- Safety & Recalls
- Regulatory Updates
- Drug Coverage
- COPD
- Cardiovascular
- Obstetrics-Gynecology & Women's Health
- Ophthalmology
- Clinical Pharmacology
- Pediatrics
- Urology
- Pharmacy
- Idiopathic Pulmonary Fibrosis
- Diabetes and Endocrinology
- Allergy, Immunology, and ENT
- Musculoskeletal/Rheumatology
- Respiratory
- Psychiatry and Behavioral Health
- Dermatology
- Oncology
Targeting the incretin system: Novel advances in the treatment of diabetes
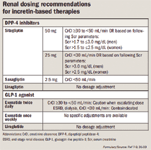
Incretin-based therapies are now the most promising therapies for type 2 diabetes.
Abstract
Type 2 diabetes mellitus remains a complex and prevalent disease in the United States today. Therapies including sulfonylureas and thiazolidinediones have been effective in its management but their limits in the long term are problematic. Incretin-based therapies are now the most promising alternatives or additions, including glucagon-like peptide-1 (GLP-1) agonists and dipeptidyl peptidase-4 (DPP-4) inhibitors. The efficacy and clinical differences of GLP-1 agonists and DPP-4 inhibitors have been thoroughly studied in recent years. This article provides details of these findings as an aid to pharmacists helping to advise patients with type 2 diabetes about their treatment options. (Formulary. 2012; 47:330–339.)

GLP-1 AGONISTS

In a 30-week, triple-blind, multicenter, placebo-controlled, parallel-group trial evaluating the change in A1c, exenatide 5 µg and 10 µg were found to be significantly better than placebo in reducing A1c values, at -0.40% and -0.78%, respectively (P<.05), among 336 patients on metformin therapy (1,500 mg/d for at least 3 months).12 Patients were randomized to exenatide 5 µg twice daily for 30 weeks, exenatide 5 µg twice daily for 4 weeks and then 10 µg twice daily thereafter, or placebo injection twice daily, in addition to their original metformin therapy. The incidence of serious adverse effects was low and consistent among all treatment arms, but the most commonly reported adverse effect was nausea, which was more than 10% in all groups, with a higher incidence in the exenatide arms. Many reports of nausea occurred at the initiation of exenatide and declined shortly thereafter. The incidence of hypoglycemia for the exenatide 5 µg, 10 µg, and placebo arms was 4.5%, 5.3%, and 5.3%, respectively. No severe hypoglycemic episodes were noted in the trial.12
In a double-blind, randomized, crossover, multicenter study, exenatide was significantly better than sitagliptin in reducing 2-hour postprandial glucose concentrations (P<.05) in 336 patients who were maintained on a stable metformin regimen.13 Patients were randomly assigned to exenatide 5 µg twice daily for 1 week, then titrated to 10 µg twice daily for 1 week or sitagliptin 100 mg daily for 2 weeks. After 2 weeks, patients crossed over to the alternate arm. Switching from exenatide to sitagliptin significantly increased postprandial glucose concentrations, while switching from sitaglitpin to exenatide further decreased postprandial values (P<.05 for both). The most common adverse effects reported in this trial were nausea (12% and 34%) and vomiting (3% and 24%), for sitagliptin and exenatide, respectively. There was only 1 reported occurrence of hypoglycemia with exenatide.13
Clinical investigations explored the use of exenatide in combination with basal insulin therapy. The rationale for such combination therapy is enhanced glycemic control, offsetting insulin-induced weight gain, and the complementing mechanisms of actions for basal and mealtime coverage. The combination of insulin and exenatide therapy was overall well tolerated. In a 30-week, randomized, parallel, placebo-controlled trial, 261 individuals with type 2 diabetes receiving at least 20 U daily of insulin glargine alone or in combination with either metformin or pioglitazone over 3 months, were randomly assigned to receive either exenatide (5 µg twice daily for 4 weeks. then increased to 10 µg twice daily) or placebo injection within 60 minutes prior to morning and evening meals.14 Insulin glargine dosages were either continued or decreased by 20% in patients with an A1c lower than 8%. At study conclusion, patients who received exenatide plus insulin glargine had a significant reduction in their A1c value from baseline (-1.7% vs -1.0%, P<.001), lower nonfasting SMBG concentrations (P<.05), and achieved better weight outcomes (-1.8 kg vs +1.0 kg, P<.05) when compared to the placebo group. There was no greater propensity for minor hypoglycemic episodes in the exenatide group (P>.05); and interestingly, major hypoglycemia occurred twice in 1 patient receiving insulin glargine alone. In this study, exenatide in addition to insulin glargine demonstrated a beneficial glycemic effect without a significant propensity for toxicity.14
In another study, the combination of exenatide and insulin glargine in metformin-treated patients significantly lowered the 6-hour postprandial excursions (P=.0036) and significantly reduced body weight (P=.0377) when compared to metformin and glargine alone.15 The incidence of hypoglycemia (<50 mg/dL) was comparable among the treatment groups (n=2 for both groups). The authors concluded that exenatide in addition to glargine and metformin is effective for postprandial glycemic control without requiring insulin reduction and without increasing the risk of hypoglycemia beyond that expected with insulin therapy.15 Currently exenatide twice daily is the only GLP-1 agonist with FDA approval for use with insulin therapy.7-9
Exenatide long-acting release. Exenatide long-acting release (LAR), commonly referred to as exenatide once weekly, received FDA approval January 2012 for adjunct therapy to diet and exercise in individuals with inadequate glycemic control.8 According to the manufacturer, however, exenatide LAR should not be recommended as first-line therapy. Similar to exenatide twice daily, the long-acting formulation has a 53% homology to naturally occurring GLP-1. It differs from the currently available exenatide twice daily due to its microsphere suspension and a biodegradable polymer that allows prolonged release.5,16 The microsphere delivery system eliminates the daily fluctuations of serum medication concentrations and the need for the dosage titration that is seen with exenatide twice daily.16 Exenatide once weekly has a median half-life of 2 weeks, achieves a serum concentration of greater than 50 pg/mL, which significantly reduces postprandial glucose readings by 2 weeks, and reaches steady state between 6 and 10 weeks.17 At the recommended dose of 2 mg every 7 days, exenatide once weekly produced stable plasma levels that were comparable to those seen with exenatide twice daily, allowing for a once-weekly dosing regimen.18 Unlike exenatide twice daily and liraglutide, which are available in prefilled pens, exenatide once weekly is a powder that must be reconstituted by the patient. Exenatide once weekly is provided in a single-dose tray that contains 1 vial of exenatide 2 mg powder, 1 prefilled diluent syringe, and 2 needles.8
In clinical trials, exenatide once weekly demonstrated efficacy in decreasing overall A1c levels, postprandial glucose levels, and fasting glucose levels, a benefit that is seen to a lesser extent with exenatide twice daily. Furthermore when compared to exenatide twice daily, exenatide once weekly demonstrated greater gastrointestinal (GI) tolerability.19 The DURATION-1 trial, a randomized, noninferiority, comparator-controlled, open-label trial, evaluated 2 mg exenatide once weekly to 10 µg exenatide twice daily for 30 weeks in 303 patients who continued their previous medications of metformin, a sulfonylurea, a TZD, or any dual therapy of those agents.19 At study conclusion, the exenatide once-weekly group experienced significantly greater reductions in A1c (-1.9% vs -1.5%; P=.0023), significantly greater achievements of target A1c of 7% or less (P=.0039), and a significantly greater propensity to decrease fasting plasma glucose values (P<.0001) when compared to individuals treated with exenatide twice daily. The most common adverse effect reported in this trial was nausea, which was significantly higher in patients treated with exenatide twice daily. The incidence of hypoglycemia was similar among treatment arms, with the majority of cases associated with concomitant sulfonylurea use.19
The DURATION-2 superiority trial of double-blind, double-dummy design evaluated the change in A1c from baseline to week 26 in 514 randomized patients maintaining stable doses of metformin to either exenatide 2 mg once weekly, sitagliptin 100 mg daily, or pioglitazone 45 mg daily.20 The observed A1c reduction was greater with exenatide once weekly compared to sitagliptin and pioglitazone, at -1.5%, -0.9%, and -1.2%, respectively. Statistical significance was observed in the A1c reduction of -0.6% between the exenatide and sitagliptin treatment groups only (P<.0001).20
In the DURATION-2 extension, a 26-week, open-label, efficacy study, patients either continued their exenatide once-weekly therapy or were switched from their previous oral study medications (sitagliptin or pioglitazone) to exenatide once weekly.21 At study conclusion, the change in A1c between weeks 26 and 52 was -0.31%, -0.10%, and 0.06% for the sitagliptin-switched group, the pioglitazone-switched group, and the exenatide-continued group, respectively. Overall, at week 52, individuals who continued exenatide once weekly and those switched from pioglitazone maintained the initial improvement in A1c, while the sitagliptin-switched group experienced a significant reduction in A1c (-0.3%; P=.0010) from the initial baseline value.21
The DURATION-3, open-label, parallel study, randomly assigned 456 patients to either exenatide 2 mg once weekly or insulin glargine (starting dose 10 U/d, adjusted thereafter according to the Initiate Insulin by Aggressive Titration and Education [INITIATE] algorithm) for 26 weeks to compare the change in A1c between these therapies.22 At study conclusion, exenatide was significantly better than insulin at reducing A1c, at -1.5 and -1.3%, respectively, equating to a treatment difference of -0.16% (P=.017). Similarly, weight reductions significantly favored exenatide therapy (P<.0001). As expected, based on the mechanisms of action, exenatide produced greater postprandial reductions than insulin glargine. A greater proportional of patients receiving exenatide experienced A1c reduction and weight loss compared to the insulin-treated group. Limitations to this study included the open-label design, possible lack of supervision to the INITIATE protocol, and lack of adherence monitoring.22
Liraglutide. Liraglutide, in contrast to the animal-derived origin of exenatide, is designed by recombinant DNA technology.23 Liraglutide received FDA approval in January 2010 as an adjunct to diet and exercise to improve glycemic control in patients with type 2 diabetes; however, the manufacturer does not recommend it as a first-line option.9 Like exenatide, liraglutide has been studied in combination with metformin, sulfonylureas, and TZDs, but there are currently no published studies of liraglutide with basal insulin. With 97% homology to GLP-1, liraglutide is more structurally similar to endogenous GLP-1 than exenatide.5 Liraglutide is designed to resist DPP-4 enzymatic degradation via 2 mechanisms: amino acid substitutions at positions 26 and 34 and slower dissociation of the molecule through the presence of multiple hydrophobic residues. Together, these result in a prolonged half-life of 12 to 14 hours, which allows for once-daily subcutaneous dosing.5 Its extended duration of action results in a more robust effect on fasting glucose concentrations than exenatide, presumably due to its sustained serum GLP-1 exposure.24 The recommended initial dose, although not a therapeutic dose, of liragludtide is 0.6 mg daily for 1 week to enahance GI tolerability..9,23 Weekly the dose should be titrated by 0.6-mg intervals to the recommended target doses of 1.2 mg or 1.8 mg daily. Liraglutide does not undergo any appreciable metabolism, and is excreted in the urine and feces. No dosage adjustments are necessary for renal or hepatic impairment.9
The effectiveness, determined by A1c reduction, of liraglutide 1.2 mg daily and 1.8 mg daily was compared to glimepiride 8 mg daily in the 52-week LEAD-3 double-blind, double-dummy, active-control, parallel-group study among 746 patients.25 At study completion, patients taking liraglutide 1.2 mg and 1.8 mg had significantly greater reductions in their A1c when compared to glimepiride, at -0.84%, -1.14%, and -0.51%, respectively. In terms of adverse effects, the incidence of overall hypoglycemia with both liraglutide groups was significantly lower than that occurring with glimepiride (P<.0001); however, nausea was reported to a greater degree in liraglutide patients (P<.0001 for both doses).25
The LEAD-6 trial randomly assigned 464 patients uncontrolled on metformin and/or a sulfonylurea to either liraglutide 0.6 mg titrated up to 1.8 mg after 2 weeks or exenatide 5 µg twice daily, titrated up to 10 µg twice daily after 4 weeks, in addition to their previous antiglycemic medications.24 This study demonstrated significance favoring liraglutide's ability to reduce A1c values (-1.12% vs -0.79%, respectively; P<.0001) and achieve a goal A1c of lower than 7% when compared to exenatide (P=.0015). Exenatide, however, demonstrated a greater ability to reduce postprandial glucose concentrations over liraglutide (P<.05).24
The LEAD-6 extension trial was comprised of 200 liraglutide-treated and 186 exenatide-treated patients.26 Individuals either continued their current liraglutide therapy or were switched from exenatide to liraglutide 1.8 mg daily. At the 14-week study conclusion, patients who were switched to liraglutide demonstrated further reductions in A1c values of approximately -0.32% (P<.0001). Patients who had continued liraglutide demonstrated a further but insignificant reduction of -0.06% (P=.1222).26
An open-label, 52-week, parallel-group trial comparing the change in A1c reduction between liraglutide 1.2 mg daily, liraglutide 1.8 mg daily, or sitagliptin 100 mg daily in 665 patients taking metformin (>1,500 mg/d) with inadequately controlled type 2 diabetes favored liraglutide therapy.27 At study conclusion, patients who were treated with liraglutide 1.2 mg and 1.8 mg exhibited greater A1c reductions, (-1.29% and -1.51%, respectively), than that observed with sitagliptin (-0.88%), resulting in a significant mean treatment difference favoring liraglutide (P<.0001 for both doses). A dose-dependent incidence of liraglutide-induced nausea was observed. The incidence of hypoglycemia reported as more than 5% was relatively consistent among treatment arms.27
Although both short-acting and long-acting GLP-1 agonists are effective as glycemic-lowering agents, GLP-1 agonists with durations of actions longer than 24 hours appear to be affiliated with greater A1c and fasting plasma glucose lowering, although decreases in postprandial glucose concentrations are observed.18 They also appear to be associated with larger increases in fasting insulin as well as larger decreases in fasting glucagon concentrations. Evidence further suggests long-acting GLP-1 agonists are associated with a greater ability to slow gastric emptying, less treatment-related nausea and hypoglycemia, and provide equivalent weight reductions when compared to their shorter-duration formulations.18
When selecting between a short-acting or long-acting GLP-1 agonist practitioners should consider patient preference, the degree of fasting and/or postprandial glucose lowering desired, concomitant therapies and conditions, side effects including the risk of and concerns with hypoglycemia, insurance coverage, and/or cost.
DPP-4 INHIBITORS
DPP-4 inhibitors, prescribed alone or in concert with other antidiabetic therapies, offer a new therapeutic option for the treatment of type 2 diabetes through their ability to increase endogenous GLP-1 levels 3- to 4-fold within 5 minutes of oral administration.28,29 Weight neutrality and low propensity for severe hypoglycemia have made them an attractive therapy for patients with type 2 diabetes.28,30 When these are used in combination therapy with insulin or insulin secretagogues, however, the risk of hypoglycemia can be expounded; thus an insulin or insulin secretagogue dosage reduction may be required.31-33 Although DPP-4 inhibitors share the same mechanism of action for glycemic lowering their structural heterogenicity creates several pharmacologic differences that are discussed herein as well as found in Table 4.11,31-35
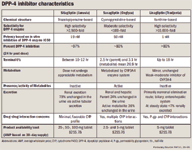
Sitagliptin. Sitagliptin, approved by FDA in October 2006, was the first DPP-4 inhibitor released into the US market for the treatment of type 2 diabetes as either monotherapy or adjunctive therapy with metformin, a sulfonylurea, or a TZD.31 Sitagliptin, which has an elimination half-life of 12.4 hours, is highly selective for the DPP-4 enzyme and inhibits approximately 80% of DPP-4 enzymatic activity when administered at the recommended dose of 100 mg daily.31,35 Sitagliptin's metabolism is inappreciable-79% of the parent agent is excreted, primarily unchanged, by the kidneys-and thus requires multiple dosage recommendations based on the degree of renal dysfunction (Table 3).31,34,35 Sitagliptin does not inhibit cytochrome (CYP) 450 isozymes or induce the CYP 3A4 system and thus has a favorable profile with regard to its interaction with the CYP system.35
A 24-week multicenter, randomized, double-blind, placebo-controlled, parallel-group trial evaluating the efficacy of sitagliptin in 206 elderly patients (≥65 years) with poorly controlled lifestyle-managed type 2 diabetes found a significant reduction in A1c favoring sitagliptin therapy (-0.5% sitagliptin vs +0.2% placebo; P<.001), with a between group difference of -0.7%.36 There were no reports of hypoglycemia in either treatment group. The authors concluded that sitagliptin effectively improved glycemic parameters and is well tolerated among the elderly population.36
A 24-week, multicenter, randomized, placebo-controlled, parallel-group study was conducted to determine the change in A1c at study conclusion when comparing sitagliptin 100 mg to placebo in 641 adults (≥21 years) taking stable insulin therapy (defined as ≥15 U/d intermediate- or long-acting or premixed insulin) with or without metformin (>1,500 mg/d).37 At study conclusion, there was a significant difference in A1c value in the sitagliptin group as demonstrated by a 0.6% reduction from baseline (P<.001) compared to no change in the placebo group. No statistical difference was observed between individuals based on the presence or absence of metformin or insulin type. The incidence of hypoglycemia was significantly greater in the sitagliptin group when compared to the placebo group (16% vs 8%, respectively; P=.003). Other adverse effects experienced with a prevalence of 2% or greater in both groups were influenza, nasopharyngitis, upper respiratory tract infections (URTI), urinary tract infections (UTI), and headache. All forenamed adverse effects were higher in the sitagliptin group with the exception of URTIs. The authors concluded that the addition of sitagliptin, at the recommended dose, to ongoing insulin therapy, with or without concomitant metformin therapy, improved overall glycemic control, and is generally well tolerated, even in terms of hypoglycemia.37
A 12-month, international, multicenter, randomized, double-blind study compared the effects of adding sitagliptin 100 mg daily or metformin 850 mg twice daily in 137 adults with uncontrolled diabetes despite therapy with pioglitazone 30 mg daily and lifestyle modifications.38 Study parameters included effect on weight, glycemic control, beta-cell function, insulin resistance, and inflammatory state. Patients in both groups experienced a significant reduction in A1c compared to baseline (P<.01), with no statistically or clinically significant difference between the 2 groups. The mean change in A1c at 12 months was -1.1% and -1% for the sitagliptin and metformin groups, respectively. Additionally, both medications resulted in a significant improvement in fasting plasma glucose and postprandial glucose at 12 months (P<.01 compared to baseline for both groups). This study showed a statistically similar reduction in A1c, fasting plasma glucose, and postprandial glucose between sitagliptin or metformin when added to pioglitazone therapy.38
Saxagliptin. Saxagliptin, a potent, reversible, selective DPP-4 inhibitor specifically designed for extended enzymatic inhibition, received FDA approval July 2009 as adjunct therapy to diet and exercise in adults with type 2 diabetes.32,39 The parent compound of saxagliptin and its active metabolite inhibit 97% of DPP-4 enzymatic activity for 24 hours, allowing for once-daily dosing, with an elimination half-life of 2.5 and 3.1 hours, respectively.34,35 Although saxagliptin 10 mg daily was evaluated in clinical trials, the recommended dose for saxagliptin is 2.5 mg to 5 mg daily, with the lower dose recommended for patients with a CrCl of 50 mL/min or less and/or taking a strong cytochrome P450 3A4 and 3A5 (CYP3A4/5) inhibitor.32,39 Saxagliptin is primarily metabolized by CYP3A4/5; enzymes. Strong CYP3A4/5 inhibitors and inducers alter saxagliptin's pharmacokinetic profile resulting in various drug-drug interactions.39 Some of the drug-drug interactions of greatest significance are with fluconazole (azoles), omperazole, macrolides, and HIV antiretrovirals.32,39 Saxagliptin is also excreted via hepatic pathways; although studies have found that individuals with mild, moderate, and severe hepatic impairment experienced a less than 2-fold higher medication exposure, indicating that dose reductions for hepatic insufficiency are not warranted.32,35
A multicenter, randomized, parallel-group, double-blind, placebo-controlled study evaluating multiple doses of saxagliptin (2.5 mg, 5 mg, or 10 mg daily) to placebo in 401 treatment-naïve adults (18-77 years) with poor lifestyle-controlled diabetes demonstrated significant improvement in change in A1c (primary endpoint) with all saxagliptin doses (P<.0001) when compared to placebo.28 The adjusted mean change in A1c values for saxagliptin 2.5 mg, 5 mg, 10 mg, and placebo was -0.43%, -0.46%, -0.54%, and +0.19%, respectively. The most commonly reported adverse effects (>5% incidence) in the saxagliptin groups were URTI, headache, UTI, nasopharyngitis, sinusitis, and dermatologic conditions. The adverse effects listed herein with the exception of UTI and sinusitis also occurred at a more than 5% incidence in the placebo group, with URTIs occurring more in the placebo group than in any saxagliptin group. The reported incidence of hypoglycemia (all unconfirmed) for saxagliptin 2.5 mg, 5 mg, and 10 mg, and placebo was 2.9%, 5%, 8%, and 6%, respectively. The authors concluded that clinical improvement in glycemic parameters favored treatment with saxagliptin therapy over placebo and saxagliptin was well tolerated in patients currently on lifestyle modifications for diabetes control.28
An 18-week, multicenter, randomized, double-blind, active-controlled, parallel-group study comparing saxagliptin 5 mg daily to sitagliptin 100 mg daily in 801 individuals with type 2 diabetes who had inadequate glycemic control on monotherapy metformin (1,500 mg-3,000 mg/d) demonstrated noninferiority between the 2 DPP-4 inhibitors.40 Adding saxagliptin or sitagliptin to a stable metformin dose reduced the A1c (primary end point) by 0.52% and 0.62%, respectively. The difference in A1c reduction between the treatment groups was 0.09%. For the entire study, the overall incidence of reported adverse effects was 47.1% and 47.2%, for saxagliptin and sitagliptin, respectively, with infections and infestations being the most common, occurring equally in 25.1% of the patients in each treatment arm. Frequently reported infections (>5%) were influenza and UTIs. Additionally, hypoglycemia occurred at similar rates of 3.2% and 2.8% for saxagliptin and sitagliptin therapy, respectively. This study proved that both saxagliptin and sitagliptin similarly and effectively improved glycemic control and were generally well tolerated.40
A 24-week, multicenter, randomized, double-blind, placebo-controlled study evaluating the efficacy of saxagliptin (2.5 or 5 mg daily) or placebo as add-on therapy to a TZD in 565 inadequately controlled individuals with type 2 diabetes demonstrated glycemic improvements favoring saxagliptin.41 TZD regimens (rosiglitazone 4 mg or 8 mg daily; pioglitazone 30 mg or 45 mg daily) remained constant during the study. Significance was observed for the primary end point of change in baseline A1c concentration, for both the 2.5-mg (P=.0007) and 5-mg (P<.0001) saxagliptin arms when compared to placebo at week 24. Adjusted mean change in A1c from baseline to study conclusion was -0.66%, -0.94%, and -0.30%, respectively, for saxagliptin 2.5 mg, 5 mg, and placebo. The adverse effects with a reported greater than 5% incidence among saxagliptin-treated patients were (in descending order): URTI, UTI, nasopharyngitis, arthralgia, headache, dizziness, and peripheral edema. The occurrence of hypoglycemia was similar between groups with a reported incidence of 4.1%, 2.7%, and 3.8% for saxagliptin 2.5 mg, 5 mg, and placebo, respectively. This study showed treatment with saxagliptin statistically improves glycemic parameters when added to TZD therapy.41
Linagliptin. Linagliptin, a reversible, highly selective, novel, xanthine-based, dose-dependent DPP-4 inhibitor was approved by FDA in April/May 2011 for use in lifestyle-treated patients with type 2 diabetes as either monotherapy or combination therapy.33,42 Similar to the other DPP-4 inhibitors discussed previously, linagliptin is dosed once daily for the treatment of type 2 diabetes. At the recommended dose of 5 mg daily, linagliptin reduces A1c by approximately 0.4% and inhibits 80% or more of DPP-4 enzymatic activity in a dose-dependent manner throughout a 24-hour period.33,42 Additionally, in vitro assays of the mean 50% maximum inhibitory concentration (IC50) suggest that linagliptin is more potent than sitagliptin and saxagliptin, respectively.2 Like saxaglitpin and sitagliptin, linagliptin has a low propensity for hypoglycemia and was generally well tolerated by patients in clinical trials.42 Linagliptin is a weak to moderate inhibitor of CYP 3A4, but does not inhibit the other CYP isoenzymes.35 Although no dosage adjustments are indicated for potential drug interactions, it is recommended that alternative treatment be used when linagliptin is administered with a permeability glycoprotein (P-gp) or CYP 3A4 inducer, as these can reduce linagliptin efficacy.33 Differing from other FDA-approved DPP-4 inhibitors, linagliptin has a favorable pharmacokinetic profile secondary to its predominantly nonrenal elimination route, which bypasses the need for dose reductions in renal impairment.33 Linagliptin would therefore be the preferred agent in patients with renal insufficiency.
A 24-week, phase 3, multicenter, randomized, double-blind, placebo-controlled, parallel-group study investigating the efficacy and safety of linagliptin 5 mg daily versus placebo as add-on therapy to metformin (stable dose >1,500 mg/d for >12 weeks) in 701 individuals with uncontrolled type 2 diabetes found that linagliptin therapy resulted in significant reductions in mean A1c concentrations.43 At the end of study, the reduction in A1c was -0.49% for linagliptin versus an increase by 0.15% for placebo, which was associated with a treatment difference of -0.64% (P<.0001). Similarly, a significant percentage of patients in the linagliptin group achieved an A1c of less than 7% compared to the placebo group, 26% vs 9%, respectively (P=.0001). Furthermore, although not statistically significant, the biomarkers of insulin sensitivity and beta-cell function improved secondary to linagliptin treatment. Regarding linagliptin's safety profile, the most common and more frequently occurring side effects (compared to placebo) were influenza, URTIs, and diarrhea, although not statistically different between treatment groups.43
Another 24-week, multicenter, randomized, double-blind, placebo-controlled study evaluated the efficacy and safety of initial combination therapy with linagliptin 5 mg daily or placebo with pioglitazone 30 mg daily in 389 patients with inadequately controlled type 2 diabetes.2 The results of the primary end point, change in A1c from baseline to study completion, favored linagliptin, with an A1c reduction of -1.06% and -0.56% for linagliptin and placebo, respectively, and an adjusted mean A1c of -0.51% (P<.0001). Additionally, at the end of the study, 42.9% and 30.5% of the linagliptin- and placebo-treated patients, respectively, achieved an A1c of less than 7%. Furthermore, although not statistically significant, the biomarkers of insulin sensitivity and beta-cell function improved secondary to linagliptin treatment. Regarding linagliptin's safety profile, the most common and more frequently occurring side effects (compared to placebo) were GI disorders (gastritis, nausea, vomiting), facial and local edema, headache and hypoesthesia, although none was statistically or clinically significant.2
Similar to GLP-1 agonists, all DPP-4 inhibitors are efficacious with glucose lowering. Selection of a specific DPP-4 inhibitor may be based on renal function, drug-drug interactions, insurance coverage, and/or cost.
DISCUSSION
Incretin-based therapies exhibit an extremely unique mode of glucose lowering that is multifactorial. No single antidiabetic agent currently available targets the multitude of dysfunctions seen in type 2 diabetes like the incretin-based therapies, particularly the GLP-1 agonists. Practitioners should be aware that there are no therapeutic alternatives for incretin-based therapies, as this class exhibits numerous characteristics that are not found in other antiglycemic therapies (Table 1).4
Given their numerous advantages, healthcare providers should consider the use of these agents more judiciously in patients with poorly-controlled diabetes, especially individuals who present with concerns of postprandial hyperglycemia and obesity. Evidence suggests the addition of an incretin-based therapy, specifically a GLP-1 agonist, to a standard diabetes regimen in patients with longstanding disease may be beneficial in reducing overall disease burden and medical costs, which can subsequently enhance the patient's quality of life.
All incretin-based therapies have demonstrated efficacy in clinical trials, yet their place in therapy varies based on desired glycemic outcomes, the need for weight reduction, and their safety profile. Furthermore, the place in therapy differs between the AACE and American Diabetes Association (ADA) guidelines. Although the 2009 AACE algorithm acknowledges the use of both GLP-1 agonists and DPP-4 inhibitors as first-line options, the 2008 ADA algorithm endorses the use of GLP-1 agonists in tier 2, consisting of less-validated diabetes therapies, and only acknowledges the use of DPP-4 inhibitors in "special populations," specifically those with concerns of hypoglycemia or weight gain.3,44 In our clinical practice we prefer the use of a GLP-1 agonist over a DPP-4 inhibitor secondary to their weight reduction properties. We have thus used GLP-1 agonists as first-line therapy, monotherapy, and as add-on therapy to oral medications and insulin.
CONCLUSION
Incretin-based therapies are a new class of medications that represent innovative and useful options for patients with type 2 diabetes mellitus, with evidence of improving pertinent biomarkers in glycemic control. Definitely as the endemic of diabetes continues to grow, we will witness the widespread use of these agents, as their adverse events permits. With their increased use and further clinical investigations, more will be revealed regarding the long-term implications of incretin-based therapies and best practice recommendations can be developed regarding their clinical utility and strategies for abating undesirable events.
Given the increased prevalence of type 2 diabetes and the polypharmacy approaches to treating diabetes, the pharmacist as part of the interprofessional medical team can successfully impact the selection and monitoring requirements of incretin based therapies as well as assist with the overall medication therapy management.
Dr Williams is assistant professor of pharmacy practice, Butler University College of Pharmacy and Health Sciences, Indianapolis, IN. Dr Prasad-Reddy is assistant professor of clinical and administrative sciences, Sullivan University College of Pharmacy, clinical pharmacist, University of Louisville Hospital, Internal Medicine Department, Louisville, KY.
Acknowledgements: Tracy Costello, PharmD, assistant professor of pharmacy practice, Butler University; Yolanda M. Hardy, PharmD, associate professor Chicago State University; Barbara Howes, associate professor of library science; Leslie Tindal, PharmD, RPh, pharmacy manager, Walmart.
Disclosure Information: The authors report no financial disclosures as related to products discussed in this article.
REFERENCES
1. Centers for Disease Control and Prevention. National Diabetes Fact Sheet 2011. http://www.cdc.gov/diabetes/pubs/pdf/ndfs_2011.pdf. Updated May 2011. Accessed May 2, 2012.
2. Gomis R, Espadero RM, Jones R, Woerle HJ, Dugi KA. Efficacy and safety of initial combination therapy with linagliptin and pioglitazone in patients with inadequately controlled type 2 diabetes: A randomized, double-blind, placebo-controlled study. Diabetes Obes Metab. 2011;13:653–661.
3. Rodbard HW, Davidson JA, Garber AJ, et al. AACE/ACE Consensus Statement. Statement by an American Association of Clinical Endocrinologists/American College of Endocrinology Consensus Panel on Type 2 Diabetes Mellitus: An algorithm for glycemic control. http://www.aace.com/files/glycemic-control-algorithm.pdf. Accessed May 2, 2012.
4. Davidson JA, Parente EB, Gross JL. Incretin mimetics and dipeptidyl peptidase-4 inhibitors: Innovative treatment therapies for type 2 diabetes. Arq Bras Endocrinol Metabol. 2008;52:1039–1049.
5. Kruger DF, Bode B, Spollett GR. Understanding GLP-1 analogs and enhancing patient success. Diabetes Educ. 2010;36(suppl 3):44S-72S.
6. Neumiller JJ. Differential chemistry (structure), mechanism of action, and pharmacology of GLP-1 receptor agonists and DPP-4 inhibitors J Am Pharm Assoc. 2009;49(suppl 1):S16–S29.
7. Byetta (exenatide). http://www.accessdata.fda.gov/drugsatfda_docs/label/2011/021773s029s030lbl.pdf.
8. Bydureon (exenatide extended-release). http://www.accessdata.fda.gov/drugsatfda_docs/label/2012/022200Orig1s000lbledt.pdf. Accessed May 5, 2012.
9. Victoza (liraglutide). http://www.accessdata.fda.gov/drugsatfda_docs/label/2012/022341s007s009s013lbl.pdf. Accessed May 5, 2012.
10. Pinkney J, Fox T, Ranganath L. Selecting GLP-1 agonists in the management of type 2 diabetes: Differential pharmacology and therapeutic benefits of liraglutide and exenatide. Ther Clin Risk Manag. 2010;6:401–411.
11. Red Book Online via Micromedex. http://www.redbook.com/redbook/index.html. Accessed May 2, 2012
12. Defronzo RA, Ratner RE, Han J, et al. Effects of exenatide (exendin-4) on glycemic control and weight over 30 weeks in metformin-treated patients with type 2 diabetes. Diabetes Care. 2005;28:1092–1100.
13. DeFronzo RA, Okerson T, Viswanathan P, et al. Effects of exenatide versus sitagliptin on postprandial glucose, insulin and glucagon secretion, gastric emptying, and caloric intake: A randomized, cross-over study. Curr Med Res Opin. 2008;24:2943–2952.
14. Buse JB, Bergenstal RM, Glass LC, et al. Use of twice-daily exenatide in Basal insulin-treated patients with type 2 diabetes: A randomized, controlled trial. Ann Intern Med. 2011;154:103–112.
15. Arnolds S, Dellweg S, Clair J, et al. Further improvement in postprandial glucose control with addition of exenatide or sitagliptin to combination therapy with insulin glargine and metformin: A proof-of-concept study. Diabetes Care. 2010;33:1509–1515.
16. Aroda VR, DeYoung MB. Clinical implications of exenatide as a twice-daily or once-weekly therapy for type 2 diabetes. Postgrad Med. 2011;123:228–238.
17. Tzefos M, Harris K, Brackett A. Clinical efficacy and safety of once-weekly glucagon-like peptide-1 agonists in development for treatment of type 2 diabetes mellitus in adults. Ann Pharmacother. 2012;46:68–78.
18. Madsbad S, Kielgast U, Asmar M, et al. An overview of once-weekly glucagon-like peptide-1 receptor agonists--Available efficacy and safety data and perspectives for the future. Diabetes Obes Metab. 2011;13:394–407.
19. Drucker DJ, Buse JB, Taylor K, et al; DURATION-1 Study Group. Exenatide once weekly versus twice daily for the treatment of type 2 diabetes: A randomised, open-label, non-inferiority study. Lancet. 2008;372:1240–1250.
20. Bergenstal RM, Wysham C, Macconell L, et al; DURATION-2 Study Group. Efficacy and safety of exenatide once weekly versus sitagliptin or pioglitazone as an adjunct to metformin for treatment of type 2 diabetes (DURATION-2): A randomised trial. Lancet. 2010:376:431–439.
21. Wysham C, Bergenstal R, Malloy J, et al. DURATION-2: Efficacy and safety of switching from maximum daily sitagliptin or pioglitazone to once-weekly exenatide. Diabet Med. 2011;28:705–714.
22. Diamant M, Van Gaal L, Stranks S, et al. Once weekly exenatide compared with insulin glargine titrated to target in patients with type 2 diabetes (DURATION-3): An open-label randomised trial. Lancet. 2010;375:2234–2243.
23. Davies MJ, Kela R, Khunti K. Liraglutide - Overview of the preclinical and clinical data and its role in the treatment of type 2 diabetes. Diabetes Obes Metab. 2011;13:207–220.
24. Buse JB, Rosenstock J, Sesti G, et al. Liraglutide once a day versus exenatide twice a day for type 2 diabetes: A 26-week randomised, parallel-group, multinational, open-label trial (LEAD-6). Lancet. 2009;374:39–47.
25. Garber A, Henry R, Ratner R, et al; LEAD-3 (Mono) Study Group. Liraglutide versus glimepiride monotherapy for type 2 diabetes (LEAD-3 Mono): A randomised, 52-week, phase III, double-blind, parallel-treatment trial. Lancet. 2009;373:473–481.
26. Buse JB, Sesti G, Schmidt WE, et al; Liraglutide Effect Action in Diabetes-6 Study Group. Switching to once-daily liraglutide from twice-daily exenatide further improves glycemic control in patients with type 2 diabetes using oral agents. Diabetes Care. 2010;33:1300–1303.
27. Pratley R, Nauck M, Bailey T, et al; 1860-LIRA-DPP-4 Study Group. One year of liraglutide treatment offers sustained and more effective glycaemic control and weight reduction compared with sitagliptin, both in.0. combination with metformin, in patients with type 2 diabetes: A randomized, parallel-group, open-label trial. Int J Clin Pract. 2011;65:397–407.
28. Rosenstock J, Aguilar-Salinas C, Klein E, et al; CV181-011 Study Investigators. Effect of saxagliptin monotherapy in treatment-naïve patients with type 2 diabetes. Curr Med Res Opin. 2009;25:2401–2411.
29. Deacon CF. Dipeptidyl peptidase-4 inhibitors in the treatment of type 2 diabetes: A comparative review. Diabetes Obes Metab. 2011;13:7–18.
30. Kirby M, Yu DM, O'Connor S, Gorrell MD. Inhibitor selectivity in the clinical application of dipeptidyl peptidase-4 inhibition. Clin Sci (Lond). 2010;118:31–41.
31. Januvia (sitagliptin). http://www.accessdata.fda.gov/drugsatfda_docs/label/2012/021995s023lbl.pdf. Accessed May 5, 2012.
32. Onglyza (saxagliptin). http://www.accessdata.fda.gov/drugsatfda_docs/label/2011/022350s004lbl.pdf. Accessed May 5, 2012.
33. Tradjenta (Linagliptin). http://www.accessdata.fda.gov/drugsatfda_docs/label/2011/201280lbl.pdf. Accessed May 5, 2012.
34. Gerich J. DPP-4 inhibitors: What may be the clinical differentiators? Diabetes Res Clin Pract. 2010;90:131–140.
35. Baetta R, Corsini A. Pharmacology of dipeptidyl peptidase-4 inhibitors: Similarities and differences. Drugs 2011;71:1441-1467.
36. Barzilai N, Guo H, Mahoney EM, et al. Efficacy and tolerability of sitagliptin monotherapy in elderly patients with type 2 diabetes: A randomized, double-blind, placebo-controlled trial. Curr Med Res Opin. 2011;27:1049–1058.
37. Vilsbøll T, Rosenstock J, Yki-Järvinen H, et al. Efficacy and safety of sitagliptin when added to insulin therapy in patients with type 2 diabetes. Diabetes Obes Metab. 2010;12:167–177.
38. Derosa G, Maffioli P, Salvadeo SA, et al. Effects of sitagliptin or metformin added to pioglitazone monotherapy in poorly controlled type 2 diabetes mellitus patients. Metabolism. 2010;59:887–895.
39. Sharma MD. Role of saxagliptin as monotherapy or adjunct therapy in the treatment of type 2 diabetes. Ther Clin Risk Manag. 2010;6:233–237.
40. Scheen AJ, Charpentier G, Östgren CJ, Hellqvist A, Gause-Nilsson I. Efficacy and safety of saxagliptin in combination with metformin compared with sitagliptin in combination with metformin in adults patients with type 2 diabetes mellitus. Diabetes Metab Res Rev. 2010;26:540–549.
41. Hollander P, Li J, Allen E, Chen R; CV181-013 Investigators. Saxagliptin added to a thiazolidinedione improves glycemic control in patients with type 2 diabetes and inadequate control on thiazolidinedione alone. J Clin Endocrinol Metab. 2009;94:4810-4819.
42. Scott LJ. Linagliptin: In type 2 diabetes mellitus. Drugs. 2011;71:611–624.
43. Taskinen MR, Rosenstock J, Tamminen I, et al. Safety and efficacy of linagliptin as add-on therapy to metformin in patients with type 2 diabetes: A randomized, double-blind, placebo-controlled study. Diabetes Obes Metab. 2011;13:65–74.
44.Nathan DM, Buse JB, Davidson MB, et al; American Diabetes Association; European Association for Study of Diabetes. Medical management of hyperglycemia in type 2 diabetes: A consensus algorithm for the initiation and adjustment of therapy: A consensus statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care. 2009;32:193-203.
Coalition promotes important acetaminophen dosing reminders
November 18th 2014It may come as a surprise that each year Americans catch approximately 1 billion colds, and the Centers for Disease Control and Prevention estimates that as many as 20% get the flu. This cold and flu season, 7 in 10 patients will reach for an over-the-counter (OTC) medicine to treat their coughs, stuffy noses, and sniffles. It’s an important time of the year to remind patients to double check their medicine labels so they don’t double up on medicines containing acetaminophen.
Support consumer access to specialty medications through value-based insurance design
June 30th 2014The driving force behind consumer cost-sharing provisions for specialty medications is the acquisition cost and not clinical value. This appears to be true for almost all public and private health plans, says a new report from researchers at the University of Michigan Center for Value-Based Insurance Design (V-BID Center) and the National Pharmaceutical Council (NPC).
Management of antipsychotic medication polypharmacy
June 13th 2013Within our healthcare-driven society, the increase in the identification and diagnosis of mental illnesses has led to a proportional increase in the prescribing of psychotropic medications. The prevalence of mental illnesses and subsequent treatment approaches may employ monotherapy as first-line treatment, but in many cases the use of combination of therapy can occur, leading to polypharmacy.1 Polypharmacy can be defined in several ways but it generally recognized as the use of multiple medications by one patient and the most common definition is the concurrent use of five more medications. The presence of polyharmacy has the potential to contribute to non-compliance, drug-drug interactions, medication errors, adverse events, or poor quality of life.
Medical innovation improves outcomes
June 12th 2013I have been diagnosed with stage 4 cancer of the pancreas, a disease that’s long been considered not just incurable, but almost impossible to treat-a recalcitrant disease that some practitioners feel has given oncology a bad name. I was told my life would be measured in weeks.
