- Safety & Recalls
- Regulatory Updates
- Drug Coverage
- COPD
- Cardiovascular
- Obstetrics-Gynecology & Women's Health
- Ophthalmology
- Clinical Pharmacology
- Pediatrics
- Urology
- Pharmacy
- Idiopathic Pulmonary Fibrosis
- Diabetes and Endocrinology
- Allergy, Immunology, and ENT
- Musculoskeletal/Rheumatology
- Respiratory
- Psychiatry and Behavioral Health
- Dermatology
- Oncology
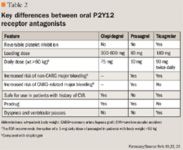
Ticagrelor: An oral, non-thienopyridine P2Y12 receptor antagonist for the treatment of acute coronary syndromes
Clinical studies of patients with both ST-elevation and non-ST-elevation acute coronary syndromes have shown that ticagrelor, when compared with clopidogrel, reduces the rates of vascular death and myocardial infarction while increasing the rate of non-coronary artery bypass graft-related major bleeding. Ticagrelor was also associated with a higher incidence of dyspnea and ventricular pauses.

Key Points
Abstract
Coronary heart disease and acute coronary syndromes (ACS) are a significant cause of morbidity and mortality in the United States. The cornerstone of ACS management is combination oral antiplatelet therapy, usually with aspirin and a thienopyridine. Clopidogrel, the most commonly used thienopyridine, is limited by a high degree of interpatient variability and inconsistent inhibition of platelets. Ticagrelor, a new, oral, direct-acting P2Y12 receptor antagonist, produces a more profound and consistent antiplatelet effect than clopidogrel. Clinical studies of patients with both ST-elevation and non-ST-elevation ACS have shown that ticagrelor, when compared with clopidogrel, reduces the rates of vascular death and myocardial infarction while increasing the rate of non-coronary artery bypass graft-related major bleeding. Ticagrelor was also associated with a higher incidence of dyspnea and ventricular pauses. A new drug application was submitted to FDA for ticagrelor in November 2009. If approved, ticagrelor will be an attractive alternative antiplatelet agent for patients with ACS. (Formulary. 2010; 45:148–154.)
Coronary heart disease (CHD) and acute coronary syndromes (ACS) remain a significant cause of morbidity and mortality in the United States. It is estimated that more than 1 million Americans will suffer an ACS this year, resulting in approximately 150,000 fatalities.1 In 2006, CHD resulted in more than 1.3 million percutaneous coronary interventions (PCI) and more than 400,000 coronary artery bypass grafts (CABG).1 The overall estimated cost of CHD for 2010 is approximately $177 billion.1
Although clopidogrel has been shown to improve outcomes in patients with ACS when added to aspirin therapy, significant limitations are associated with its use.3 Response to clopidogrel is hampered by slow and variable transformation of the prodrug to the active metabolite, as well as modest and variable degrees of platelet inhibition.4 Inhibition or induction of one of the cytochrome P450 enzymes responsible for the conversion of clopidogrel to its active metabolite can explain some of this variable response. Examples of factors that can affect the activity of these CYP450 enzymes include genetic polymorphisms (CYP2C19), smoking status (CYP1A2), or concomitant drug therapy (CYP2C9).5–7 Ultimately, patients who have lower degrees of platelet inhibition with clopidogrel may be at increased risk of adverse ischemic outcomes.8
Prasugrel is a new thienopyridine that produces a more potent and consistent inhibition of platelets compared with clopidogrel.9 While prasugrel has been shown to reduce the risk of recurrent myocardial infarction (MI) in patients with ACS when compared with clopidogrel, it also significantly increases the risk of major bleeding (especially in CABG patients).10 An antiplatelet agent that produces more potent and consistent inhibition of platelets without substantially increasing the risk of bleeding would therefore be highly desirable. Ticagrelor (pronounced tye" ka grel' or) is a new oral antiplatelet agent that is currently under review by FDA as of November 2009 for the treatment of ACS.
CHEMISTRY AND PHARMACOLOGY
The pathophysiology of ACS is characterized by rupture of an atherosclerotic plaque within the coronary artery, with subsequent platelet aggregation, thrombus formation, and ischemia. Before platelets can aggregate, they must first be activated to express glycoprotein IIb/IIIa receptors on the cell surface. This activation is the result of stimulation from endogenous platelet agonists, such as thromboxane A2, epinephrine, or adenosine diphosphate (ADP).11 ADP activates platelets by binding to P2Y12 receptors on the cell surface. These P2Y12 receptors are irreversibly antagonized by thienopyridines like clopidogrel and ticlopidine.12
Ticagrelor, formerly known as AZD6140, is the first of a new chemical class of antiplatelet agents, the cyclo-pentyl-triazolo-pyrimidines.13 Similar to the thienopyridines, ticagrelor is an oral P2Y12 receptor antagonist that exerts its antiplatelet effects by blocking ADP. However, unlike the thienopyridines, ticagrelor is not a prodrug, and therefore does not require metabolic activation in order to inhibit platelets. Furthermore, ticagrelor completely inhibits ADP-induced platelet inhibition ex vivo through reversible binding reversibly to the P2Y12 receptors.13 These properties give ticagrelor several theoretical advantages over clopidogrel. Since ticagrelor does not require metabolic activation, it avoids the variability seen with the CYP450 system and therefore produces a more consistent antiplatelet effect. Also, since ticagrelor is reversible, it does not have to be withheld for 5 to 7 days prior to CABG like clopidogrel and prasugrel.
There have been several studies comparing the pharmacodynamic effects of ticagrelor with clopidogrel. In a randomized study, 200 patients with atherosclerosis received ticagrelor 50, 100, or 200 mg twice daily, ticagrelor 400 mg daily, or clopidogrel 75 mg daily for 28 days.13 The main pharmacodynamic measure was the inhibition of ADP-induced platelet aggregation as measured by optical aggregometry in response to 20 µM ADP. On day 1, peak inhibition of platelet aggregation (IPA) was observed 2 to 4 hours post-dose with ticagrelor, whereas clopidogrel minimally inhibited platelet aggregation (mean percentage IPA, <20%, all time points). On day 14, the 3 higher dosages of ticagrelor produced comparable degrees of platelet inhibition (90% to 95%), which exceeded that with ticagrelor 50 mg twice daily or clopidogrel (~60%). This study demonstrates that ticagrelor in doses ≥100 mg twice daily produces faster and more effective IPA than clopidogrel. It is worth noting that there was no loading dose of clopidogrel used in this study.
A substudy of DISPERSE (Dose confirmation Study assessing anti-Platelet Effects of AZD6140 vs clopidogRel in non-ST-segment Elevation myocardial infarction)-2 compared the antiplatelet effects of ticagrelor and clopidogrel and assessed the effects of ticagrelor in clopidogrel-pretreated patients.14 Patients were randomly assigned to receive either ticagrelor 90 mg twice daily, ticagrelor 180 mg twice daily, or clopidogrel 75 mg once daily for up to 12 weeks. One-half the patients in each ticagrelor group were randomly assigned to receive a 270-mg loading dose. Patients randomly assigned to receive clopidogrel were given a loading dose of 300 mg if clopidogrel naïve (n=45) or a maintenance dose of 75 mg once daily if clopidogrel-pretreated (n=44). The primary outcome was again inhibition of ADP-induced platelet aggregation as measured by optical aggregometry in response to 20 µmol/L ADP. In clopidogrel-naïve patients, ticagrelor produced a statistically significant increase in IPA at 2 hours over clopidogrel (~60% to 80% vs 20%, respectively; P<.05). This difference was dose-dependent, and it persisted throughout the 12-week study period. In clopidogrel-pretreated patients, the mean baseline level of platelet aggregation in the group of patients allocated to continue taking clopidogrel 75 mg daily was 38%. On the other hand, the patients who were switched to ticagrelor experienced substantial further inhibition of platelet aggregation in a dose-dependent fashion, with patients in the 180-mg twice daily group reaching mean platelet aggregation levels of less than 5%. This study demonstrates that superior platelet inhibition is achieved with ticagrelor in both clopidogrel-naïve and clopidogrel-pretreated patients. This superior response was seen despite the use of a 300-mg loading dose of clopidogrel.
The ONSET/OFFSET study sought to further delineate the antiplatelet effects of ticagrelor compared to clopidogrel.15 Patients with stable CHD were randomly assigned to receive either a ticagrelor 180-mg loading dose followed by 90 mg twice daily (n=54), clopidogrel 600-mg loading dose followed by 75 mg daily (n=50), or placebo (n=12). The primary pharmacodynamic measure was the same as in the DISPERSE-2 substudy. Ticagrelor inhibited platelets to a greater extent than did clopidogrel at 30 minutes (mean IPA, 40% versus ~5%, respectively; P<.0001) and at 2 hours (mean IPA, ~90% versus ~40%, respectively; P<.0001). After stopping the study medication, levels of platelet inhibition with ticagrelor had decreased by roughly 50% after 24 hours and were numerically similar to the IPA produced by clopidogrel at that time point. The ticagrelor group had significantly lower IPA at 72 (P<.05) and 120 (P<.005) hours than the clopidogrel group had after stopping the study medication. Hence, ticagrelor produces more rapid and profound platelet inhibition over clopidogrel, despite the use of a 600-mg loading dose. Furthermore, the dissipation of antiplatelet effect of ticagrelor is more rapid than that of clopidogrel, which is evidenced by the lower degree of IPA in the ticagrelor group within 72 hours of stopping therapy.
PHARMACOKINETICS
Ticagrelor is rapidly absorbed following oral administration, with an onset of antiplatelet effect within 30 minutes and Tmax occurring around 3 hours.13,14 The estimated Cmax is 770 ng/mL; the estimated AUC is 4,762 ng-h/mL.14 Total clearance is 26 L/h, with an apparent half-life of approximately 8 to 12 hours.14,16
Ticagrelor is converted by CYP3A4 to an active metabolite, AR-C124910XX. This compound peaks in about 3 hours and is present in blood at about one-third the concentration of ticagrelor.14 This metabolite is approximately as potent as ticagrelor at blocking the P2Y12 receptor in vitro and is thought to contribute to antiplatelet effects after oral dosing with ticagrelor.13 Although no formal drug-drug interaction studies have been conducted with ticagrelor, it is important to note that patients taking inhibitors, inducers, or substrates of CYP3A were generally excluded from clinical trials.
CLINICAL TRIALS
Ticagrelor has been evaluated in several clinical trials for the treatment of ACS. In the DISPERSE-2 trial, 990 patients with non-ST-elevation ACS were randomly assigned to ticagrelor 90 mg twice daily, ticagrelor 180 mg twice daily, or clopidogrel 300-mg loading dose followed by 75 mg daily for up to 3 months.17 All patients received aspirin, and the use of other standard therapies for ACS such as heparin or a IIb/IIIa inhibitor was allowed at the discretion of the treating physician. The primary objective was to assess the safety and tolerability of the different doses of ticagrelor versus clopidogrel in patients with non-ST-segment elevation ACS by evaluating total bleeding events observed within the first 4 weeks of treatment. Bleeding was primarily defined according to the Thrombolysis in Myocardial Infarction (TIMI) criteria.18 The rate of the primary end point-TIMI major or minor bleeding at 4 weeks-was not different among the 3 groups, with 26 patients (8.1%) experiencing a bleeding episode in the clopidogrel group, compared with 32 (9.8%) in the ticagrelor 90-mg group and 25 (8.0%) in the 180-mg group (P=.43 and P=.96 vs clopidogrel, respectively). Rates of major and fatal or life-threatening bleeding were also not different between the 3 groups. Although secondary end points, such as rates for cardiovascular death or MI, were not different between groups, there was a trend toward a numerically lower rate of MI in favor of the ticagrelor groups (P=NS). Overall, the study demonstrated similar safety and tolerability for ticagrelor compared with clopidogrel. This study was relatively small, however, and not powered to detect differences in efficacy end points.

The primary end point, death from vascular causes, MI, or stroke, occurred significantly less often in the ticagrelor group than in the clopidogrel group (9.8% vs 11.7% at 12 months; HR, 0.84; 95% CI, 0.77–0.92; P<.001). This difference was apparent as early as 30 days after the start of treatment, and was driven by statistically significant reductions in both vascular death and MI (P<.01 for both). The ticagrelor and clopidogrel groups did not differ significantly with regard to the rates of major bleeding as defined in the trial (11.6% and 11.2%, respectively; P=.43). The 2 treatment groups also did not differ significantly in the rates of CABG-related TIMI major bleeding, despite the fact that ticagrelor was only withheld for 24 to 72 hours prior to surgery while clopidogrel was withheld for 5 days. However, there was a higher rate of non-CABG-related major bleeding according to the study criteria (4.5% vs 3.8%, P=.03) and TIMI criteria (2.8% vs 2.2%, P=.03) with ticagrelor as compared with clopidogrel. There were also more episodes of intracranial bleeding (26 [0.3%] vs 14 [0.2%], P=.06), including fatal intracranial bleeding (11 [0.1%] vs 1 [0.01%], P=.02).
Based on the results of the PLATO study, treating 1,000 ACS patients with ticagrelor instead of clopidogrel would prevent 11 vascular deaths and 11 MIs at the cost of 6 non-CABG-related major bleeding episodes. The overall results of this study demonstrate that in patients who had ACS, treatment with ticagrelor significantly reduced the rate of death from vascular causes and MI, without an increase in the rate of overall major bleeding but with an increase in the rate of non-procedure-related bleeding.
A subgroup analysis of the 13,408 patients in PLATO who underwent an early invasive management strategy was presented separately.20 Overall, the results of this subgroup were similar to those in the main PLATO study. The primary composite end point of death from vascular causes, MI, and stroke occurred in fewer patients in the ticagrelor group than in the clopidogrel group (9.0% vs 10.7% at 12 months; HR, 0.84; 95% CI, 0.75–0.94; P=.0025). This was again driven by statistically significant reductions in vascular death and MI (P<.05 for both). There was no difference between ticagrelor and clopidogrel for CABG-related TIMI major bleeding (5.3% vs 5.9%, respectively; P=.19). However, there was a strong trend toward higher rates of non-CABG-related TIMI major bleeding with ticagrelor over clopidogrel (2.8% vs 2.2%, respectively; P=.08). Rates of major bleeding using other definitions (Global Use of Strategies to Open Occluded Coronary Arteries or GUSTO study criteria) were not different between agents. In summary, the results of this analysis confirm the efficacy and safety of ticagrelor in patients with ACS who are managed with an early invasive strategy.
ADVERSE EVENTS
Ticagrelor was well-tolerated in clinical trials, with only 7.4% of patients prematurely discontinuing therapy because of adverse drug events.19 Bleeding is the most serious adverse effect of ticagrelor therapy. Overall rates of major bleeding varied depending on the definition used, and generally ranged between 2.8% and 11.6%.17,19,20 Rates of life-threatening or fatal bleeding were as high as 6%; rates of intracranial hemorrhage were 0.3%.
In addition to bleeding, patients receiving ticagrelor had significantly more episodes of dyspnea (10% to 20%) and ventricular pauses lasting more than 3 seconds (6%). Most of the episodes of dyspnea were minor, and only 1% of patients in the ticagrelor group stopped therapy because of this adverse event. Also, the episodes of ventricular pauses were usually self-limiting (no difference between groups by day 30) and were not associated with an increased risk of pacemaker insertion or syncope.19
Finally, ticagrelor therapy was associated with an increase in serum uric acid (15% increase from baseline value) and serum creatinine (11% increase from baseline value). Both of these lab derangements appear to be reversible, as there were no differences in these values between clopidogrel- and ticagrelor-treated patients within 30 days of stopping treatment.19
DOSING AND ADMINISTRATION
Ticagrelor has been studied in dosages of 50, 90, 100, 180, and 200 mg twice daily, or 400 mg once daily. Early studies found that ticagrelor 100 mg and 200 mg twice a day appeared to have a more beneficial safety and tolerability profile than ticagrelor 400 mg daily and were superior to ticagrelor 50 mg twice daily with regard to antiplatelet efficacy.13 Later studies showed higher rates of minor bleeding with ticagrelor 180 mg twice daily versus 90 mg twice daily.17 In the large, randomized trials, the dosage of ticagrelor was a 180-mg load followed by 90 mg twice daily.19,20 This is likely the dosage that will be recommended by FDA if this drug is approved for the management of ACS.
FORMULARY CONSIDERATIONS
Clinical studies have demonstrated the superiority of ticagrelor, an oral, direct inhibitor of the P2Y12 receptor, compared with clopidogrel in patients with ACS. These favorable outcomes were seen in a wide range of patients, including those managed invasively and conservatively and those with and without ST-elevation ACS. The ability of ticagrelor to reduce both vascular death and MI is noteworthy, as both clopidogrel and prasugrel have not demonstrated vascular mortality reduction in the management of patients with ACS.3,10 This improvement in outcomes was coupled with an increased incidence of non-CABG-related major bleeding, dyspnea, and ventricular pauses.
The improved outcomes seen in clinical trials was likely due to the more complete and consistent inhibition of platelets seen with ticagrelor over clopidogrel. Additionally, the relatively fast "offset" of antiplatelet effect that is seen with ticagrelor would offer clinicians a new dimension of flexibility when managing ACS patients. Currently, an ACS patient who receives clopidogrel or prasugrel must wait for 5 to 7 days before undergoing CABG, due to the increased risk of major bleeding. Therefore, some clinicians are hesitant to use a thienopyridine in a patient with ACS until the coronary anatomy is defined and the possibility of CABG is excluded. On the other hand, clinical studies show that holding ticagrelor for 24 to 72 hours produced rates of bleeding that were comparable to holding clopidogrel for 5 days. This means that clinicians can use ticagrelor to treat a patient with ACS without needing to postpone CABG for up to a week.
There are several potential drawbacks to ticagrelor therapy in patients with ACS. First, there is a theoretical concern that the fast "offset" of its antiplatelet effect would expose nonadherent patients to increased risk for coronary artery stent thrombosis if they missed 1 or 2 doses. Therefore, clinicians who prescribe ticagrelor will have to be especially diligent about counseling patients regarding the importance of medication adherence. Another limitation of ticagrelor therapy is the potential for dyspnea and ventricular pauses. These adverse drug events were usually mild and self-limiting in the clinical trials; however, clinicians should monitor patients closely when initiating ticagrelor therapy. Finally, as clopidogrel will soon be off patent, ticagrelor will likely be a more expensive antiplatelet agent. Whether this increased medication cost will be offset by reduced overall costs from decreased rates of MI and vascular death remains to be studied in appropriate pharmacoeconomic analyses. It is worth noting that while prasugrel is more expensive than clopidogrel, it may be a more cost-effective agent when factoring in the potential for improved patient outcomes.21
In summary, ticagrelor provides a more potent and consistent inhibition of platelets than clopidogrel. It has been studied across the spectrum of ACS patients, and has been shown to reduce both vascular death and recurrent MI, while increasing the rate of non-CABG-related major bleeding. If approved, ticagrelor would likely represent an attractive alternative antiplatelet agent for patients with ACS.
Dr Jennings is a clinical pharmacy specialist, cardiovascular intensive care unit, Department of Pharmacy Services, Henry Ford Hospital, and adjunct assistant professor, Department of Pharmacy Practice, Wayne State University, Detroit, Mich.
Disclosure Information: The author reports no financial disclosures as related to products discussed in this article.
In each issue, the "Focus on" feature reviews a newly approved or investigational drug of interest to pharmacy and therapeutics committee members. The column is coordinated by Robert A. Quercia, MS, RPh, clinical manager, Department of Pharmacy Services, Hartford Hospital, Hartford, Conn, and adjunct associate professor, University of Connecticut School of Pharmacy, Storrs, Conn; and by Craig I. Coleman, PharmD, associate professor of pharmacy practice, University of Connecticut School of Pharmacy, and director, Pharmacoeconomics and Outcomes Studies Group, Hartford Hospital.
EDITORS' NOTE: The clinical information provided in "Focus on" articles is as current as possible. Due to regularly emerging data on developmental or newly approved drug therapies, articles include information published or presented and available to the author up until the time of the manuscript submission.
REFERENCES
1. Adams RJ, Brown TM, Carnethon M; American Heart Association. Heart Disease and Stroke Statistics–2010 Update. Circulation. 2010;121:e46-e215.
2. Anderson JL, Adams CD, Antman EM, et al; American College of Cardiology; American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 2002 Guidelines for the Management of Patients with Unstable Angina/Non-ST-Elevation Myocardial Infarction); American College of Emergency Physicians; Society for Cardiovascular Angiography and Interventions; Society of Thoracic Surgeons; American Association of Cardiovascular and Pulmonary Rehabilitation; Society for Academic Emergency Medicine. ACC/AHA 2007 guidelines for the management of patients with unstable angina/non-ST-elevation myocardial infarction: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Revise the 2002 Guidelines for the Management of Patients With Unstable Angina/Non-ST-Elevation Myocardial Infarction) developed in collaboration with the American College of Emergency Physicians, the Society for Cardiovascular Angiography and Interventions, and the Society of Thoracic Surgeons endorsed by the American Association of Cardiovascular and Pulmonary Rehabilitation and the Society for Academic Emergency Medicine. J Am Coll Cardiol. 2007;50:e1–e157.
3. Yusuf S, Zhao F, Mehta SR, Chrolavicius S, Tognoni G, Fox KK; Clopidogrel in Unstable Angina to Prevent Recurrent Events Trial Investigators. Effects of clopidogrel in addition to aspirin in patients with acute coronary syndromes without ST-segment elevation. N Engl J Med. 2001;345:494–502.
4. Jernberg T, Payne CD, Winters KJ, et al. Prasugrel achieves greater inhibition of platelet aggregation and a lower rate of non-responders compared with clopidogrel in aspirin-treated patients with stable coronary artery disease. Eur Heart J. 2006;27:1166–1173.
5. Sibbing D, Koch W, Gebhard D, et al. Cytochrome 2C19*17 allelic variant, platelet aggregation, bleeding events, and stent thrombosis in clopidogrel-treated patients with coronary stent placement. Circulation. 2010;121:512–518.
6. Gremmel T, Steiner S, Seidinger D, Koppensteiner R, Panzer S, Kopp CW. Smoking promotes clopidogrel-mediated platelet inhibition in patients receiving dual antiplatelet therapy. Thromb Res. 2009;124:588–591.
7. Gilard M, Arnaud B, Cornily JC, et al. Influence of omeprazole on the antiplatelet action of clopidogrel associated with aspirin: the randomized, double-blind OCLA (Omeprazole CLopidogrel Aspirin) study. J Am Coll Cardiol. 2008;51:256–260.
8. Bliden KP, DiChiara J, Tantry US, Bassi AK, Chaganti SK, Gurbel PA. Increased risk in patients with high platelet aggregation receiving chronic clopidogrel therapy undergoing percutaneous coronary intervention: is the current antiplatelet therapy adequate? J Am Coll Cardiol. 2007;49:657–666.
9. Wiviott SD, Trenk D, Frelinger AL, et al; PRINCIPLE-TIMI 44 Investigators. Prasugrel compared with high loading and maintenance-dose clopidogrel in patients with planned percutaneous coronary intervention: the Prasugrel in Comparison to Clopidogrel for Inhibition of Platelet Activation and Aggregation–Thrombolysis in Myocardial Infarction 44 trial. Circulation. 2007;116:2923–2932.
10. Wiviott SD, Braunwald E, McCabe CH, et al; for the TRITON-TIMI 38 Investigators. Prasugrel versus clopidogrel in patients with acute coronary syndromes. N Engl J Med. 2007;357:2001–2015.
11. Badion JJ, Ibanez B, Fuster V, Badimon L. Coronary thrombosis: local and systemic factors. In: Fuster V, O'Rourke RA, Walsh RA, Poole-Wilson P, eds. Hurst's The Heart. 12th ed. New York: McGraw-Hill; 2008.
12. Majerus PW, Tollefsen D. Anticoagulant, thrombolytic, and antiplatelet drugs. In: Brunton LL, ed. Goodman and Gilman's, The Pharmacological Basis of Therapeutics. 10th ed. New York: McGraw-Hill; 2001:1519–1538.
13. Husted S, Emanuelsson H, Heptinstall S, Sandset PM, Wickens M, Peters G. Pharmacodynamics, pharmacokinetics, and safety of the oral reversible P2Y12 antagonist AZD6140 with aspirin in patients with atherosclerosis: a double-blind comparison to clopidogrel with aspirin. Eur Heart J. 2006;27:1038–1047.
14. Storey RF, Husted S, Harrington RA, et al. Inhibition of platelet aggregation by AZD6140, a reversible oral P2Y12 receptor antagonist, compared with clopidogrel in patients with acute coronary syndromes. J Am Coll Cardiol. 2007;50:1852–1856.
15. Gurbel PA, Bliden KP, Butler K, et al. Randomized double-blind assessment of the ONSET and OFFSET of the antiplatelet effects of ticagrelor versus clopidogrel in patients with stable coronary artery disease: the ONSET/OFFSET study. Circulation. 2009;120:2577–2585.
16. Teng R, Butler K. Pharmacokinetics, pharmacodynamics, tolerability and safety of single ascending doses of ticagrelor, a reversibly binding oral P2Y12 receptor antagonist, in healthy subjects. Eur J Clin Pharmacol. 2010;66:487–496.
17. Cannon CP, Husted S, Harrington RA, et al; DISPERSE-2 Investigators. Safety, tolerability, and initial efficacy of AZD6140, the first reversible oral adenosine diphosphate receptor antagonist, compared with clopidogrel, in patients with non-ST-segment elevation acute coronary syndrome: primary results of the DISPERSE-2 trial. J Am Coll Cardiol. 2007;50:1844–1851.
18. Rao SV, O'Grady K, Pieper KS, et al. A comparison of the clinical impact of bleeding measured by two different classifications among patients with acute coronary syndromes. J Am Coll Cardiol. 2006;47:809–816.
19. Wallentin L, Becker RC, Budaj A, et al; PLATO Investigators. Ticagrelor versus clopidogrel in patients with acute coronary syndromes. N Engl J Med. 2009;361:1045–1057.
20. Cannon CP, Harrington RA, James S, et al; PLATelet inhibition and patient Outcomes Investigators. Comparison of ticagrelor with clopidogrel in patients with a planned invasive strategy for acute coronary syndromes (PLATO): a randomised double-blind study. Lancet. 2010;375:283–293.
21. Mahoney EM, Wang K, Arnold SV, et al. Cost-effectiveness of prasugrel versus clopidogrel in patients with acute coronary syndromes and planned percutaneous coronary intervention: results from the trial to assess improvement in therapeutic outcomes by optimizing platelet inhibition with Prasugrel-Thrombolysis in Myocardial Infarction TRITON-TIMI 38. Circulation. 2010;121:71–79.
22. Plavix [package insert]. Bridgewater, NJ: Sanofi-Aventis; 2010.
23. Effient [package insert]. Daiichi Sankyo and/or Lilly USA; 2010.
Coalition promotes important acetaminophen dosing reminders
November 18th 2014It may come as a surprise that each year Americans catch approximately 1 billion colds, and the Centers for Disease Control and Prevention estimates that as many as 20% get the flu. This cold and flu season, 7 in 10 patients will reach for an over-the-counter (OTC) medicine to treat their coughs, stuffy noses, and sniffles. It’s an important time of the year to remind patients to double check their medicine labels so they don’t double up on medicines containing acetaminophen.
Support consumer access to specialty medications through value-based insurance design
June 30th 2014The driving force behind consumer cost-sharing provisions for specialty medications is the acquisition cost and not clinical value. This appears to be true for almost all public and private health plans, says a new report from researchers at the University of Michigan Center for Value-Based Insurance Design (V-BID Center) and the National Pharmaceutical Council (NPC).
Management of antipsychotic medication polypharmacy
June 13th 2013Within our healthcare-driven society, the increase in the identification and diagnosis of mental illnesses has led to a proportional increase in the prescribing of psychotropic medications. The prevalence of mental illnesses and subsequent treatment approaches may employ monotherapy as first-line treatment, but in many cases the use of combination of therapy can occur, leading to polypharmacy.1 Polypharmacy can be defined in several ways but it generally recognized as the use of multiple medications by one patient and the most common definition is the concurrent use of five more medications. The presence of polyharmacy has the potential to contribute to non-compliance, drug-drug interactions, medication errors, adverse events, or poor quality of life.
Medical innovation improves outcomes
June 12th 2013I have been diagnosed with stage 4 cancer of the pancreas, a disease that’s long been considered not just incurable, but almost impossible to treat-a recalcitrant disease that some practitioners feel has given oncology a bad name. I was told my life would be measured in weeks.
