- Safety & Recalls
- Regulatory Updates
- Drug Coverage
- COPD
- Cardiovascular
- Obstetrics-Gynecology & Women's Health
- Ophthalmology
- Clinical Pharmacology
- Pediatrics
- Urology
- Pharmacy
- Idiopathic Pulmonary Fibrosis
- Diabetes and Endocrinology
- Allergy, Immunology, and ENT
- Musculoskeletal/Rheumatology
- Respiratory
- Psychiatry and Behavioral Health
- Dermatology
- Oncology
Therapy Update: Nonanthracycline regimens for adjuvant treatment of operable breast cancer
Nonanthracycline-containing chemotherapy regimens for the adjuvant treatment of operable stage I to III breast cancer are discussed, including efficacy and toxicity results from recent randomized clinical trials comparing anthracycline to nonanthracycline-containing regimens.
Abstract
Nonanthracycline-containing chemotherapy regimens for the adjuvant treatment of operable stage I to III breast cancer are discussed, including efficacy and toxicity results from recent randomized clinical trials comparing anthracycline to nonanthracycline-containing regimens. Adverse effect profiles and monitoring approaches are also discussed. Although anthracyclines such as doxorubicin have been the backbone of adjuvant breast cancer treatment for nearly 25 years, they are a known cause of cardiotoxicity, and therefore, research investigating less toxic yet equally efficacious treatment options is of great interest. Recently published and presented clinical trials have shown nonanthracycline regimens to be comparable to anthracycline-containing regimens in terms of efficacy, but with lower rates of cardiotoxicity. Therefore, the use of the nonanthracycline regimens such as docetaxel plus cyclophosphamide for human epidermal growth factor receptor 2 (HER2) negative tumors and a combination of docetaxel, carboplatin, and trastuzumab for HER2 receptor positive disease for adjuvant treatment has increased. Anthracycline-containing and nonanthracycline-containing chemotherapy regimens have differing adverse effect profiles, and therefore, monitoring recommendations differ. Nonanthracycline-containing regimens are included as feasible treatment options in recent treatment guidelines, and their use has increased. Pharmacists can play a vital role in recognizing the adverse effects and necessary monitoring for these newer regimens.
Breast cancer is the most common nondermatologic cancer among American women with more than 190,000 cases diagnosed in 2009 alone. With earlier diagnoses due to mammographic screening and improved treatment options, mortality from breast cancer is continually decreasing. However, breast cancer continues to be the second leading cause of cancer death in women, and therefore, continued research into better treatment options for this common disease is still paramount.1
Treatment guideline overview
Nonmetastatic, or operable stage I to III, breast cancer is localized to the breast and axillary lymph nodes and is characterized as not having spread to distant locations. The National Comprehensive Cancer Network (NCCN) provides recent and updated treatment guidelines for all stages of breast cancer. In respect to nonmetastatic breast cancer, treatment includes local treatment with surgery and in some cases, radiation, and possible adjuvant systemic therapy includes endocrine therapy and/or chemotherapy. The necessity for and selection of a therapeutic modality depends on a number of prognostic factors including the size of the tumor, axillary lymph node status, tumor hormone receptor content, tumor HER2 receptor status, and nuclear grade. Although most patients with estrogen receptor positive breast cancer receive endocrine therapy, the decision to use systemic chemotherapy, including traditional cytotoxic and biologic therapy is more complex. In general, for small tumors (less than 0.5 cm) that do not involve any lymph nodes, prognosis is so favorable that systemic chemotherapy or endocrine therapy is not recommended by the NCCN guidelines. However, endocrine therapy with a selective estrogen receptor modulator such as tamoxifen may be considered to reduce the risk of future breast cancer development. For larger tumors greater than 1 cm, tumors with negative prognostic features, and patients with lymph node involvement, recommended treatment includes chemotherapy, and if the tumor is hormone receptor positive, the addition of endocrine therapy.2 A defined number of treatment cycles of adjuvant treatment is given to eradicate micrometastatic disease that may remain after the complete surgical removal of the breast tumor, in an effort to reduce the chance of recurrence.3
The NCCN guidelines give recommendations for a number of appropriate combination adjuvant chemotherapy regimens for nonmetastatic, operable breast cancer that have been evaluated in phase 3 clinical trials. Both the anthracycline- and nonanthracycline-containing chemotherapy regimens are now included as preferred regimens (Tables 1 and 2). The introduction of the nonanthracycline options is a relatively new addition to the guidelines.
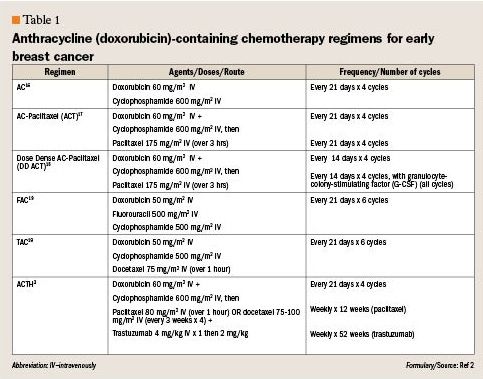
Table 1
The discovery of the upregulation and impact of the HER2 receptor on breast tumor cells has been a significant advance in the treatment of breast cancer, and current guidelines delineate differences in recommended treatment regimens depending on HER2 status. Although upregulation of the HER2 receptor occurs in 20% to 25% of breast cancers and portends a poorer prognosis, it also provides a potential target for treatment with HER2 receptor inhibitors such as trastuzumab, a monoclonal antibody directed against the HER2 receptor. Adjuvant use of trastuzumab has been shown to increase survival in patients with HER2 receptor positive breast cancers when used with chemotherapy and the combination is recommended in the NCCN guidelines for all HER2 receptor positive tumors eligible for systemic adjuvant therapy.3,4 Romond et al reported a 50% reduction in disease recurrence and a significant increase in overall survival after 52 weeks of trastuzumab (H)with an anthracycline (A)/cyclophosphamide (C)/taxane (T) chemotherapy regimen compared to an ACT regimen alone (Table 1).3

Table 2
Recent clinical trials
Although anthracycline, i.e. doxorubicin-based regimens such as AC have been used for several decades for the treatment of breast cancer, anthracyclines are a known cause of cardiotoxicity and therefore, research into less toxic yet efficacious regimens is of great interest. The introduction of the taxanes, paclitaxel and docetaxel, as very effective agents for breast cancer treatment has made the investigation of taxane-based, nonanthracycline-containing chemotherapy regimens a possibility. Several phase 3, randomized clinical trials have been recently published or presented comparing nonanthracycline-containing regimens to anthracycline-containing chemotherapy regimens. The US Oncology Trial 9735 compared 4 cycles of docetaxel and cyclophosphamide (TC) to 4 cycles of adjuvant doxorubicin and cyclophosphamide (AC) in 1016 cases of operable stages I to III breast cancer. At the 7-year follow-up, the authors reported superior disease-free survival with TC [81% versus 75% AC, P=.033, HR= 0.74; 95% CI, 0.56-0.98)], and also a significant overall survival benefit [87% TC versus 82% AC, P=.032, (HR 0.69; 95% CI, 0.5-0.97]). This trial demonstrated that cardiac toxicity was lower in the TC arm. There was 1 death from congestive heart failure (CHF) and 4 cases of myocardial infarction (MI) in the AC arm compared with no CHF and 2 cases of MI in the TC arm. However, neutropenia was increased among patients receiving TC.5
Although anthracyclines have been found in some previous trials to be more effective for HER2 receptor positive cancers than nonanthracycline-containing regimens, the recent results of the Breast Cancer International Research Group (BCIRG) trial 006, challenge those findings.6 This phase 3 randomized clinical trial of more than 3,000 women with HER2 receptor positive cancers tested AC followed by adjuvant TH with an ACT control group (Tables 1 and 2). This study’s updated 5-year analysis from the ongoing trial was presented at the Annual San Antonio Breast Cancer Symposium in December 2009. The investigators reported that both the ACTH and TCH regimens were superior to the ACT regimen without trastuzumab, and that the ACTH and TCH regimens were similarly efficacious. In terms of overall survival, compared to the ACT control group, the ACTH group had an HR of 0.63 (95% CI, 0.48-0.81; PP=.038). Although patients in the TCH group experienced more myelosuppression, the anthracycline-containing ACT and ACTH regimens were associated with more serious toxicities including leukemias (7 patients vs 1 patient), and CHF (25 patients vs 4 patients, P=.0015).4,12
To summarize these recent prospective randomized clinical trial results, TC appears to be an option for HER2 receptor negative patients and TCH appears to be a reasonable option for HER2 receptor positive patients; both chemotherapy regimens have been added to the updated NCCN practice guidelines in oncology for invasive breast cancer.2 At this time, there is no consensus on whether anthracycline-containing regimens should be abandoned in favor of nonanthracycline regimens and additional study and longer follow up are required. However, these regimens are a valuable addition to the breast cancer treatment armamentarium and may provide options for patients in whom cardiotoxicity should be avoided, such as the elderly and those with heart disease.
Toxicities and suggested monitoring
The US Oncology Trial 9735 and BCIRG 006 have demonstrated differing adverse effect profiles between anthracycline and nonanthracycline-containing regimens, and the differences are related to the known toxicities of doxorubicin in AC, docetaxel in TC, and in the case of TCH, carboplatin. For example, AC patients in the US Oncology Trial experienced significantly more cardiotoxicity, nausea, and vomiting compared with patients randomly assigned to TC. 5
Anthracyclines are known to cause cardiotoxicity, including potentially irreversible cardiomyopathy and CHF, which may appear months to years after treatment. The average incidence of cardiotoxicity is 5.1% but is highly dependent on the lifetime cumulative dose, which should not exceed 550 mg/m2 or 450 mg/m2 if the patient has received prior chest radiotherapy. Age has also been reported to increase the risk of anthracycline-induced cardiotoxicity, as have prior mediastinal radiation; concomitant cyclophosphamide, taxanes, or trastuzumab; preexisting heart disease; hypertension; female gender; and liver disease.7,8 Hershman et al reported a 29% increase in the risk of CHF among women older than 65 years receiving anthracyclines, which is 2 to 3 times the risk seen in the general population receiving anthracyclines.9 Suggested monitoring for cardiotoxicity includes cardiac evaluation employing determination of left ventricular ejection fraction with electrocardiograms or multigated acquisition (MUGA) scans performed prior to initiation of therapy. Therapy is not recommended for patients with impaired cardiac function.
To prevent the nausea and vomiting associated with anthracycline treatment, patients should be premedicated with a triple regimen of a steroid, serotonin antagonist, and neurokinin-1 antagonist, as per supportive care antiemesis guidelines.2,10,11
Other adverse effects associated with anthracyclines include myelosuppression, mouth sores, red-tinged urine, chemical phlebitis, alopecia, and increased toxicity in patients with hepatic impairment. Suggested monitoring includes a complete blood count (CBC) prior to and at intervals during therapy, regular oral examinations, and liver function tests prior to each dose. Dosage of doxorubicin should be reduced in patients with impaired hepatic function, and withheld if there are clinical symptoms of CHF, such as dyspnea.12
TC patients in the US Oncology Trial experienced significantly more edema, myalgia, arthralgia, and neutropenia, compared to patients in the AC arm, secondary to the use of docetaxel in TC.5 Other adverse effects commonly associated with docetaxel include hypersensitivity infusion reactions, peripheral neuropathy, fatigue, nail changes, and alopecia. Additionally, increased toxicities can be observed in those with liver impairment. Suggested monitoring for patients taking docetaxel includes a CBC and liver function tests prior to each cycle of therapy, and close observation for infusion reactions, especially during the first and second infusions, with immediate discontinuation for significant reactions. Premedication with dexamethasone 8 mg twice daily for 3 days starting 1 day prior to therapy is suggested to reduce the severity of edema and hypersensitivity reactions. The dosage of docetaxel should be reduced or withheld in patients with impaired hepatic function, neutropenia, myelosuppression, and peripheral neuropathy, according to the package labeling.13
In the BCIRG 006 trial, more thrombocytopenia was observed in the TCH arm than in the anthracycline-containing treatment arms, therefore platelet counts should be monitored closely during therapy.4
Trastuzumab used in the ACTH and TCH arms of the BCIRG 006 trial, is also associated with cardiotoxicity similar to that seen with anthracycline use, however, the mechanisms and risk factors are poorly delineated. Unlike anthracycline cardiotoxicity, trastuzumab-induced cardiotoxicity appears to be generally reversible when the agent is discontinued and standard heart failure medications seem to hasten recovery.7,14 Echocardiograms or MUGA scans should be performed at regular intervals during therapy to monitor cardiac function. Trastuzumab should be withheld when there is a significant reduction in ejection fraction symptoms of heart failure, and unlike anthracyclines, may be reintroduced after recovery with close cardiac monitoring.8
Studies have shown that the concurrent use of anthracyclines and trastuzumab notably increases levels of cardiotoxicity, therefore, most anthracycline-containing regimens complete the doxorubicin cycles first, 2 to 3 weeks before starting trastuzumab (Tables 1 and 2).15
Trastuzumab is generally well tolerated, with the most common side effects being infusion-related reactions consisting of fever and chills. If this occurs, retreatment may be attempted with premedications.12 Rarely, more serious infusion-related reactions, such as bronchospasm, have been reported.
Conclusions
With an increasing number of long-term breast cancer survivors and the increased incidence of cancers as the American population ages, the number of patients experiencing anthracycline-induced cardiotoxicity may also continue to grow. Older women or those with hypertension or cardiac disease may be at higher risk of cardiotoxicity associated with anthracyclines, and heart failure is already the leading reason for hospitalization in elderly populations.9 Therefore, continued research to find effective, yet less toxic regimens for breast cancer treatment is paramount. Recent phase 3 randomized trials have shown adjuvant, nonanthracycline regimens to be equivalent or superior to anthracycline-containing regimens for the treatment of operable breast cancers. These results indicate that nonanthracycline regimens are viable treatment options and such regimens have been incorporated into recent NCCN treatment guidelines for breast cancer. However, the optimal regimen is not known at this time, and additional clinical trials and data from extended follow-up of existing studies are required. Clinical trials investigating nonanthracycline-containing regimens are ongoing, and may help determine optimal treatment strategies. Until then, the introduction of nonanthracycline regimens has increased treatment options for patients and may offer options for patients in whom avoidance of cardiotoxicity is advised. The use of nonanthracycline-containing regimens has increased in recent years and pharmacists can play a vital role in recognizing the adverse effects of and additional monitoring needed with these newer regimens.
Dr Schwartz is an assistant professor in the Department of Pharmacy Practice, Albany College of Pharmacy and Health Sciences, Colchester, Vt.
References
1. National Cancer Institute (NCI). Available at: http://www.cancer.gov/cancertopics/types/breast. Accessed April 1, 2010.
2. NCCN Clinical Practice Guidelines in Oncology: Breast Cancer v.2.2010. Available at: www.nccn.org. Accessed April 1, 2010.
3. Romond EH, Perez EA, Bryant J, et al. Trastuzumab plus adjuvant chemotherapy for operable HER2-positive breast cancer. N Engl J Med. 2005;353(16):1673–1684.
4. Slamon D EW, Robert N, et al. Phase III trial comparing AC-T with AC-TH and with TCH in the adjuvant treatment of HER2 positive early breast cancer patients: Third interim efficacy analysis. Presented at the San Antonio Breast Cancer Symposium, San Antonio, TX;2009, Abstract 62.
5. Jones S, Holmes FA, O'Shaughnessy J, et al. Docetaxel with cyclophosphamide is associated with an overall survival benefit compared with doxorubicin and cyclophosphamide: 7-year follow-up of US Oncology Research Trial 9735. J Clin Oncol. 2009;27(8):1177–1183.
6. Gianni L, Valagussa P. Anthracyclines and early breast cancer: The end of an era? J Clin Oncol. 2009;27(8):1155–1157.
7. Seidman A, Hudis C, Pierri MK, et al. Cardiac dysfunction in the trastuzumab clinical trials experience. J Clin Oncol. 2002;20(5):1215–1221.
8. Hershman DL, Shao T. Anthracycline cardiotoxicity after breast cancer treatment. Oncology (Williston Park). 2009 Mar;23(3):227–234.
9. Hershman DL, McBride RB, Eisenberger A, et al. Doxorubicin, cardiac risk factors, and cardiac toxicity in elderly patients with diffuse B-cell non-Hodgkin's lymphoma. J Clin Oncol. 2008;26(19):3159–3165.
10. NCCN Clinical Practice Guidelines in Oncology: Antiemesis v.1.2010. Available at: www.nccn.org. Accessed April 1, 2010.
11. Kris MG, Hesketh PJ, Somerfield MR, et al. American Society of Clinical Oncology guideline for antiemetics in oncology: update 2006. J Clin Oncol. 2006;24(18):2932–2947.
12. Adriamycin [package insert]. Bedford, OH; Bedford Laboratories: 2006.
13. Taxotere [package insert]. Bridgewater, NJ; Sanofi-Aventis: 2008.
14. Ewer MS, Vooletich MT, Durand JB, et al. Reversibility of trastuzumab-related cardiotoxicity: new insights based on clinical course and response to medical treatment. J Clin Oncol. 2005;23(31):7820–7826.
15. Slamon DJ, Leyland-Jones B, Shak S, et al. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpresses HER2. N Engl J Med. 2001;344(11):783–792.
16. Fisher B, Brown AM, Dimitrov NV, et al. Two months of doxorubicin-cyclophosphamide with and without interval reinduction therapy compared with 6 months of cyclophosphamide, methotrexate, and fluorouracil in positive-node breast cancer patients with tamoxifen-nonresponsive tumors: results from the National Surgical Adjuvant Breast and Bowel Project B-15. J Clin Oncol. 1990;8(9):1483–1496.
17. Henderson IC, Berry DA, Demetri GD, et al. Improved outcomes from adding sequential paclitaxel but not from escalating doxorubicin dose in an adjuvant chemotherapy regimen for patients with node-positive primary breast cancer. J Clin Oncol. 2003;21(6):976–983.
18. Citron ML, Berry DA, Cirrincione C, et al. Randomized trial of dose-dense versus conventionally scheduled and sequential versus concurrent combination chemotherapy as postoperative adjuvant treatment of node-positive primary breast cancer: first report of Intergroup Trial C9741/Cancer and Leukemia Group B Trial 9741. J Clin Oncol. 2003;21(8):1431–1439.
19. Martin M, Pienkowski T, Mackey J, et al. Adjuvant docetaxel for node-positive breast cancer. N Engl J Med. 2005;352(22):2302–2313.
FDA Issues Complete Response Letter for Pz-Cel to Treat Epidermolysis Bullosa
April 22nd 2024Prademagene zamikeracel is a cell therapy designed to incorporate the functional collagen-producing COL7A1 gene into a patient’s own skin cells. The FDA is asking for additional information on manufacturing practices.
