- Safety & Recalls
- Regulatory Updates
- Drug Coverage
- COPD
- Cardiovascular
- Obstetrics-Gynecology & Women's Health
- Ophthalmology
- Clinical Pharmacology
- Pediatrics
- Urology
- Pharmacy
- Idiopathic Pulmonary Fibrosis
- Diabetes and Endocrinology
- Allergy, Immunology, and ENT
- Musculoskeletal/Rheumatology
- Respiratory
- Psychiatry and Behavioral Health
- Dermatology
- Oncology
Tigecycline: A novel glycylcycline antiobiotic
Highly resistant strains of both gram-positive and gram-negative bacteria are becoming commonplace in both the inpatient and outpatient setting. Recently developed antimicrobials have targeted resistant gram-positive pathogens, but the problem of resistant gram-negative pathogens remains. Tigecycline (Tygacil, Wyeth) is an injectable antimicrobial and the first in a new class of agents (the glycylcyclines) that possesses activity against key gram-positive and gram-negative bacterial pathogens. Tigecycline overcomes common tetracycline resistance mechanisms and has shown in vitro and in vivo activity against multidrug-resistant organisms. Tigecycline treatment produced clinical and microbiologic outcomes similar to standard comparator agents in patients with complicated skin and skin structure infections and complicated intra-abdominal infections and was approved for these indications in June 2005. The most common adverse events associated with tigecycline's administration during clinical trials were..
Abstract
Highly resistant strains of both gram-positive and gram-negative bacteria are becoming commonplace in both the inpatient and outpatient setting. Recently developed antimicrobials have targeted resistant gram-positive pathogens, but the problem of resistant gram-negative pathogens remains. Tigecycline (Tygacil, Wyeth) is an injectable antimicrobial and the first in a new class of agents (the glycylcyclines) that possesses activity against key gram-positive and gram-negative bacterial pathogens. Tigecycline overcomes common tetracycline resistance mechanisms and has shown in vitro and in vivo activity against multidrug-resistant organisms. Tigecycline treatment produced clinical and microbiologic outcomes similar to standard comparator agents in patients with complicated skin and skin structure infections and complicated intra-abdominal infections and was approved for these indications in June 2005. The most common adverse events associated with tigecycline's administration during clinical trials were gastrointestinal in nature. Tigecycline appears to be a promising alternative agent for the management of infections in which resistant pathogens are suspected or documented.(Formulary. 2005;40:245–254.)
Antimicrobial resistance continues to escalate in many key gram-positive and gram-negative bacterial pathogens as we move forward into the 21st century. Methicillin-resistant Staphylococcus aureus (MRSA) comprises over half of all strains isolated from hospitalized patients and now has become a key pathogen in many community-associated staphylococcal infections.1,2 Penicillin resistance in Streptococcus pneumoniae (PRSP) occurs in approximately one-quarter of all isolated strains, and a similar prevalence of vancomycin resistance is present in Enterococcus faecium (VREF).2 Fluoroquinolone and multidrug resistance are increasing at an alarming rate in Pseudomonas aeruginosa, while more and more infections are being described related to highly resistant strains of bacteria such as Acinetobacter and Stenotrophomonas species.3 Although recent advances have been made for the treatment of resistant gram-positive organisms, a paucity of agents exist that can target the problem of antimicrobial resistance in both gram-positive and gram-negative bacteria. In June 2005, a novel injectable glycylcycline antimicrobial, tigecycline (Tygacil, Wyeth), was approved by FDA for the treatment of complicated skin and skin structure infections (cSSSIs) and complicated intra-abdominal infections (cIAIs). Tigecycline represents a significant advance in antimicrobial therapy as it has activity against resistant strains of gram-positive and gram-negative bacteria.
The tetracyclines were first isolated 60 years ago and maintained activity for many years against a wide variety of bacterial pathogens. Currently, tetracycline resistance is widespread amongst most isolates of human pathogenic bacteria. In the early 1990s, new derivatives of tetracycline were synthesized that contained an N,N-dimethylglycylamido (DMG) substitution at the 9 position of the minocycline molecule.4 The addition of a bulky side chain at position 9 results in a derivative that is less affected by the presence of the 2 most common mechanisms of resistance to the tetracycline antimicrobials: tetracycline efflux pump proteins and ribosomal protection proteins. Further optimization of these 9-substituted glycylcycline compounds for improved activity and tolerability resulted in tigecycline, a 9-t-butylglycylamido derivative of minocycline (GAR-936, chemical name (4S,4aS,5aR,12aS)-9-[2-(tert-butylamino)acetamido]-4,7-bis(dimethylamino)-1,4,4a,5,5a,6,11,12a-octahydro-3,10,12,12a-tetrahydroxy-1,11-dioxo-2-naphthacenecarboxamide).5
Both tigecycline and tetracycline bind to the same region of the 30S subunit of the bacterial ribosome (tetracycline binding site-1) to inhibit protein synthesis, but the glycylcycline derivatives bind approximately 5 times more effectively to this common site than tetracycline.6 Tigecycline likely binds to the common tetracycline binding site-1 in a differentorientation than the parent tetracycline molecule, or the conformation of the ribosome becomes altered in order to accommodatethe bulky 9-t-butylglycylamido group.7 Early preclinical investigations verified that tigecycline had significantly improved in vitro and in vivo activity against a wide variety of tetracycline-susceptible and tetracycline-resistant aerobic and anaerobic gram-positive and gram-negative bacteria.8
IN VITRO SUSCEPTIBILITY INVESTIGATIONS

A commercially prepared dry-form broth microdilution MIC panel was developed for tigecycline that shows excellent reproducibility and compares well with traditional broth microdilution MIC testing methods.16 Breakpoint MICs for susceptibility are ≤0.5 mcg/mL for S aureus, ≤0.25 mcg/mL for vancomycin-susceptible Enterococcus faecalis and Streptococcus species other than S pneumoniae, ≤2 mcg/mL for Enterobacteriaceae, and ≤4 mcg/mL for anaerobic bacteria.5 Resistance breakpoints for these latter 2 bacteria are ≥8 and ≥16 mcg/ mL, respectively.
RESISTANCE TO TIGECYCLINE AND MECHANISMS
High-level resistance to tigecycline is currently rare and selection of resistance appears to occur at very low frequencies in strains that have been studied to date.17 Tigecycline is not affected by well-characterized resistance mechanisms for tetracycline antibiotics (ie, tetracycline-specific efflux pumps such as tetA–tetE and tetK; ribosomal protection due to genes such as tetM or tetO). The presence of these common tetracycline resistance determinants resulted in minimal elevations of tigecycline MICs (typically 2- to 4-fold increases) for gram-negative organisms such as Escherichia coli, Enterobacter spp, Klebsiella spp, and Serratia marcescens.18 Similar increases in MICs were observed for Staphylococcus aureus that possessed either tetM or tetK; the presence of tetM appears to impart a slightly higher level of resistance for strains of Streptococcus pneumoniae (2- to 16-fold increases in MICs).18
There are a select number of organisms that appear to possess intrinsic resistance and/or decreased susceptibility to tigecycline. P aeruginosa has high tigecycline MICs (MIC90 >8 g/mL); the mechanism of its intrinsic resistance mechanism involves active efflux of the drug by at least 3 distinct 3-protein multidrug efflux pump systems (MexXY-OprM, MexAB-OprM, and MexCD-OprJ).19 Efflux-based resistance appears to occur via similar multicomponent efflux transporters (ie, the AcrAB and AcrEF efflux pump systems) in other gram-negative bacteria including Morganella morganii, Proteus spp, and some strains of Klebsiella pneumoniae and E coli.20–23 The overexpression of a novel efflux pump (mepA) was recently described as a resistance mechanism for laboratory-derived strains of S aureus obtained after repeated growth of the organism in increasing concentrations of tigecycline.24
PHARMACODYNAMICS
There are limited published data on the pharmacodynamic parameters that correlate with tigecycline efficacy. The pharmacodynamic activity of tigecycline was determined for strains of S pneumoniae, S aureus, E coli, and K pneumoniae using a well-established neutropenic mouse thigh infection model.25 Tigecycline provided bacteriostatic activity against the various strains of S pneumoniae at doses ranging from 0.8 to 5.9 mg/kg/d, whereas significantly higher doses were needed for bacteriostatic activity against the other strains of bacteria (range, 4.3–23 mg/kg/d). Both time above a certain multiple of the organism's MIC (eg, 0.5 or 1 times the MIC) and the area under the concentration-time curve (AUC) correlated best with antibacterial activity (R2 >0.73 for both parameters against all organisms). The in vivo post-antibiotic effect (PAE) was determined against 1 strain each of E coli and S pneumoniae and was found to be 4.9 and 8.9 hours, respectively.
Tigecycline had activity against both tetracycline-susceptible and -resistant strains of E faecalis and E faecium in a mouse model of peritonitis.26 Although the 50% protective subcutaneous dose (PD50) ranged from 0.6 to 5.7 mg/kg and increased for tetracycline-resistant strains, tigecycline continued to provide a protective effect, whereas tetracycline was completely ineffective versus the resistant strains even at doses as high as 200 mg/kg.26 Tigecycline has been evaluated as an option for the treatment of bacterial endocarditis caused by Enterococci and Staphylococci. It had bacteriostatic activity similar to vancomycin versus E faecalis and a strain of MRSA in a rat model of infective endocarditis and resulted in significantly lower end-of-treatment viable bacterial counts of E faecalis in a rabbit model of endocarditis.27,28 Autoradiographic analysis indicated that radiolabeled tigecycline diffused homogenously throughout the vegetations. Tigecycline was compared to vancomycin (both alone or in combination with rifampin) in a rabbit osteomyelitis infection model.29 All antimicrobial treatment regimens produced greater than 80% infection clearance rates that were significantly greater than the untreated control group.
Tigecycline may prove useful for the treatment of Legionella infections based on results obtained from a guinea pig infection model.15 In this model, tigecycline therapy resulted in survival of 13 of 16 treated animals at a 12-day post-infection evaluation, which was similar to the 11 of 12 survival rate observed with azithromycin therapy. The authors concluded that tigecycline would fall into an "intermediate" activity category (good but not excellent) and that it should be effective against mild infections when given as a longer (14–21 d) treatment regimen.15 Further investigation of its clinical utility in Legionella pneumonia appears warranted.

It is common to combine antimicrobials in an attempt to either provide a broader spectrum of antibacterial activity, provide synergistic activity, or to prevent resistance. The effect of combining tigecycline with other common gram-positive antimicrobials was evaluated against 2 strains of VRE, 2 strains of vancomycin-intermediate S aureus, and 1 MRSA using test tube time-kill methods. The combination of tigecycline with gentamicin resulted in improvement or enhancement of killing against 5 of the 6 strains, whereas no significant effect was observed by combining the drug with vancomycin, rifampin, or doxycycline.31 In contrast, the combination of tigecycline and cefepime was antagonistic versus 2 clinical strains of methicillin-resistant S aureus evaluated using an in-vitro pharmacodynamic infection model.32 However, this antagonism was only observed during simulations of the concentration-time profiles of tigecycline 100 mg once-daily combined with cefepime 2 g every 12 hours; indifferent activity was observed when tigecycline was combined with cefepime 2 g every 8 hours or as a simulated continuous infusion.
PHARMACOKINETICS
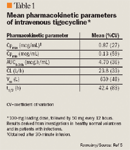
The maximum tolerated doses of tigecycline in fasting and fed patients were 100 mg and 200 mg in a single- and multiple-ascending-dose pharmacokinetic investigation.34 In this study, mean serum Cmax values were unchanged by the presence of fed or fasting conditions, and a linear relationship was observed between Cmax and AUC values over the dosing range studied (12.5–300 mg/d). In the multiple-dose portion of the study, patients who received doses of 75 mg or more experienced substantial nausea and vomiting, but there were no clinically relevant changes in clinical laboratory parameters, blood pressure, or ECG. Although the prevalence of adverse events did not vary significantly between study patients that received varying infusion volumes (100 or 250 mL), subjects given a 30-minute infusion had a lower prevalence of nausea and vomiting, while subjects given a 60-minute infusion had fewer infusion-related reactions.
Tigecycline's steady-state pharmacokinetic profile was determined in both serum and blister fluid in a single-center, open-label, multiple-dose study of 10 healthy volunteers.35 Tigecycline penetrated well into blister fluid, as the mean percent penetration was 74%±7%. Tigecycline may rapidly redistribute from the peripheral skin blister fluid compartment, as the half-life in this compartment was approximately 4 times lower than the serum tigecycline half-life.

PHARMACOKINETICS IN SPECIAL POPULATIONS
Tigecycline is not extensively metabolized; administration of radiolabeled tigecycline to healthy males revealed less than 10% recovery of a glucuronide metabolite and a N-acetyl metabolite.5 The majority of radioactivity was recovered in feces and urine; approximately 60% of a dose was eliminated by biliary/fecal excretion and 22% was renally excreted as unchanged tigecycline.37 The presence of moderate/ severe hepatic impairment (Child Pugh class B/C) reduced systemic clearance of the drug by 25%/55 % and increased elimination half-life by 23%/43%, respectively. Based on these data, the product labeling recommends an initial 100-mg loading dose, followed by a reduced maintenance dose of 25 mg every 12 hours for patients with severe hepatic impairment.5
No alterations in pharmacokinetics occurred in patients with severe renal impairment (including dialysis-dependent end-stage renal disease patients); dosage adjustments are therefore not needed in this population. Pharmacokinetic profiles are unaltered in geriatric patients, as well as in men versus women, and in patients of different race (Asian, Black, Hispanic, and White subjects).5
CLINICAL EFFICACY
The efficacy and safety of tigecycline for the treatment of cSSSIs was investigated in a phase 2, multicenter, randomized, open-label study.33 A total of 164 hospitalized patients aged 18 to 85 years were enrolled and randomized to receive 1) tigecycline 50-mg IV loading dose, then 25 mg every 12 hours, or 2) tigecycline 100-mg IV loading dose, then 50 mg every 12 hours for 7 to 14 days. The primary efficacy end point was clinical cure rate in clinically evaluable patients (n=109). The clinical cure rates and microbial eradication rates at the end of therapy were statistically similar for the 25-mg treatment group (78% and 62%, respectively) versus the 50-mg treatment group (85% and 74%, respectively). The trial determined that tigecycline was efficacious in treating patients with cSSSIs, had a favorable pharmacokinetic profile, and had an acceptable safety profile that justified further investigation with an adequate comparator in phase 3 trials.
As of July 2005, there have been results reported for three phase 3 comparative trials of tigecycline in abstract form only: 1 for patients with cSSSIs and 2 for patients with cIAIs. Integrated results from these phase 3 investigations are also summarized in the tigecycline product labeling.5

The efficacy of tigecycline was compared to imipenem/cilastatin (IMI) in 2 double-blind, phase 3, mulicenter, multinational studies of patients with cIAIs.39,40 In the Tigecycline 306 Study, 817 patients were randomized and received at least 1 dose of study medication (mITT population). A total of 404 patients received 50 mg of tigecycline every 12 hours (after a loading dose of 100 mg) and 413 patients received IMI dosed as per renal function. Patients were well-matched with respect to the most common demographic variables. The most common cIAI was complicated appendicitis. In the microbiologically evaluable (ME) group at the test-of-cure visit, the overall clinical success rate was 91.3% for tigecycline and 89.9% for IMI. Clinical cure rates were also similar when compared across baseline infection diagnoses; for the presence of monomicrobial versus polymicrobial infections; and across the various gram-positive, gram-negative, and anaerobic organisms that were isolated from the infections. The study design was similar for the second phase 3 cIAI investigation (Tigecycline 301 Study), with the exception of a patient stratification step based on baseline APACHE II score (stratification by APACHE II score of <15 or 16 to <31).18 A total of 825 patients received at least 1 dose of study medication and comprised the mITT population. The clinical cure rates at test-of-cure for microbiologically evaluable patients (n=502) were 80.6% for tigecycline-treated patients and 82.4% for IMI-treated patients. In summary, tigecycline was shown to be both safe and efficacious in the treatment of patients with cIAIs as compared to patients treated with IMI.
INDICATIONS AND USAGE
Based on data submitted to FDA, tigecycline was approved for the treatment of cSSSIs caused by susceptible strains of E coli, vancomycin-susceptible E faecalis, methicillin-susceptible and -resistant S aureus, S agalactiae, S anginosus group, S pyogenes, and B fragilis.5 It was approved for treatment of cIAIs caused by susceptible strains of C freundii, E cloacae, E coli, K oxytoca, K pneumoniae, vancomycin-susceptible E faecalis, methicillin-susceptible S aureus, S anginosus group, B fragilis, B thetaiotaomicron, B uniformis, B vulgatus, C perfringens, and Peptostreptococcus micros.
SAFETY AND ADVERSE EFFECTS

The gastrointestinal adverse effects of tigecycline are hypothesized to result from excess serotonin release. Although the administration of ondansetron (a serotonin antagonist antiemetic) appeared to reduce this adverse effect in healthy normal volunteers, there were no documented differences in the concentrations of the serotonin metabolite 5-hydroxyindoleacetic acid for subjects that experienced nausea and/or vomiting after tigecycline infusions versus subjects that received a placebo infusion.34
Although elevated serum transaminases and serum alkaline phosphatase considered possibly or probably related to tigecycline were reported in early clinical investigations of the drug, the incidence of these laboratory abnormalities was approximately 5% for both tigecycline- and comparator-treated patients.5 Increased amylase, bilirubin, or BUN occurred in 3.1%, 2.3%, and 2.1% of tigecycline-treated patients as compared with 1.4%, 0.9%, and 0.2% of comparator-treated patients.5 Rash and/or pruritis occurred in 2.6% and 2.4% of tigecycline-treated patients versus 4.1% and 4.1% of comparator-treated patients.5
As with other tetracycline antimicrobials, tigecycline is contraindicated in pregnant women and in patients aged ≤8 years due to documented effects on ossification and because of tooth/bone discoloration.5 Tigecycline is considered Pregnancy Category D and should be used in pregnant women only if the potential benefits of the drug outweigh the potential risks. Its safety and effectiveness in pediatric patients aged <18 years is unknown. Other tetracycline class-specific adverse effects such as photosensitivity, pseudotumor cerebri, pancreatitis, and anti-anabolic activity may occur with tigecycline but were not well-documented during clinical investigations with the drug.5
DRUG INTERACTIONS

Tigecycline did not inhibit metabolism mediated by the 1A2, 2C8, 2C9, 2C19, 2D6, and 3A4 isozymes of cyctochrome P450 during in vitro human liver microsome experiments.5 The concomitant administration of tigecycline (100 mg followed by 50 mg every 12 hours) and warfarin (25 mg, single-dose) was evaluated in healthy subjects.5 The clearances of R- and S-warfarin were decreased by 40% and 23%, respectively. The R- and S-warfarin Cmax and AUC were both increased (by 38%/43% and 68%/29%, respectively). The effects of warfarin on the international normalized ratio (INR) were unchanged, but monitoring of suitable anticoagulation tests is recommended for patients on concomitant warfarin and tigecycline.5
DOSAGE AND ADMINISTRATION
Tigecycline should be administered as an initial 100-mg loading dose followed by a maintenance dose of 50 mg every 12 hours. Each dose should be infused over 30 to 60 minutes.5 The recommended duration of therapy is 5 to 14 days and should be determined based on the severity of the infection and the patient's response to therapy. Tigecycline comes in single-dose 5-mL glass vials that contain 50 mg of lyophilized powder. Reconstitution of the powder with 5.3 mL of 0.9% sodium chloride or 5% dextrose will result in a 10 mg/mL solution that is yellow-to-orange in color.5 Concentrations of tigecycline in IV infusion bags should not exceed 1 mg/mL. The reconstituted drug can be stored for up to 6 hours at room temperature or up to 24 hours if refrigerated.5 Tigecycline can be administered through a Y-site, but the line should be flushed with either 0.9% sodium chloride or 5% dextrose before and after drug infusion. The drug is compatible for Y-site infusion with dobutamine, dopamine, lidocaine, potassium chloride, ranitidine, and theophylline; infusion through the same Y-site is not recommended with amphotericin B, chlorpromazine, methylprednisolone, or voriconazole.5
In each issue, the "Focus on" feature reviews a newly approved or investigational drug of interest to pharmacy and therapeutics committee members. The column is coordinated by Robert A. Quercia, MS, RPh, director of Drug Information Services at Hartford Hospital in Hartford, Conn, and adjunct associate professor, University of Connecticut School of Pharmacy, Storrs, Conn; and by Craig I. Coleman, PharmD, assistant professor of pharmacy practice, University of Connecticut School of Pharmacy, and director, Pharmacoeconomics and Outcomes Studies Group, Hartford Hospital.
EDITORS' NOTE: The clinical information provided in "Focus on" articles is as current as possible. Due to regularly emerging data on developmental or newly approved drug therapies, articles include information published or presented and available to the author up until the time of the manuscript submission.
REFERENCES
1. Wisplinghoff H, Bischoff T, Tallent SM, Seifert H, Wenzel RP, Edmond MP. Nosocomial bloodstream infections in US hospitals: analysis of 24,179 cases from a prospective nationwide surveillance study. Clin Infectious Dis. 2004;39: 309–317.
2. NNIS. National Nosocomial Infections Surveillance (NNIS) System Report, data summary from January 1992 through June 2003, issued August 2003. Am J Infect Control. 2003;31: 481–498.
3. Neuhauser MM, Weinstein RA, Rydman R, Danziger LH, Karam G, Quinn JP. Antibiotic resistance among gram-negative bacilli in US intensive care units: implications for fluoroquinolone use. JAMA. 2003;289:885–888.
4. Testa RT, Petersen PJ, Jacobus NV, Sum PE, Lee VJ, Tally FP. In vitro and in vivo antibacterial activities of the glycylcyclines, a new class of semisynthetic tetracyclines. Antimicrob Agents Chemother. 1993;37:2270-2277.
5. Tygacil [package insert]. Philadelphia, Penn: Wyeth Pharmaceuticals; 2005.
6. Bergeron J, Ammirati M, Danley D, James L, Norcia M, Retsema J, et al. Glycylcyclines bind to the high-affinity tetracycline ribosomal binding site and evade Tet(M)- and Tet(O)-mediated ribosomal protection. Antimicrob Agents Chemother. 1996;40:2226-2228.
7. Bauer G, Berens C, Projan SJ, Hillen W. Comparison of tetracycline and tigecycline binding to ribosomes mapped by dimethylsulphate and drug-directed Fe2+ cleavage of 16S rRNA. J Antimicrob Chemother. 2004;53:592–599.
8. Petersen PJ, Jacobus NV, Weiss WJ, Sum PE, Testa RT. In vitro and in vivo antibacterial activities of a novel glycylcycline, the 9-t-butylglycylamido derivative of minocycline (GAR-936). Antimicrob Agents Chemother. 1999;43:738-744.
9. Fritsche TR, Kirby JT, Jones RN. In vitro activity of tigecycline (GAR-936) tested against 11,859 recent clinical isolates associated with community-acquired respiratory tract and gram-positive cutaneous infections. Diagn Microbiol Infect Dis. 2004;49:201–209.
10. LaPlante KL, Rybak MJ. Clinical glycopeptide-intermediate staphylococci tested against arbekacin, daptomycin, and tigecycline. Diagn Microbiol Infect Dis. 2004;50:125–130.
11. Stevens T, Johnson B, Johnson J, Bouchillon S, Hoban D, Gaylord C, McCarthy M, Dowzicky M. Tigecycline evaluation surveillance trial (T.E.S.T)––in vitro antibacterial activity against methicillin resistant and methicillin sensitive Staphylococcus aureus isolates [abstract]. Presented at: the 44th Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC); October 30–November 2, 2004; Washington, DC. Abstract E-2061.
12. Stevens T, Johnson B, Johnson J, et al. Tigecycline Evaluation Surveillance Trial (T.E.S.T)––in vitro antibacterial activity against selected species of enterobacteriaceae [abstract]. Presented at: the 44th Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC); October 30–November 2, 2004; Washington, DC. Abstract E-2060.
13. Wallace RJ Jr, Brown-Elliott BA, Crist CJ, Mann L, Wilson RW. Comparison of the in vitro activity of the glycylcycline tigecycline (formerly GAR-936) with those of tetracycline, minocycline, and doxycycline against isolates of nontuberculous mycobacteria. Antimicrob Agents Chemother. 2002;46:3164–3167.
14. Rhomberg PR, Jones RN. In vitro activity of 11 antimicrobial agents, including gatifloxacin and GAR936, tested against clinical isolates of Mycobacterium marinum. Diagn Microbiol Infect Dis. 2002;42:145–147.
15. Edelstein PH, Weiss WJ, Edelstein MA. Activities of tigecycline (GAR-936) against Legionella pneumophila in vitro and in guinea pigs with L pneumophila pneumonia. Antimicrob Agents Chemother. 2003; 47:533–540.
16. Rhomberg PR, Jones RN. Reproducibility assessment of tigecycline MIC results by broth microdilution methods using commercially prepared dry-form panels. Diagn Microbiol Infect Dis. 2005;52:67–69.
17. Projan SJ. Preclinical pharmacology of GAR-936, a novel glycylcycline antibacterial agent. Pharmacotherapy. 2000;20:219S–223S; discussion 224S–228S.
18. Fluit AC, Florijn A, Verhoef J, Milatovic D. Presence of tetracycline resistance determinants and susceptibility to tigecycline and minocycline. Antimicrob Agents Chemother. 2005;49:1636–1638.
19. Dean CR, Visalli MA, Projan SJ, Sum PE, Bradford PA. Efflux-mediated resistance to tigecycline (GAR-936) in Pseudomonas aeruginosa PAO1. Antimicrob Agents Chemother. 2003;47: 972–978.
20. Ruzin A, Visalli MA, Keeney D, Bradford PA. Influence of transcriptional activator RamA on expression of multidrug efflux pump AcrAB and tigecycline susceptibility in Klebsiella pneumoniae. Antimicrob Agents Chemother. 2005;49:1017–1022.
21. Ruzin A, Keeney D, Bradford PA. AcrAB efflux pump plays a role in decreased susceptibility to tigecycline in Morganella morganii. Antimicrob Agents Chemother. 2005;49:791-793.
22. Hirata T, Saito A, Nishino K, Tamura N, Yamaguchi A. Effects of efflux transporter genes on susceptibility of Escherichia coli to tigecycline (GAR-936). Antimicrob Agents Chemother. 2004;48:2179–2184.
23. Visalli MA, Murphy E, Projan SJ, Bradford PA. AcrAB multidrug efflux pump is associated with reduced levels of susceptibility to tigecycline (GAR-936) in Proteus mirabilis. Antimicrob Agents Chemother. 2003;47:665–669.
24. McAleese F, Petersen P, Ruzin A, Dunman PM, Murphy E, Projan SJ, et al. A novel MATE family efflux pump contributes to the reduced susceptibility of laboratory-derived Staphylococcus aureus mutants to tigecycline. Antimicrob Agents Chemother. 2005;49:1865–1871.
25. van Ogtrop ML, Andes D, Stamstad TJ, Conklin B, Weiss WJ, Craig WA, et al. In vivo pharmacodynamic activities of two glycylcyclines (GAR-936 and WAY 152,288) against various gram-positive and gram-negative bacteria. Antimicrob Agents Chemother. 2000;44:943-949.
26. Nannini EC, Pai SR, Singh KV, Murray BE. Activity of tigecycline (GAR-936), a novel glycylcycline, against Enterococci in the mouse peritonitis model. Antimicrob Agents Chemother. 2003;47:529–532.
27. Lefort A, Lafaurie M, Massias L, et al. Activity and diffusion of tigecycline (GAR-936) in experimental enterococcal endocarditis. Antimicrob Agents Chemother. 2003;47:216–222.
28. Murphy TM, Deitz JM, Petersen PJ, Mikels SM, Weiss WJ. Therapeutic efficacy of GAR-936, a novel glycylcycline, in a rat model of experimental endocarditis. Antimicrob Agents Chemother. 2000;44:3022–3027.
29. Yin LY, Lazzarini L, Li F, Stevens CM, Calhoun JH. Comparative evaluation of tigecycline and vancomycin, with and without rifampicin, in the treatment of methicillin-resistant Staphylococcus aureus experimental osteomyelitis in a rabbit model. J Antimicrob Chemother. 2005; 55(6): 995–1002.
30. Labthavikul P, Petersen PJ, Bradford PA. In vitro activity of tigecycline against Staphylococcus epidermidis growing in an adherent-cell biofilm model. Antimicrob Agents Chemother. 2003;47: 3967–3969.
31. Mercier RC, Kennedy C, Meadows C. Antimicrobial activity of tigecycline (GAR-936) against Enterococcus faecium and Staphylococcus aureus used alone and in combination. Pharmacotherapy. 2002;22:1517–1523.
32. Huang V, Rybak MJ. Pharmacodynamics of cefepime alone and in combination with various antimicrobials against methicillin-resistant Staphylococcus aureus in an in vitro pharmacodynamic infection model. Antimicrob Agents Chemother. 2005;49:302–308.
33. Postier RG, Green SL, Klein SR, Ellis-Grosse EJ, Loh E. Results of a multicenter, randomized, open-label efficacy and safety study of two doses of tigecycline for complicated skin and skin-structure infections in hospitalized patients. Clin Ther. 2004;26:704–714.
34. Muralidharan G, Micalizzi M, Speth J, Raible D, Troy S. Pharmacokinetics of tigecycline after single and multiple doses in healthy subjects. Antimicrob Agents Chemother. 2005;49:220–229.
35. Sun HK, Ong CT, Umer A, Harper D, Troy S, Nightingale CH, et al. Pharmacokinetic profile of tigecycline in serum and skin blister fluid of healthy subjects after multiple intravenous administrations. Antimicrob Agents Chemother. 2005;49:1629-1632.
36. Conte JE Jr, Golden JA, Kelly MG, Zurlinden E. Steady-state serum and intrapulmonary pharmacokinetics and pharmacodynamics of tigecycline. Int J Antimicrob Agents. 2005;25:523–529.
37. Zimmerman JJ, Harper D, Matschke K, Speth J, Raible DG, Fruncillo RJ. Pharmacokinetics and pharmacodynamics of tigecycline and digoxin coadministered to healthy men [abstract]. Presented at: the 44th Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC); October 30–November 2, 2004; Washington, DC. Abstract A-460.
38. Dartois N, for the Tigecycline 305 cSSSI Study group. Results of a phase 3, double-blind, safety and efficacy study comparing tigecycline with vancomycin/aztreonam to treat complicated skin and skin structure infections [abstract]. Presented at: the 44th Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC); October 30–November 2, 2004; Washington, DC. Abstract L-986.
39. Dartois N, Gioud-Paquet M, Ellis-Grosse EJ, Loh E, for the Tigecycline 306 Study group. Tigecycline vs. imipenem/cilastatin for treatment of complicated intra-abdominal infections [abstract]. Presented at: the 44th Interscience Conference on Antimicrobial Agents and Chemotherapy (ICAAC); October 30–November 2, 2004; Washington, DC. Abstract L-992c.
40. Ellis-Grosse EJ, Loh, E. Tigecycline compared with imipenem/cilastatin in the treatment of complicated intra-abdominal infections [abstract]. Presented at: the 15th European Congress of Clinical Microbiology and Infectious Diseases (ECCMID); April 2–5, 2005; Copenhagen, Denmark. Abstract P1317.
41. Muralidharan G, Fruncillo RJ, Micalizzi M, Raible DG, Troy SM. Effects of age and sex on single-dose pharmacokinetics of tigecycline in healthy subjects. Antimicrob Agents Chemother. 2005;49:1656-1659.
Employers Face Barriers With Adopting Biosimilars
March 1st 2022Despite the promise of savings billions of dollars in the United States, adoption of biosimilars has been slow. A roundtable discussion among employers highlighted some of the barriers, including formulary design and drug pricing and rebates.
