEV1/forced vital capacity (FVC) ratio less than 70%. Patients first completed a 4-week, single-blind run-in period and were subsequently randomly assigned in a 2:2:1 fashion to receive roflumilast (250 µg or 500 µg) or placebo once daily for 24 weeks. The primary efficacy outcomes included changes in the postbronchodilator FEV1 and the change in the St. George's respiratory questionnaire (SGRQ) total score from baseline. Other pulmonary function tests were also evaluated throughout follow-up. Overall, patients receiving either dose of roflumilast showed significant improvement in FEV1 from baseline (P<.0006 for both doses vs placebo), starting as early as week 4. Similar improvements in postbronchodilator FVC, FEV in the first 6 seconds (FEV6), and the forced expiratory flow between 25% and 75% of the vital capacity (FEF25-75) were seen between roflumilast and placebo (P<.05 for all comparisons). Although significant improvements in health-related quality of life (HRQL) as measured by the SGRQ were seen for all the groups from baseline, there were no statistically significant differences between either roflumilast 250 µg (P=.077) or 500 µg or (P=.053) groups versus placebo. Of note, patients receiving roflumilast 500 µg experienced significantly fewer COPD exacerbations than the 250 µg or placebo groups (P value for trend test=.014). Overall, roflumilast use, at either 250 µg or 500 µg daily, resulted in significant improvements in various pulmonary function tests in patients with moderate-to-severe COPD. Even though direct comparisons between the roflumilast 250 µg and 500 µg groups were not made, it seems patients receiving the higher dose had better and earlier responses in most outcomes. In fact, a small 4-week double-blind, placebo-controlled crossover trial demonstrated that roflumilast 500 µg significantly improved the pre- and postbronchodilator FEV1 compared to placebo (P<.0001 pre-, and P=.018 postbronchodilator).22 Further clinical investigations evaluating this dose in adult COPD populations would seem warranted.
A multicenter randomized, double-blind, placebo-controlled, parallel-group study was conducted on 1,513 patients with severe, stable COPD.23 Participants had to be at least 40 years of age, current or former smokers with at least a 10 pack-year history, clinically stable COPD and a postbronchodilator FEV1 of 50% or less, FEV1:FVC ratio of 0.7 or less, or FEV1 reversibility of 5% or less. After a 4-week run-in period where patients received placebo and salbutamol (as rescue medication), patients were randomly assigned to receive placebo (n=753) or roflumilast 500 µg (n=760) once daily for 52 weeks. The primary end points included change in postbronchodilator FEV1 from baseline and the number of moderate-to-severe exacerbations per patient year. Secondary end points included change from baseline in the SGRQ total score and other pulmonary function tests. Commonly used medications prior to randomization included short-acting anticholinergics (49%), inhaled corticosteroids (28%) and inhaled long-acting beta-2 agonists (30%). Post-bronchodilator FEV1 was improved from baseline when compared with placebo (12 mL ± 11 mL for roflumilast vs -26 mL ± 11 mL for placebo, P=.0001). No significant differences in the number of moderate or severe exacerbations were seen between groups (0.857/patient/year for roflumilast vs 0.918/patient/year for placebo, P=.451). However, patients in the roflumilast group experienced 36% fewer severe COPD exacerbations compared to placebo (1.014/patient/year vs 1.588/patient/year, P=.024). Moreover, significantly fewer patients receiving roflumilast required systemic corticosteroids for their exacerbations (P=.029). Although both the roflumilast and placebo groups demonstrated improvements in the SGRQ total score from baseline, there was no significant difference between the groups (P=.651). These results extend the findings of a study of 6 months of therapy showing that improvements in pulmonary function tests are extended through 12 months.21 Improvements in HRQL and overall exacerbations were not observed, however, with differences only seen in the number of severe COPD exacerbations.
The impact of roflumilast use on clinical outcomes was further examined in 2 multicenter, randomized, double-blind, placebo-controlled trials that were pooled together.24 To be included in these studies, patients had to be former or current smokers with at least a 20 pack-year history, at least 40 years of age, and have a diagnosis of clinical COPD (confirmed by postbronchodilator FEV1/FVC of at least 70%, and a FEV1 at least 50% of predicted). Following a 4-week placebo run-in period, patients were randomly assigned to roflumilast 500 µg (n=1,537) or placebo (n=1,554) once daily for 52 weeks. The primary end points included the change in pre-bronchodilator FEV1 from baseline and the rate of exacerbations over follow-up. Secondary end points included postbronchodilator FEV1, time to death, C-reactive protein, and transition dyspnea index (TDI) focal score. In the pooled analysis, prebronchodilator FEV1 was significantly increased from baseline in the roflumilast group and decreased in the placebo group (40 ± 6 mL vs -9 ± 5 mL, respectively; P<.0001). An increase in postbronchodilator FEV1 was also observed in the roflumilast group compared with placebo (P<.0001). This difference was irrespective of concurrent long-acting beta-2 agonist use. The rate of moderate or severe exacerbations was significantly lower in the roflumilast group compared with placebo (1.14/patient/year vs 1.37/patient/year; P=.0003). A significant decrease was observed in time to death (P=.5452), or C-reactive protein levels (P=.8670) between the groups. A small improvement occurred in the focal TDI score in the roflumilast group vs the placebo group (P=.0009). The study showed significant reductions in exacerbation rate with roflumilast use in addition to the similar improvements in pre- and postbronchodilator FEV1.
Although clinical trials seem to indicate that roflumilast has beneficial clinical effects in patients with moderate-to-severe COPD, only 40% to 50% of patients were receiving other treatments for COPD. Thus, the studies evaluated to this point do not answer the question of whether roflumilast use would be beneficial when used in combination with other currently available treatment options. In this context, researchers undertook to answer this question using data from 2 multicenter, randomized, double-blind, placebo-controlled studies and pooling them in a single report.25 Patients included in both studies had moderate-to-severe COPD (defined as a postbronchodilator FEV1 40% to 70% of predicted, FEV1/FVC ratio of <70% and partial reversibility to albuterol), were older than 40 years of age, and had to be current or former smokers with a 10 pack-year history. After a 4-week run-in period, all patients were randomly assigned to receive either roflumilast 500 µg or placebo once daily for 24 weeks. In the M2-127 trial, patients received roflumilast (n=466) or placebo (n=467) in addition to daily salmeterol treatment (dose not specified), whereas the M2-128 trial gave all roflumilast (n=371) and placebo (n=372) patients daily tiotropium treatment (dose not specified). Patients in the M2-128 trial were more symptomatic due to the requirement of a chronic cough with sputum production and frequent short-acting beta-2 agonist use during the run-in period. The primary end point was the same in both studies: change in mean prebronchodilator FEV1 from baseline. Secondary end points included postbronchodilator FEV1 and FVC, TDI score, shortness of breath questionnaire (SOBQ), rate of exacerbations, and use of rescue medications. In both trials, pre- and postbronchodilator FEV1 increased significantly in the roflumilast groups versus placebo when combined with either salmeterol or tiotropium (P<.0001 for all). Roflumilast use also resulted in significant improvements in the postbronchodilator FVC versus placebo (M2-127: P=.0028; M2-128: P=.0004). Variable effects on symptomatic outcomes were seen, including slight improvements in SOBQ (M2-127: P=.5457, M2-128: P=.0051) and in the amount of rescue medications used (M2-127: P=.3689, M2-128: P=.0004) in the roflumilast groups compared with placebo. Interestingly, the per-patient-per-year rate of mild, moderate, or severe COPD exacerbations was not significantly reduced by either combining roflumilast plus salmeterol versus salmeterol alone [relative risk (RR) 0.79, 95% confidence interval (CI) 0.58-1.08) or roflumilast plus tiotropium versus tiotropium alone (RR 0.84, 95% CI 0.57-1.23). However, reductions in both the median time to a first exacerbation and proportion of patients with a moderate or severe exacerbation were reduced in both populations (P<.05 for all).
ADVERSE EVENTS
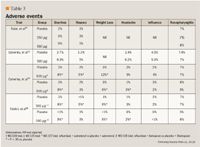
Of note, none of the clinical trials reported clinically meaningful changes in vital signs, electrocardiogram measurements or clinical laboratory parameters during treatment with roflumilast. In particular, a recent study reported no significant increases with roflumilast versus placebo in cardiovascular events including atrial fibrillation (1% vs <1%) and rhythm disturbances (2.1% vs 1.4%).24 This is noteworthy due to the reported incidence of atrial tacchyarrhythmias associated with theophylline use, especially with higher serum concentrations.26,27
DRUG INTERACTIONS
In vivo investigations show that roflumilast is metabolized by N-oxidation by cytochrome P450 3A4 and CYP1A2 isoenzymes, and CYP1A1 and CP2C19 to a lesser extent, to an inactive metabolite that is subsequently dealkylated by CYP3A4, glucuronidated, and eliminated renally.28 Thus, the potential for drug-drug interactions exists with many currently available pharmacologic agents.
Effects of other drugs on roflumilast. The impact of the potent CYP3A4 inhibitor ketoconazole on the pharmacokinetics of roflumilast and its active metabolite was evaluated in a single and repeated doses study of healthy subjects.28 The investigators studied the interaction between single-dose ketoconazole and steady-state roflumilast as well as the effect of steady-state ketoconazole on roflumilast. During steady-state roflumilast use (500 µg/d x 11 days), a single dose of ketoconazole increased the AUC by 34% with no impact on the Cmax. Repeated doses of ketoconazole resulted in a 2-fold increase in AUC and 23% increase in Cmax of roflumilast. Interestingly, despite these pharmacokinetic alterations, no change in total PDE4 inhibitory activity was seen. Thus, it appears that no dosage changes are required when potent CYP3A4 inhibitors, represented by ketoconazole, are administered concomitantly with roflumilast.
Researchers investigated the impact of the potent CYP1A2 and CYP2C19 inhibitor fluvoxamine on the pharmacokinetics of roflumilast.29 They demonstrated that coadministration of steady-state fluvoxamine increased the AUC and Cmax of roflumilast by 2.6-fold and 12%, respectively. Of note, an increase in PDE4 inhibitory activity by 1.6-fold was seen in this study. Despite these alterations, no serious or unexpected adverse events were observed, although the population was small and healthy at baseline. Interestingly, the impact of smoking, a leading cause of COPD and known to contain potent CYP1A2 inducers, was evaluated in 12 healthy smokers and 12 non-smokers given a single dose of roflumilast 500 µg.30 No significant differences in either pharmacokinetic or safety parameters of roflumilast or its primary metabolite were seen between smokers and nonsmokers, although the data were only presented in abstract form and specific data were not provided.
An evaluation of concomitant roflumilast administration with a magnesium hydroxide/aluminium hydroxide-containing antacid showed no pharmacokinetic alterations of either roflumilast or its primary metabolite.31 This suggests that alterations in GI acidity are unlikely to impact the absorption or metabolism of roflumilast and should not result in adverse effects.
Studies have also demonstrated no significant interaction between roflumilast and other drugs commonly used in patients with pulmonary disorders, including the inhaled corticosteroid budesonide or the leukotriene receptor antagonist montelukast.32,33 This included no alterations in either the pharmacokinetic disposition of the investigated drugs or their safety and tolerability profiles.
Effects of roflumilast on other drugs. Due to the prominent role CYP3A4 plays in the metabolism of roflumilast, its propensity for enzymatic inhibition was studied against the prominent CYP3A4 substrate midazolam.34 Overall, steady-state roflumilast was not found to alter the pharmacokinetic disposition of midazolam suggesting that roflumilast is unlikely to alter the clearance of drugs that are metabolized by CYP3A4. Similarly, roflumilast has been shown to have no significant impact on the pharmacokinetics or pharmacodynamics of warfarin suggesting that these agents can be administered concomitantly without requiring dosing alterations.35
DOSING AND ADMINISTRATION
Based on available clinical trials, the dosage of roflumilast most likely to be clinically used is 500 µg orally once daily.21-25 Investigations have shown that roflumilast can be taken with or without food.14,15 Pharmacokinetic data suggests that no dosage adjustments are required for patients with severe renal impairment or mild and moderate liver cirrhosis.19,20 Roflumilast pharmacokinetics does not appear to differ in either children (6-10 years) or adolescents (11-16 years) as compared with adults in doses up to 250 µg/d.36,37 However, clinical trial data in these populations are insufficient to recommend a dose at this time.
FORMULARY CONSIDERATIONS
Currently available clinical guidelines for the management of patients with COPD recommend various treatment options based on disease severity.1 In particular, patients with stage II (moderate) disease (FEV1/FVC ratio <70%, FEV1 >50% but <80%) are recommended to receive daily treatment with a long-acting bronchodilator, such as an anticholinergic agent (eg, tiotropium) or a beta-2 agonist (eg, salmeterol or formoterol). When patients are deemed to have more severe disease (FEV1/FVC ratio <70%, FEV1 >30% but <50%), or are experiencing frequent exacerbations, inhaled corticosteroids (eg, triamcinolone or fluticasone) should be added to the long-acting bronchodilator treatment.1 The benefit of these therapies is limited, however, especially in patients with more advanced disease. Moreover, morbidity and mortality trends related to COPD continue to rise suggesting that new, novel treatment strategies are required.38
Numerous clinical trials have demonstrated that the oral PDE4 inhibitor roflumilast exhibits moderate anti-inflammatory properties in patients with COPD and improves markers of pulmonary function including pre- and postbronchodilator FEV1 and FVC in patients with moderate-to-severe COPD. For example, the postbronchodilator FEV1 with roflumilast use was significantly improved from baseline when compared with placebo (39-97 mL, P<.05 for all).21-24 These reductions appear to be additive when roflumilast is combined with both salmeterol (60 mL, P<.05) and tiotropium (81 mL, P<.05) versus either agent alone.25 Studies of longer duration23,24 show less-pronounced benefits with roflumilast than shorter-duration ones suggesting that roflumilast does not attenuate the rate of pulmonary decline in COPD patients.21,25 Other anti-inflammatory agents have demonstrated similar effects. A pooled analysis of individual patient data from 7 randomized controlled trials showed that inhaled corticosteroid use significantly improved FEV1, an effect that was most pronounced during the first 6 months of therapy especially in patients who no longer smoked.39 This effect declined with continued use of steroid therapy in studies with up to 3 years of follow-up. Although longer-duration studies are required with roflumilast, a similar effect to the inhaled corticosteroids seems apparent.
Roflumilast has been shown to have benefits when added to either long-acting beta-2 agonist or anticholinergic therapy.25 There are no clinical trials, however, directly comparing its use to these agents. The oral route of administration of roflumilast suggests that patient compliance could be improved with this drug as compared with the currently available inhaled therapies, but this requires further study. Similarly, whether the addition of roflumilast to patients already treated with inhaled corticosteroids is appropriate from both an efficacy and a safety standpoint remains to be determined. Reassuringly, the known gastrointestinal adverse events related to roflumilast therapy were not main reasons for patients withdrawing from most clinical trials. Most patients could tolerate these side effects and continue therapy.
Whether roflumilast will prove to be a cost-prohibitive treatment option in patients with moderate-to-severe COPD remains to be seen. A preliminary cost-effectiveness analysis showed that roflumilast use increased the overall treatment costs of COPD, although this was almost entirely related to the cost of the drug.40
Roflumilast was shown to be cost-effective in the subset of patients with severe COPD experiencing frequent exacerbations, however, and those with relatively high healthcare utilization.
Further studies will be required if the drug is FDA approved to better determine the more broad-reaching pharmacoeconomic impact and benefits of this agent in treating COPD patients.
Dr Erica L. Baker is a senior research scientist, University of Connecticut/Hartford Hospital Evidence-Based Practice Center, Hartford, Conn.
Dr William L. Baker is an assistant clinical professor, University of Connecticut School of Pharmacy, and project manager, University of Connecticut/Hartford Hospital Evidence-Based Practice Center.
Disclosure Information: The authors report no financial disclosures as related to products discussed in this article.
In each issue, the "Focus on" feature reviews a newly approved or investigational drug of interest to pharmacy and therapeutics committee members. The column is coordinated by Robert A. Quercia, MS, RPh, clinical manager, Department of Pharmacy Services, Hartford Hospital, Hartford, Conn, and adjunct associate professor, University of Connecticut School of Pharmacy, Storrs, Conn; and by Craig I. Coleman, PharmD, associate professor of pharmacy practice, University of Connecticut School of Pharmacy, and director, Pharmacoeconomics and Outcomes Studies Group, Hartford Hospital.
EDITORS' NOTE: The clinical information provided in "Focus on" articles is as current as possible. Due to regularly emerging data on developmental or newly approved drug therapies, articles include information published or presented and available to the author up until the time of the manuscript submission.
REFERENCES
1. Global Initiative for Chronic Obstructive Lung Disease. Global strategy for the diagnosis, management, and prevention of chronic obstructive pulmonary disease (updated December 2009). Available at: http://www.goldcopd.com/. Accessed December 8, 2009.
2. Halbert RJ, Isonaka S, George D, Iqbal A. Interpreting COPD prevalence estimates: what is the true burden of disease? CHEST. 2003;123:1684-1692.
3. Essayan DM. Cyclic nucleotide phosphodiesterase (PDE) inhibitors and immunomodulation. Biochem Pharmacol. 1999;57:965-973.
4. Wilt TJ, Niewoehner D, MacDonald R, Kane RL. Management of stable chronic obstructive pulmonary disease: a systematic review for a clinical practice guideline. Ann Intern Med. 2007;147:639-653.
5. Qaseem A, Snow V, Shekelle P, et al. Clinical Efficacy Assessment Subcommittee of the American College of Physicians. Diagnosis and management of stable chronic obstructive pulmonary disease: a clinical practice guideline from the American College of Physicians. AnnInternMed. 2007;147:633-638.
6. Lipworth BJ. Phosphodiesterase-4 inhibitors for asthma and chronic obstructive pulmonary disease. Lancet. 2005;365:167-175.
7. Murciano D, Auclair MH, Pariente R, Aubier M. A randomized, controlled trial of theophylline in patients with severe chronic obstructive pulmonary disease. NEnglJMed. 1989;320:1521-1525.
8. Currie GP, Butler CA, Anderson WJ, Skinner C. Phosphodiesterase 4 inhibitors in chronic obstructive pulmonary disease: a new approach to oral treatment. BrJClinPharmacol. 2008;65:803-810.
9. Hatzelmann A, Schudt C. Anti-inflammatory and immunomodulatory potential of the novel PDE4 inhibitor roflumilast in vitrol. JPharmacol Exp Ther. 2001;297:267-279.
10. Bundschuh DS, Eltze M, Barsig J, Wollin L, Hatzelmann A, Beume R. In vivo efficacy in airway disease models of roflumilast, a novel orally active PDE4 inhibitor. JPharmacolExpTher. 2001;297:280-290.
11. Jones NA, Boswell-Smith V, Lever R, Page CP. The effect of selective phosphodiesterase isoenzyme inhibition on neutrophil function in vitro. PulmPharmacolTher. 2005;18:93-101.
12. Kwak HJ, Song JS, Heo JY, Yang SD, Nam JY, Cheon HG. Roflumilast inhibits lipopolysaccharide-induced inflammatory mediators via suppression of nuclear factor-kB, p38 mitogen-activated protein kinase, and c-Jun NH2-terminal kinase activation. JPharmacolExpTher. 2005;315:1188-1195.
13. David M, Zech K, Seiberling M, Weimar C, Bethke TD. Roflumilast, a novel, oral, selective PDE4 inhibitor, shows high absolute bioavailability. JAllergy Clin Immunol. 2004;113(suppl2):S220-S221. [Abstract]
14. Nassr N, Hauns B, Bethke M, et al. Influence of food intake on the pharmacokinetics of roflumilast and its active metabolite roflumilast-N-oxide. EurJClinPharmacol. 2002;58:S82. [Abstract]
15. Hauns B, Hermann R, Hünnemeyer A, et al. Investigation of a potential food effect on the pharmacokinetics of roflumilast, an oral, once-daily phosphodiesterase 4 inhibitor, in healthy subjects. JClinPharmacol. 2006;46:1146-1153.
16. Bethke TD, Bohmer GM, Hermann R, et al. Dose-proportional intraindividual single- and repeated-dose pharmacokinetics of roflumilast, an oral, once-daily phosphodiesterase 4 inhibitor. JClinPharmacol. 2007;47:26-36.
17. Hünnemeyer A, Hauns B, David M, et al. Pharmacokinetics and safety of roflumilast, a once-daily, oral, selective PDE4 inhibitor, and its active metabolite roflumilast N-oxide in healthy subjects. JAllergyClinImmunol. 2004;113(suppl):S222. [Abstract]
18. Hünnemeyer A, Zech K, Bethke TD, Boehmer G, Gleiter CH. Oral, once-daily roflumilast and its active N-oxide metabolite exhibit dose-proportional pharmacokinetics between 250 µg and 500 µg. CHEST. 2004;126:804S-805S. [Abstract]
19. Hermann R, Nassr N, Lahu G, et al. Steady-state pharmacokinetics of roflumilast and roflumilast N-oxide in patients with mild and moderate liver cirrhosis. ClinPharmacokinet. 2007;46:403-416.
20. Nassr N, Bethke T, Hartmann M, et al. No dose adjustment of roflumilast in patients with severe renal impairment. EurJClinPharmacol. 2002;58:S82. [Abstract]
21. Rabe KF, Bateman ED, O'Donnell D, Witte S, Brodenbröker D, Bethke TD. Roflumilast – an oral anti-inflammatory treatment for chronic obstructive pulmonary disease: a randomized controlled trial. Lancet. 2005;366:563-571.
22. Grootendorst DC, Gauw SA, Verhoosel RM, et al. Reduction in sputum neutrophil and eosinophil numbers by the PDE4 inhibitor roflumilast in patients with COPD. Thorax. 2007;62:1081-1087.
23. Calverley PM, Sanchez-Toril F, Mclvor A, Teichmann P, Brodenbröeker D, Fabbri LM. Effect of 1-year treatment with roflumilast in severe chronic obstructive pulmonary disease. AmJRespirCritCareMed. 2007;176:154-161.
24. Calverley PM, Rabe KF, Goehring UM, Kristiansen S, Fabbri LM, Martinez FJ; M2-124 and M2-125 study groups. Roflumilast in symptomatic chronic obstructive pulmonary disease: two randomized clinical trials. Lancet. 2009;374:685-694.
25. Fabri LM, Calverley PM, Izquierdo-Alonso JL, et al. M2-127 and M2-128 study groups. Roflumilast in moderate-to-severe chronic obstructive pulmonary disease treated with longacting bronchodilators: two randomised clinical trials. Lancet. 2009;374:695-703.
26. Levine JH, Michael JR, Guarnieri T. Multifocal atrial tachycardia: a toxic effect of theophylline. Lancet. 1985;1(8419):12-14.
27. Varriale P, Ramaprasad S. Aminophylline induced atrial fibrillation. PacingClinElectrophysiol. 1993;16:1953-1955.
28. Lahu G, Hünnemeyer A, von Richter O, et al. Effect of single and repeated doses of ketoconazole on the pharmacokinetics of roflumilast and roflumilast N-oxide. JClinPharmacol. 2008;48:1339-1349.
29. von Richter O, Lahu G, Hünnemeyer A, Herzog R, Zech K, Hermann R. Effect of fluvoxamine on the pharmacokinetics of roflumilast and roflumilast N-oxide. ClinPharmacokinet. 2007;46:613-622.
30. Nassr N, Hünnemeyer A, Hauns B, et al. Smoking has no influence on the pharmacokinetics of roflumilast and its active metabolite roflumilast N-oxide. EurJClinPharmacol. 2002;58:41. [Abstract]
31. Nassr N, Lahu G, Hünnemeyer A, et al. Magnesium hydroxide/aluminium hydroxide-containing antacid does not affect the pharmacokinetics of the targeted phosphodiesterase 4 inhibitor roflumilast. JClinPharmacol. 2007;47:660-666.
32. Hermann R, Siegmund W, Giessmann T, ete al. The oral, once-daily phosphodiesterase 4 inhibitor roflumilast lacks relevant pharmacokinetic interactions with inhaled budesonide. JClinPharmacol. 2007;47:1005-1013.
33. Bohmer GM, Nassr N, Wenger M, et al. The targeted oral, once-daily phosphodiesterase 4 inhibitor roflumilast and the leukotriene receptor antagonist montelukast do not exhibit significant pharmacokinetic interactions. JClinPharmacol. 2009;49:389-397.
34. Nassr N, Lahu G, von Richter O, et al. Lack of a pharmacokinetic interaction between steady-state roflumilast and single-dose midazolam in healthy subjects. BrJClinPharmacol. 2007;63:365-370.
35. Hünnemeyer A, Zech K, Terblanché J, Duursema L, Bethke TD, Venter L. Roflumilast shows no pharmacokinetic interaction with warfarin in healthy subjects. CHEST. 2004;126:804S. [Abstract]
36. Kearns S, Szefler S, Abdel-Rahman S, Lahu G, Zech K, Bethke T. Disposition of roflumilast in pediatrics. ClinPharmacolTher. 2005;77(2):P29. [Abstract]
37. Neville KA, Szefler SJ, Abdel-Rahman SM, et al. Single-dose pharmacokinetics of roflumilast in children and adolescents. JClinPharmacol. 2008;48:978-985.
38. O'Byrne PM, Gauvreau G. Phosphodiesterase-4 inhibition in COPD. Lancet. 2009;374:665-666.
39. Soriano JB, Sin DD, Zhang X, et al. A pooled analysis of FEV1 decline in COPD patients randomized to inhaled corticosteroids or placebo. CHEST. 2007;131:682-689.
40. Rutten-van Molken MP, van Nooten FE, Lindemann M, Caeser M, Calverley PM. A 1-year prospective cost-effectiveness analysis of roflumilast for the treatment of patients with severe chronic obstructive pulmonary disease. Pharmacoeconomics. 2007;25:695-711.
Employers Face Barriers With Adopting Biosimilars
March 1st 2022Despite the promise of savings billions of dollars in the United States, adoption of biosimilars has been slow. A roundtable discussion among employers highlighted some of the barriers, including formulary design and drug pricing and rebates.
