- Safety & Recalls
- Regulatory Updates
- Drug Coverage
- COPD
- Cardiovascular
- Obstetrics-Gynecology & Women's Health
- Ophthalmology
- Clinical Pharmacology
- Pediatrics
- Urology
- Pharmacy
- Idiopathic Pulmonary Fibrosis
- Diabetes and Endocrinology
- Allergy, Immunology, and ENT
- Musculoskeletal/Rheumatology
- Respiratory
- Psychiatry and Behavioral Health
- Dermatology
- Oncology
Update on the treatment of benign prostatic hyperplasia
Benign prostatic hyperplasia (BPH) is a common chronic disease, with the incidence of BPH increasing with age. Histopathologic features are present in almost half of all men aged >60 years. The disease can have a profoundly negative impact on patients' quality of life, often causing them to limit or avoid basic activities of daily living. Many patients who develop BPH will seek treatment for bothersome lower urinary tract symptoms (LUTS).
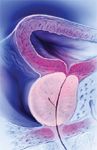
Benign prostatic hyperplasia (BPH) is a common chronic disease that affects approximately 37 million men aged >50 years.1 Histopathologic disease, which is characterized by hyperplastic stromal and epithelial cells within the prostate, can be observed in approximately 50% of men aged >60 years and up to 90% of men by the age of 85.2
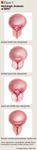
Although the incidence of BPH increases with age, it is important to note that there is often a poor correlation between the extent of histopathologic disease and the degree of clinical symptom presentation.3 Furthermore, in the absence of well-defined morbidity and mortality data, the long-term sequelae associated with the natural progression of BPH remain unclear. Despite recent clinical studies that have demonstrated a reduced risk of disease progression in patients treated with medical therapy, the incidence of many BPH-related complications (eg, retention, incontinence) in untreated patients remains low.4 Therefore, treatment initiation is often dictated by the presence and severity of urinary symptoms, which usually correspond with a perceived poor quality of life.
In August 2003, the American Urological Association (AUA) published its Guideline on the Management of Benign Prostatic Hyperplasia, an update of the 1994 report by the Agency for Healthcare Research and Quality (AHRQ).2 The revised guideline addresses current treatment options for patients with BPH, with a focus on evidence-based clinical literature. This review examines the benefits and potential risks associated with medical, minimally invasive, and surgical treatment approaches for patients diagnosed with symptomatic BPH.
DIAGNOSIS A thorough medical history, which includes a complete physical examination and digital rectal examination (DRE), is essential when considering a diagnosis of BPH. Several other conditions, such as urethral strictures, bladder cancer, prostatitis, urinary tract infection, and bladder stones, can cause also cause LUTS. These possibilities should all be carefully considered before making a diagnosis of BPH. In a patient with a typical LUTS presentation suggestive of BPH, a DRE can help identify an enlarged prostate while simultaneously allowing the physician to exclude the possibility of other pathologies, such as prostate cancer and prostatitis.
A urinalysis should be performed and evaluated for the presence of abnormal findings such as hematuria or bacteria accompanied by white blood cells, which may suggest another pathology. Routine serum prostate-specific antigen (PSA) monitoring as part of the initial evaluation of suspected BPH is not recommended in all patients, although several studies have shown a relationship between elevated baseline PSA levels, prostate volume, and disease progression.5,6 According to the AUA guideline, PSA levels should be monitored in: those patients who will most likely be living in 10 years and for whom a diagnosis of prostate cancer would alter treatment of the disease; or patients in whom a known PSA value may affect the decision regarding various treatment options.2
Finally, because the incidence of renal dysfunction in patients with BPH is not significantly higher than in patients without BPH, routine monitoring of serum creatinine as part of the initial medical examination is no longer recommended.2
SYMPTOM SEVERITY AND QUALITY OF LIFE MEASURES Prior to the development of standardized questionnaires that are now used to quantify symptoms and subsequent response to therapy, physicians would rely primarily on qualitative patient feedback (symptoms are better or worse, etc) to assess treatment efficacy. The AUA Symptom Index (AUASI) and International Prostate Symptom Score (IPSS) are now considered the gold standard measurement tools for the assessment of BPH symptoms and response to treatment, both in a clinical practice setting and as an outcome measure in randomized controlled studies.2 The AUASI is a standardized, validated, and reliable self-administered questionnaire that utilizes 7 questions to assess the frequency and severity of a patient's obstructive and irritative symptoms, with each question scored on a scale of 0 (not at all) to 5 (almost always). Total scores on the AUASI can range from 0-35; a score of 7 or less represents patients with mild symptoms; 8-19 represents those with moderate symptoms; and patients with scores from 20-35 typically have severe symptoms.2 The IPSS uses the same 7 questions as the AUASI, with an additional question that is designed to address the degree of "bother" associated with the patient's urinary symptoms (measured on a scale of 0 to 6-delighted to terrible).
The BPH Impact Index (BPHII) is a validated, self-administered 4-item questionnaire that may be used in combination with the AUASI/IPSS to further examine the relationship between urinary symptom severity and the associated impact on the patient's activities of daily living and quality of life. For example, because of the frequent and sometimes urgent need to urinate, patients often report that they avoid activities that were once enjoyable due to the lack of convenient access to restroom facilities.7 Total scores for the BPHII can range from 0-13, with higher scores indicating more severe symptoms and poorer quality of life.
GENERAL TREATMENT APPROACHES AND OUTCOME ASSESSMENT The primary goal in the treatment of BPH is to reduce the frequency and severity of LUTS, minimize complications (eg, acute urinary retention, renal insufficiency), decrease the incidence of adverse events, and improve the quality of life in patients with symptomatic disease. Treatment of BPH is usually considered upon presentation and subsequent evaluation of a patient's urinary symptoms. A strategy of watchful waiting is indicated for patients with mild (AUASI/IPSS ≤7) or moderate or severe symptoms (≥8) who feel that these symptoms do not significantly interfere with daily activities.2
By utilizing this approach, these patients can avoid the potential for medical- and surgical-related adverse events while the physician continues to closely monitor them for worsening urinary symptoms that interfere with daily activities.
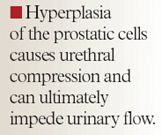
Many randomized controlled clinical trials involving these treatment strategies utilize outcome measures such as the AUASI/IPSS, peak urinary flow rate (Qmax), BPHII, and the IPSS "bother" score as an assessment of treatment efficacy and associated quality of life improvement. Although not commonly utilized in most clinical trials due to the low occurrence of BPH complications, outcome measures of clinical disease progression such as acute urinary retention, renal insufficiency, recurrent urinary tract infection, or urinary incontinence also have been studied.4,8,9 The incidence and severity of treatment-associated adverse events also are commonly assessed and reported in BPH clinical trials.
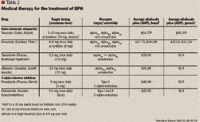
Alpha-adrenergic antagonists. The alpha-adrenergic receptor antagonists function to relieve bladder outlet obstruction by blocking the alpha-adrenergic receptors that cause smooth muscle contraction within the prostate and bladder neck.10 As a result, patients often report obstructive and irritative symptom improvements (usually within 1-2 weeks) due to the relaxation of the bladder neck and subsequent increased urinary flow rate.
Alpha-adrenergic antagonists are classified as first-, second-, and third-generation agents, based on their specificity for the prostate and bladder neck. Three subtypes of alpha1 receptors have been identified: alpha1a, alpha1b, and alpha1d. The alpha1a-receptor subtype is predominantly found within the prostatic tissue, whereas the alpha1b and alpha1d receptors primarily regulate contraction of vascular smooth muscle. First-generation agents (eg, phenoxybenzamine [Dibenzyline, Wellspring Pharmaceuticals]), which are non-specific for the alpha1 receptors, are not indicated for the treatment of BPH due to the profound vasodilatory side effects associated with their use.
The second-generation alpha-adrenergic antagonists include prazosin (Minipress, Pfizer), terazosin (Hytrin, Abbott), and doxazosin (Cardura, Pfizer). These agents are considered non-uroselective because they have affinity for all 3 alpha1-receptor subtypes. Prazosin is not FDA approved for the treatment of BPH, and it is not recommended for treating the condition due to potential adverse events such as hypotension and syncope. Terazosin and doxazosin, both of which are also indicated for the treatment of hypertension, require careful dosing titration due to their ability to significantly decrease blood pressure. The median dose of terazosin in clinical studies is between 6-7 mg, while target dosing of doxazosin for most patients is between 4-8 mg.2 Compared to the newer agents for the treatment of BPH, second-generation agents are less expensive because they are available in generic formulations.
The third-generation agents, which include tamsulosin (Flomax, Boehringer Ingelheim) and the recently approved drug alfuzosin (Uroxatral, Sanofi-Aventis), are considered to be uroselective. Unlike second-generation agents, tamsulosin exhibits a high degree of specificity for the alpha1a receptors in the prostate, while alfuzosin has been noted to concentrate within prostatic tissues.10,11 Due to the uroselective nature of these drugs, dosing titration is not necessary. The typical starting and target dose of tamsulosin is 0.4 mg per day, although there are a small number of patients who are titrated to 0.8 mg per day as a result of their worsening symptoms. The initial, target, and maximal recommended dose of alfuzosin is 10 mg once daily.
The alpha-adrenergic antagonists are considered to be equally effective for improving LUTS in clinical trials of patients with BPH.2 Patients treated with these agents, on average, have reported a noticeable 4- to 6-point decrease in their AUASI/IPSS scores with associated improvements in their Qmax, BPHII, and IPSS "bother" scores in studies lasting from 13 weeks to 9 months.12-14 The durability of alpha-blockers has also been confirmed in a recently completed 4-year open-label study that demonstrated sustained, long-term benefits in AUASI scores and Qmax in patients who continued therapy with tamsulosin for up to 6 years.15
Adverse events associated with the alpha-adrenergic antagonists are primarily related to the drug's target receptor subtype. The most commonly reported adverse events associated with the non-uroselective agents terazosin and doxazosin are dizziness (15%-26%), asthenia (10%-14%), and postural hypotension (5%-8%), due to their effects on systemic vascular smooth muscle alpha1 receptors.16 Compared to the non-selective agents, the incidence of vasodilatory side effects with the newer uroselective agents is relatively low, with postural hypotension occurring in approximately 3% of tamsulosin patients and less than 1% of alfuzosin treated patients.17 However, asthenia and dizziness have been reported in a higher percentage of tamsulosin- (7%-8%) and alfuzosin- (3%-4%) treated patients compared with those who received placebo.
A relatively low incidence of sexual side effects, such as decreased libido (3%-3.5%) and impotence (4%-6%), has been reported with all alpha-adrenergic antagonists. The incidence of ejaculatory dysfunction, defined as loss of ejaculation/decreased ejaculatory volume, has been reported more frequently in tamsulosin-treated patients (8.4%-18.1%) compared to those receiving other alpha-adrenergic antagonists.2,18 Other reported side effects with these drugs include nasal congestion and upper-respiratory tract complaints such as rhinitis, sinusitis, pharyngitis, laryngitis, and infection.
Doxazosin may not be the most appropriate agent to manage BPH symptoms in patients with hypertension, based on results from the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT), a randomized double-blind, controlled trial in 24,335 hypertensive patients.19,20 Patients who received doxazosin experienced a significantly higher risk of stroke (RR, 1.26; 95% CI, 1.10-1.46; P=.001) and combined cardiovascular disease (RR, 1.20; 95% CI, 1.13-1.27; P<.001), compared with those treated with the diuretic chlorthaladone.19 These results prompted the discontinuation of the doxazosin arm of the study in February 2000.
In January 2005, The American Society of Cataract and Refractive Surgery issued a physician advisory regarding the development of Intraoperative Floppy Iris Syndrome (IFIS), a condition that could potentially lead to intraoperative complications in cataract surgery patients who are taking tamsulosin. Although FDA has not issued an advisory statement on IFIS to date, as safety data continue to emerge, patients who are undergoing cataract surgery should advise their physician if they are being treated with tamsulosin.21,22
5-alpha-reductase inhibitors. The androgens testosterone and dihydrotestosterone are believed to play a critical role in the pathogenesis of BPH.8 The 5-alpha-reductase inhibitors improve LUTS and increase the Qmax by essentially blocking the conversion of testosterone to dihydrotestosterone (DHT), the primary androgen found within the prostate. By inhibiting the formation of dihydrotestosterone, prostate volume is reduced and bladder outlet obstruction is relieved.8 Finasteride (Proscar, Merck) and dutasteride (Avodart, GlaxoSmithKline) are currently the only 5-alpha-reductase inhibitors available for the treatment of BPH.
There are 2 types of 5-alpha-reductase, type I and type II. Finasteride selectively inhibits type II 5-alpha-reductase, which is found in the prostate, seminal vesicles, epididymides, and liver. Type II 5-alpha-reductase is responsible for up to two-thirds of circulating DHT. The recommended dosing for finasteride is 5 mg once a day. Dutasteride inhibits type I (primarily found in the skin and liver) and type II 5-alpha-reductase, thereby relieving symptoms and reducing bladder outlet obstruction. The recommended dosing for dutasteride is 0.5 mg once daily.
Compared to the alpha-adrenergic antagonists, the 5-alpha-reductase inhibitors are associated with a slower onset of symptom improvement (6-12 months or more), and most clinical trials have demonstrated more modest reported reductions in AUASI/IPSS (3.5-4.5 point decrease), Qmax, BPH Impact Index, and IPSS "bother" scores.2,8,16 However, results from a pooled analysis of 2 multicenter, randomized, placebo-controlled studies involving dutasteride-treated patients, each of 2 years' duration, followed by a 2-year open-label extension period, demonstrated AUASI score reductions comparable to studies involving alpha-adrenergic antagonists.23 The mean AUASI score change in the placebo/dutasteride group was -5.3 points and -6.1 points in the dutasteride/dutasteride group at 48 months. In addition to AUASI/IPSS, Qmax, and quality of life score improvements, several studies have also shown that the 5-alpha-reductase inhibitors reduce the risk of urinary retention and the need for BPH surgery.4,5,9
Adverse effects associated with the 5-alpha-reductase inhibitors primarily include problems with sexual function. The most commonly reported side effects with these drugs include: impotence (4%-7%); decreased libido (1%-8%); abnormal ejaculation (2%-4%); and gynecomastia (1%-2%). Although reports of breast cancer have been identified in a relatively small number of finasteride-treated patients as compared to placebo, currently available data do not support a link between the use of the drug and the risk of developing breast cancer.2
Combination therapy. The rationale for using combination therapy with an alpha-adrenergic antagonist and a 5-alpha-reductase inhibitor is to improve LUTS and reduce the overall risk of disease progression.
Two large-scale randomized, placebo-controlled trials evaluated the safety and efficacy of an alpha-adrenergic antagonist, a 5-alpha-reductase inhibitor, and the combination of both drugs in patients with BPH. 12,16 Both studies demonstrated that combination therapy was not superior to alpha-adrenergic antagonist monotherapy for decreasing LUTS; however, statistically significant symptom score improvements were observed in each group, compared with the 5-alpha-reductase and placebo groups. Although the 5-alpha-reductase inhibitor did decrease urinary symptoms, no statistically significant difference in symptom improvement was noted between the drug and placebo groups. The duration of both studies was 1 year, and neither study was designed to assess the effects of these agents on the overall risk of disease progression.
The Medical Therapy of Prostatic Symptoms (MTOPS) study was the first long-term, randomized, double-blind, placebo-controlled trial that assessed both symptomatic improvement and overall clinical disease progression in patients receiving an alpha-adrenergic antagonist, 5-alpha-reductase inhibitor, or combination therapy with both these agents. 4 Overall clinical progression was defined as worsening of symptoms, acute urinary retention, incontinence, urinary tract infection, or renal insufficiency. Results at the end of the 4-year study showed that combination therapy with doxazosin and finasteride reduced the overall risk of clinical progression by 66% (95% CI, 54-76; P<.001 vs placebo), compared to doxazosin (risk reduction 39%; 95% CI, 20-53; P=.002) and finasteride (risk reduction 34%; 95% CI, 14-50; P=.002). Combination therapy was also demonstrated to be significantly better than either drug alone at decreasing the AUASI, increasing the Qmax, and reducing the risk of clinical progression, primarily by preventing symptomatic deterioration. Although most adverse events were reported with similar frequency between treatment groups, erectile dysfunction and dizziness were reported more frequently in patients receiving both doxazosin and finasteride.
Phytotherapeutic therapy.Despite the lack of adequate evidenced-based literature and general support for their use, many BPH patients report taking phytotherapeutic agents, with an estimated $1 billion spent annually on these products in the United States alone.24 While the exact mechanism of action of these agents is not clear, it has been suggested that symptomatic improvement may be due to anti-inflammatory effects, 5-alpha-reductase inhibition, or growth factor alteration.24,25
The most commonly studied phytotherapeutic agents include Serenoa repens (saw palmetto, permixon), Pygeum africanum (African plum), beta-sitosterols such as Hypoxis rooperi (South African star grass), and those developed from the rye grass pollen, Secale cereale (cernilton). Systematic reviews and study meta-analyses have shown that several of these agents may be effective for improving LUTS. 25-27 Although most trials have only studied phytotherapeutic agents in comparison to placebo, results from a 12-month, double-blind, randomized controlled trial in Europe (N=811) found that tamsulosin and Permixon were equally effective in reducing IPSS scores.28
Although the results from several clinical trials have demonstrated symptomatic improvements, it is important to note that many of the studies involving phytotherapeutic agents contained small numbers of patients, and many lacked standardized, validated outcome measures such as the AUASI/IPSS. Furthermore, since many of these agents are derived from plant extracts, their actual composition, inter-product variability, and stability may be inaccurate or unknown.24,29 In addition, concerns over variability between product batches often raise questions regarding the safety and efficacy of these drugs. For these reasons, phytotherapeutic agents and other dietary supplements are not currently recommended by the AUA practice guidelines committee for the management of BPH.2
MINIMALLY INVASIVE TREATMENT OPTIONS Minimally invasive therapy (MIT) has become an attractive treatment option and surgical alternative, typically for those patients who have attempted and failed medical therapy. The goal of MIT is to induce necrosis of urethral prostate tissue, thereby relieving bladder outlet obstruction and decreasing urinary symptoms. Although there are several different types of MIT procedures available, most use thermotherapy devices that employ a heat source such as microwave, radio-frequency, laser, water, or high-intensity focused ultrasound to destroy the prostatic tissue that is causing the urethral obstruction.30
There are several transurethral microwave thermotherapy (TUMT) devices available (eg, Prostatron, Targis, Core-Therm, Thermatrx), all of which utilize catheters that deliver microwave energy to the prostate with safeguards such as surface cooling and temperature feedback mechanisms to minimize damage to non-target areas.30 Although some devices utilize slightly different microwave frequencies, a typical TUMT procedure can be performed with topical anesthesia in the office setting in less than 1 hour. In studies from 12 weeks through 3 years, patients who received TUMT have shown marked and sustained improvements in AUASI/IPSS scores (43%-76%) from baseline.31-34 In addition, Qmax and associated quality of life improvements have also been noted in several studies.33-35
Transurethral needle ablation (TUNA) devices (eg, Precision) utilize radio frequency energy that is delivered through a catheter that contains 2 insulated needles inserted directly into the prostatic tissue to cause coagulation necrosis. Because the extent of necrosis from the tip of the needles is less with TUNA, it is considered most effective in patients with a prostate of 60 g or less or in those with predominant lateral lobe involvement.2,30,36 Results from a 12-month randomized controlled trial involving 133 patients who received TUNA therapy showed statistically significant AUASI score improvements of >50% and an increase in Qmax from baseline.37 Furthermore, in a recently published 5-year follow-up study, improvements in AUASI scores and quality of life were maintained in patients who received TUNA therapy.38
Using a method similar to the TUNA procedure, interstitial laser coagulation (ILC) delivers radiation directly into prostatic tissue through a cystoscope that contains a laser fiber, thereby causing localized tissue necrosis.30,36 Results from a 2-year study of patients treated with either ILC or transurethral resection of the prostate (TURP) showed similar symptom score improvements, Qmax rates, and quality of life measures, despite a 16% re-treatment rate in the ILC group.39
Water-induced thermotherapy, a relatively new MIT procedure, uses the process of conductive heating to circulate hot water (140 degrees Fahrenheit) for 45 minutes through a catheter containing an expandable balloon that is placed in the prostatic urethra.30,36,40 The insulated catheter allows the non-target tissues to be protected while coagulation necrosis of the object prostatic tissue occurs. In a study evaluating 125 patients who received this procedure, IPSS scores were significantly improved (50%, P<.01) compared to baseline 12 months after the treatment period.40 In addition, Qmax and quality of life scores were also significantly improved at 12 months.
High-intensity focused ultrasound (HIFU) therapy uses low-energy ultrasonic waves, delivered via a transrectal probe, to produce very high local temperatures (176-212 degrees Fahrenheit), which cause instant coagulation necrosis of selected prostatic tissue.30,36,41 In a clinical trial evaluating 22 patients treated with a newer HIFU device compared with 35 who were treated with the prototype model, statistically significant improvements in IPSS symptoms and quality of life scores were observed in both groups at 12 months after treatment as compared to baseline.41 Although both treatments were considered efficacious compared to baseline, Qmax improvements were significantly better in the group treated with the newer HIFU device.
Most MIT procedures are generally well tolerated, with most patients reporting a progressive resolution of symptoms over the following several weeks. To minimize urinary retention, many patients will require catheter placement for 1 to 2 weeks or until they achieve resolution of symptoms and are able to void spontaneously. Other treatment-associated adverse events, which vary by procedure, include: post-procedure irritation; urinary retention (6%-23%); urinary tract infection (9%-17%); erectile dysfunction (1%-3%); loss of ejaculation/decreased ejaculatory volume (4%-16%); and treatment failure requiring re-treatment (10%-40%).2,34,42 Although the reported incidence of re-treatment associated with MIT is higher than surgical therapy, results from a 3-year randomized study comparing TUMT and TURP showed a similar re-treatment rate between both groups (19.8%, 95% CI, 10.4%-29.3% vs 12.9%, 95% CI, 4.5%-21.3%, respectively; P=.28).34 It should be noted that the number of patients undergoing re-treatment in the study was relatively small (14 in the TUMT group and 8 in the TURP group).
SURGICAL TREATMENT OPTIONS Although several newer treatment approaches for the surgical management of BPH patients have emerged, TURP remains the gold standard due to the extensive published experience and long-term follow-up data associated with the procedure.2 Other surgical options include: transurethral electrovaporization and incision of the prostate; laser vaporization; resection/enucleation; and open simple prostatectomy.
Based on the results from a multitude of randomized clinical trials, the surgical treatment options mentioned are all considered effective, with marked and sustained improvements in AUASI/IPSS scores (up to 85% from baseline), Qmax, and quality of life, compared to watchful waiting.2,34 However, unlike MIT, surgical options such as TURP are performed in the inpatient setting and require a hospital stay. The duration of hospitalization can vary based on the surgical procedure; a longer length of stay has been associated with the TURP procedure (48-50 h) compared to patients who receive holmium laser enucleation of the prostate (26-27 h).43,44
Adverse events associated with the surgical therapies include post-procedure irritative symptoms, acute urinary retention, urinary incontinence, bladder neck contracture (BNC)/urethral stricture, and sexual dysfunction.2 Although BNC/stricture can occur with either therapy, the reported incidence is higher in patients who have received a surgical procedure (3%-8%) compared to an MIT procedure (1%-3%). Sexual side effects, including sexual dysfunction (3%-13%) and ejaculatory dysfunction (17%-65%), were also more frequently associated with the surgical procedures compared to MIT.2,34 In addition, re-treatment rates or the need for a second procedure have been reported with variable frequency between the surgical therapies (1%-14%).2
TREATMENT RECOMMENDATIONS According to the 2003 AUA Guideline on Management of Benign Prostatic Hyperplasia, the treatment of a patient with BPH should be based on a thorough medical history, physical examination, and evaluation of urinary symptoms.2 For patients with mild symptoms (AUASI ≤7) or patients who are not bothered by moderate-to-severe symptoms (AUASI ≥8), a trial of watchful waiting is indicated.2 Although patients will not receive active therapy during the watchful waiting period, they should be closely followed by a physician and re-examined annually. Patients who present with complications of BPH (ie, bladder calculi, recurrent UTI, refractory gross hematuria, refractory retention, renal insufficiency) usually require surgical intervention, regardless of AUASI score.

Minimally invasive and surgical procedures are considered for BPH treatment in patients who have failed or decided against medical therapy, or who have developed intolerable side effects. Although MIT may be less efficacious and durable than surgery, it is more effective than medical therapy in reducing LUTS. MIT is often an attractive alternative to surgical therapy because most procedures can be performed in the physician's office with a local anesthetic, usually in fewer than 60 minutes. Furthermore, side effects and treatment complications associated with MIT are much less frequent compared to surgical procedures. Surgical treatment options may be considered in patients who have developed BPH-related complications and in those who have decided against or failed MIT. Currently, TURP is the gold standard of therapy and is considered the most effective treatment for the management of BPH.
Dr Douglass is an assistant clinical specialist at Northeastern University School of Pharmacy in Boston, Mass, and a clinical pharmacist in adult internal medicine at Boston Medical Center. He can be reached at m.douglass@neu.edu
. Dr Lin is a urologist at Mass Bay Urologic Associates in Milton, Mass.
Disclosures: Dr Douglass is a member of the speakers' bureau for Sanofi-Aventis, and Dr Lin is a member of the speakers’ bureau for GlaxoSmithKline.
REFERENCES 1. Benign prostatic hyperplasia. Sanofi-Aventis, Inc, 2004 (data on file).
2. AUA Practice Guidelines Committee. AUA Guideline on Management of Benign Prostatic Hyperplasia (2003). Chapter 1: Diagnosis and treatment recommendations. J Urol. 2003;170:530-547.
3. Roehrborn CG, McConnell JD. Etiology, pathophysiology, epidemiology, and natural history of benign prostatic hyperplasia. In: Walsh PC, Retik AB, Vaughan ED, Wein AJ, eds. Campbell's Urology. 8th ed. Philadelphia, Pa: WB Saunders Co; 2002:1297-1336.
4. McConnell JD, Roehrborn CG, Bautista OM, et al, and the Medical Therapy of Prostatic Symptoms (MTOPS) Research Group. The long-term effect of doxazosin, finasteride, and combination therapy on the clinical progression of benign prostatic hyperplasia. N Engl J Med. 2003;349:2387-2398.
5. Roehrborn CG, McConnell JD, Lieber M, et al, and the PLESS Study Group. Serum prostate-specific antigen concentration is a powerful predictor of acute urinary retention and need for surgery in men with clinical benign prostatic hyperplasia. Urology. 1999;53:473-480.
6. Roehrborn CG, Boyle P, Bergner D, et al, and the PLESS Study Group. Serum prostate-specific antigen and prostate volume predict long-term changes in symptoms and flow rate: results of a four-year, randomized trial comparing finasteride versus placebo. Urology. 1999;54:662-669.
7. Garraway WM, Russell EB, Lee RJ, et al. Impact of previously unrecognized benign prostatic hyperplasia on the daily activities of middle-aged and elderly men. Br J Gen Pract. 1993;43:318-321.
8. Roehrborn CG, Boyle P, Nickel JC, Hoefner K, Andriole G, and the ARIA3001 ARIA3002 and ARIA3003 Study Investigators. Efficacy and safety of a dual inhibitor of 5-alpha-reductase types 1 and 2 (dutasteride) in men with benign prostatic hyperplasia. Urology. 2002;60:434-441.
9. McConnell JD, Bruskewitz R, Walsh P, et al, and the Finasteride Long-Term Efficacy and Safety Study Group. The effect of finasteride on the risk of acute urinary retention and the need for surgical treatment among men with benign prostatic hyperplasia. N Engl J Med. 1998;338:557-563.
10. Hoffman BB. Catecholamines, sympathomimetic drugs, and adrenergic receptor antagonists. In: Hardman JG, Limbird LE, eds. Goodman & Gilman's The Pharmacological Basis of Therapeutics. 10th ed. New York, NY: McGraw-Hill; 2001:215-268.
11. Uroxatral [package insert]. New York, NY: Sanofi-Synthelabo Inc; 2003.
12. Lepor H, Williford WO, Barry MJ, et al, and the Veterans Affairs Cooperative Studies Benign Prostatic Hyperplasia Study Group. The efficacy of terazosin, finasteride, or both in benign prostatic hyperplasia. N Engl J Med. 1996;335:533-539.
13. Lepor H. Phase III multicenter placebo-controlled study of tamsulosin in benign prostatic hyperplasia. Tamsulosin Investigator Group. Urology. 1998;51:892-900.
14. Roehrborn CG. Efficacy and safety of once-daily alfuzosin in the treatment of lower urinary tract symptoms and clinical benign prostatic hyperplasia: a randomized, placebo-controlled trial. Urology. 2001;58:953-959.
15. Narayan P, Evans C, Moon T. Long-term safety and efficacy of tamsulosin for the treatmentof lower urinary tract symptoms associated with benign prostatic hyperplasia. J Urol. 2003;170:498-502.
16. Kirby R, Roehrborn CG, Boyle P. Efficacy and tolerability of doxazosin and finasteride, alone or in combination, in treatment of symptomatic benign prostatic hyperplasia: the Prospective European Doxazosin and Combination Therapy (PREDICT) trial. Urology. 2003;61:119-126.
17. Roehrborn CG. Alfuzosin: overview of pharmacokinetics, safety, and efficacy of a clinically uroselective alpha-blocker. Urology. 2001;58(6 Suppl 1):55-63.
18. Flomax [package insert]. Ridgefield, CT: Boehringer Ingelheim Pharmaceuticals Inc; 1999.
19. ALLHAT Collaborative Research Group. Diuretic versus alpha-blocker as first-step antihypertensive therapy: final results from the Antihypertensive and Lipid-Lowering Treatment to Prevent Heart Attack Trial (ALLHAT). Hypertension. 2003;42:239-246.
20. ALLHAT Collaborative Research Group. Major cardiovascular events in hypertensive patients randomized to doxazosin vs chlorthalidone: the antihypertensive and lipid-lowering treatment to prevent heart attack trial (ALLHAT). JAMA. 2000;283:1967-1975.
21. Chang DF, Campbell JR. Perspectives in lens & IOL surgery surgeons report-a new small pupil syndrome caused by Flomax. Eyeworld. 2005;10:63-64.
22. American Society of Cataract & Refractive Surgery website. ASCRS issues physician advisory on Flomax: evidence suggests link between drug and intraoperative iris complications [press release]. Available at: http:// http://www.ascrs.org/advocacy/pressrelease_011305.htm. Accessed February 1, 2005.
23. Roehrborn CG, Marks LS, Fenter T, et al. Efficacy and safety of dutasteride in the four-year treatment of men with benign prostatic hyperplasia. Urology. 2004;63:709-715.
24. Lowe FC. Phytotherapy in the management of benign prostatic hyperplasia. Urology. 2001;58(6 Suppl 1):71-77.
25. Lowe FC, Fagelman E. Phytotherapy in the treatment of benign prostatic hyperplasia: an update. Urology. 1999;53:671-678.
26. Wilt T, Ishani A, MacDonald, R. Serenoa repens for benign prostatic hyperplasia. Cochrane Database of Systematic Reviews [database online]. Chichester, UK: John Wiley & Sons, Ltd; 2004. Updated 2004.
27. Boyle P, Robertson C, Lowe F, Roehrborn C. Updated meta-analysis of clinical trials of Serenoa repens extract in the treatment of symptomatic benign prostatic hyperplasia. BJU Int. 2004;93:751-756.
28. Debruyne F, Koch G, Boyle P, et. al. [Comparison of a phytotherapeutic agent (Permixon) with an alpha-blocker (Tamsulosin) in the treatment of benign prostatic hyperplasia: a 1-year randomized international study]. [Article in French]. Prog Urol. 2002;12:384-392.
29. De Smet PA. Herbal remedies. N Engl J Med. 2002;347:2046-2056.
30. Larson BT, Bostwick DG, Corica AG, Larson TR. Histological changes of minimally invasive procedures for the treatment of benign prostatic hyperplasia and prostate cancer: clinical implications. J Urol. 2003;170:12-19.
31. Larson TR, Blute ML, Bruskewitz RC, Mayer RD, Ugarte RR, Utz WJ. A high-efficiency microwave thermoablation system for the treatment of benign prostatic hyperplasia: results of a randomized, sham-controlled, prospective, double-blind, multicenter clinical trial. Urology. 1998;51:731-742.
32. D'Ancona FC, Francisca EA, Witjes WP, Welling L, Debruyne FM, De La Rosette JJ. Transurethral resection of the prostate vs high-energy thermotherapy of the prostate in patients with benign prostatic hyperplasia: long-term results. Br J Urol. 1998;81:259-264.
33. Wagrell L, Schelin S, Nordling J, et al. Feedback microwave thermotherapy versus TURP for clinical BPH--a randomized controlled multicenter study. Urology. 2002;60:292-299.
34. Floratos DL, Kiemeney LA, Rossi C, Kortmann BB, Debruyne FM, de La Rosette JJ. Long-term followup of randomized transurethral microwave thermotherapy versus transurethral prostatic resection study. J Urol. 2001;165:1533-1538.
35. Roehrborn CG, Preminger G, Newhall P, et al. Microwave thermotherapy for benign prostatic hyperplasia with the Dornier Urowave: results of a randomized, double-blind, multicenter, sham-controlled trial. Urology. 1998;51:19-28.
36. Blute ML, Larson T. Minimally invasive therapies for benign prostatic hyperplasia. Urology. 2001;58(6 Suppl 1):33-41.
37. Roehrborn CG, Issa MM, Bruskewitz RC, et al. Transurethral needle ablation for benign prostatic hyperplasia: 12-month results of a prospective, multicenter U.S. study. Urology. 1998;51:415-421.
38. Zlotta AR, Giannakopoulos X, Maehlum O, Ostrem T, Schulman CC. Long-term evaluation of transurethral needle ablation of the prostate (TUNA) for treatment of symptomatic benign prostatic hyperplasia: clinical outcome up to five years from three centers. Eur Urol. 2003;44:89-93.
39. Kursh ED, Concepcion R, Chan S, Hudson P, Ratner M, Eyre R. Interstitial laser coagulation versus transurethral prostate resection for treating benign prostatic obstruction: a randomized trial with 2-year follow-up. Urology. 2003;61:573-578.
40. Muschter R, Schorsch I, Danielli L, et. al. Transurethral water-induced thermotherapy for the treatment of benign prostatic hyperplasia: a prospective multicenter clinical trial. J Urol. 2000;164:1565-1569.
41. Uchida T, Muramoto M, Kyunou H, Iwamura M, Egawa S, Koshiba K. Clinical outcome of high-intensity focused ultrasound for treating benign prostatic hyperplasia: preliminary report. Urology. 1998;52:66-71.
42. Laguna MP, Alivizatos G, De La Rosette JJ. Interstitial laser coagulation treatment of benign prostatic hyperplasia: is it to be recommended? J Endourol. 2003;17:595-600.
43. Tan AH, Gilling PJ, Kennett KM, Westenberg AM, Fraundorfer MR. A randomized trial comparing holmium laser enucleation of the prostate with transurethral resection of the prostate for the treatment of bladder outlet obstruction secondary to benign prostatic hyperplasia in large glands (40 to 200 grams). J Urol. 2003;170(4 Pt 1):1270-1274.
44. Gilling PJ, Cass CB, Cresswell MD, Fraundorfer MR. Holmium laser resection of the prostate: preliminary results of a new method for the treatment of benign prostatic hyperplasia. Urology. 1996;47:48-51.
45. Lacy CF, Armstrong LL, Goldman MP, Lance LL, eds. Drug Information Handbook. 11th ed. Hudson, Ohio: Lexi-Comp, Inc: 2003.
46. Clinical Pharmacology resources page. Clinical Pharmacology website. Available at: http:// http://www.cponline.gsm.com/. Accessed January 13, 2005.
47. Drug Topics Redbook 2004. Montvale, NJ: Thomson Healthcare; 2004.
48. Hoffman BB. Catecholamines, Sympathomimetic Drugs, and Adrenergic Receptor Antagonists. In: Hardman JG, Limbird LE, eds. Goodman and Gillman's Pharmacological Basis of Therapeutics. 10th ed. New York, NY: McGraw-Hill: 2001: 215-267.
49. Snyder PJ. Androgens. In: Hardman JG, Limbird LE, eds. Goodman and Gillman's Pharmacological Basis of Therapeutics. 10th ed. New York, NY: McGraw-Hill: 2001:1635-1638.
50. Hytrin [package insert]. North Chicago, Ill: Abbott Laboratories; 2001.
51. Cardura [package insert]. New York, NY: Pfizer; 2002.
52. Proscar [package insert]. Whitehouse Station, NJ: Merck; 2004.
53. Avodart [package insert]. Research Triangle Park, NC: GlaxoSmithKline; 2002.
54. Randall A. Surgical Pathology of Prostatic Obstruction. Baltimore, Md: Williams & Wilkins: 1931.
Employers Face Barriers With Adopting Biosimilars
March 1st 2022Despite the promise of savings billions of dollars in the United States, adoption of biosimilars has been slow. A roundtable discussion among employers highlighted some of the barriers, including formulary design and drug pricing and rebates.
